Explore Antibodies Drug Market Companies and Trends with Growth

- AbbVie, Inc.
- ADC Therapeutics
- AstraZeneca
- Genentech, Inc.
- Genmab A/S
- Gilead Sciences, Inc.
- GlaxoSmithKline
- GO Therapeutics, Inc.
- Hiedelberg Pharma A/G
- ImmunoGen, Inc.
- Pfizer, Inc.
- Takeda Pharmaceutical Company
- Tallac Therapeutics, Inc.
Antibody Drug Market Overview
Monoclonal Antibodies (mAbs), which seem to be antibody preparations designed to bind to a single target, have shown potential in the fight against cancer as well as autoimmune diseases.
Monoclonal antibodies are protein-based therapies or drugs that are increasingly being used to treat chronic diseases. This blog examines the global market for antibody drug products and provides an update on their applications in various disease areas. The overall market for antibody drug products is divided into four major application areas: autoimmune diseases, solid tumors, lymphoma, and leukemia, as well as other diseases such as asthma, osteoporosis, and cardiovascular disease.
The FDA has approved more than 100 antibody products, and biologics account for roughly one-fifth of all new drug approvals each year. The presence of a healthy antibody therapy pipeline increases the company's chances of achieving higher market revenue. As a result of the high demand for antibody therapy, a surge in the development of new antibody products is expected to drive the antibody therapy market share.
Which Was The First Antibody-Drug?
Orthoclone OKT3 (muromonab-CD3) was the first licensed monoclonal antibody, approved in 1986 for use in preventing kidney transplant rejection. It is a mouse monoclonal IgG2a antibody with the cognate antigen CD3. It works by binding and inhibiting the effects of CD3 on T-lymphocytes. However, due to reported side effects, its use was restricted to acute cases (e.g. human anti-mouse antibody response).
Market Growth
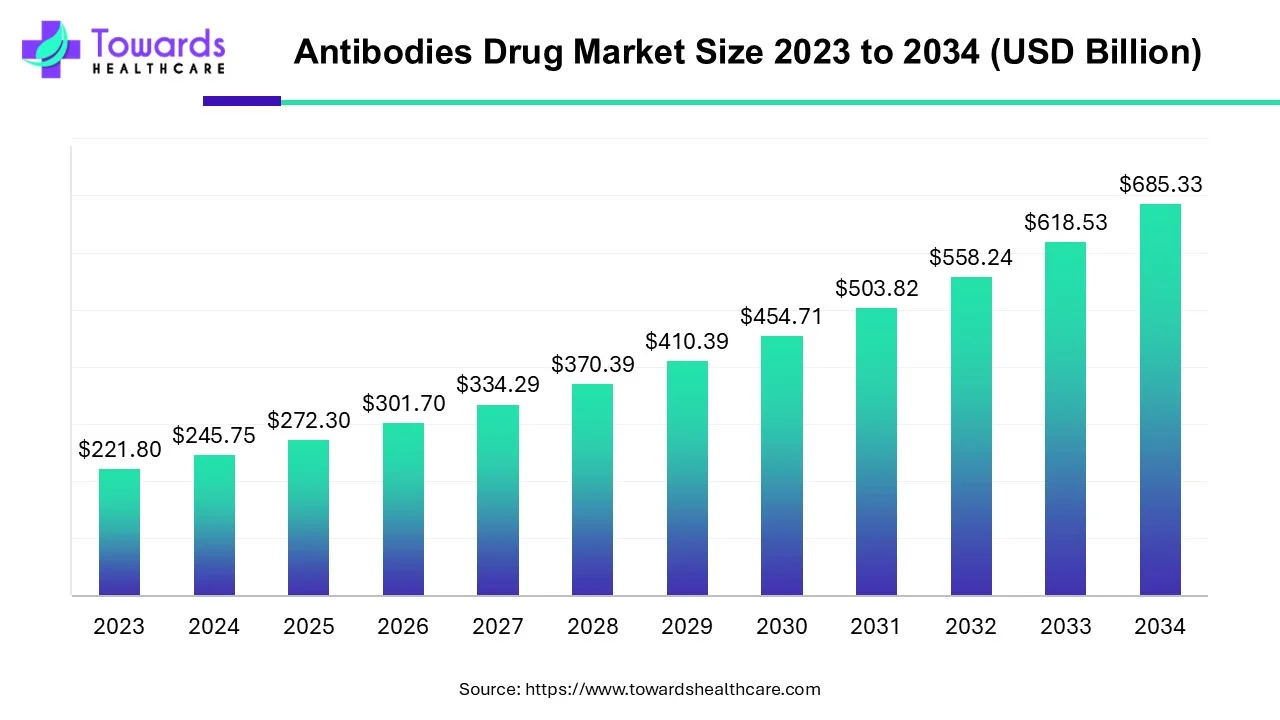
The global antibodies drug market size was calculated at USD 245.75 billion in 2024, to reach USD 272.3 billion in 2025 is expected to be worth USD 685.33 billion by 2034, expanding at a CAGR of 10.8% from 2024 to 2034.
Companies are currently funding clinical trials for over 570 mAbs. Approximately 90% of these are early-stage studies designed to assess patient populations' safety (Phase I) or safety and preliminary efficacy (Phase I/II or Phase II). The majority of mAbs in Phase I (70%) are for cancer treatment, and the proportions of mAbs in Phase II and late-stage clinical studies (pivotal Phase II, Phase II/III, or Phase III) are similar.
Antibodies Drug Market Trends
- In January 2025, Akeso, Inc. announced that it received NMPA NDA approval for its internally developed IL-17-targeting monoclonal antibody gumokimab (AK111) for the treatment of moderate-to-severe plaque psoriasis. Gumokimab was evaluated in four clinical studies, including one pivotal Phase III trial.
- In October 2024, Lonza announced the extension of its collaboration with a major biopharmaceutical firm for commercial-scale manufacture of antibody-drug conjugates (ADCs). The extended collaboration leads to the construction of a new bioconjugation suite, providing monoclonal antibody manufacturing services.
- In April 2024, the U.S. FDA approved mirvetuximab soravtansine-gynx (Elahere) for the treatment of advanced, platinum-resistant ovarian cancer. The antibody was developed by Oklahoma Health Sciences Center, and approval was obtained based on the MIRASOL trial.
Latest Announcement by Industry Leaders
Wyatt McDonnell, CEO and CO-founder of Infinimmune, commented in an interview that the company aims to become the single source of a safer and better generation of antibody therapeutics. He said that the company is currently building a pipeline of drugs and partnering with pharmaceutical companies to advance antibody programs together.
Recent Developments
- In March 2025, Masayuki Kobayashi, President and Representative Director of Taiho Pharmaceutical Co., Ltd., entered into a definitive agreement to acquire Araris Biotech AG, a Swiss biotechnology company developing next-gen antibody drug conjugates for USD 400 million. With the acquisition of the ADC drug discovery technology platform of Araris, along with Cysteinomix, Taiho intends to further expand its ongoing development portfolio in the field of oncology.
- In January 2025, ImmunoPrecise Antibodies Ltd developed a novel class of GLP-1 therapies using AI through its proprietary LENSai platform. The drugs were developed for the treatment of diabetes and obesity, enhancing efficacy, safety, therapy longevity, and patient satisfaction.
- In July 2024, researchers from Stanford University developed a novel ML-based method to predict molecular changes quickly and accurately, leading to better antibody drugs. Large language models are used to find rare and desirable mutations, eliminating the need for exhaustive experiments.
Partner with our experts to explore the Antibodies Drug Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
