Key Players' Offerings in the Cardiac Cannula Market with Supply Chain Analysis 2026

Access a free sample report covering segments, market stats, and competitor insights: Download Now
Company Offerings 2026
| Company | Description |
| Medtronic | It usually provides diverse cardiac cannulae for both adult and pediatric patients. |
| BD (Becton, Dickinson & Company) | A firm explores a comprehensive portfolio of vascular access & specialized cardiac devices. |
| Edwards Lifesciences Corporation | Its offerings comprise ThruPort systems for MIVS, EZ Glide aortic cannulae, Thin-Flex/Trim-Flex venous cannulae, & retrograde cardioplegia catheters. |
| LivaNova PLC | This facilitates a complete portfolio for conventional & minimally invasive cardiac surgery (MICS), coupled with extracorporeal life support (ECLS) |
| Smartcanula LLC | A company specializes in self-expandable venous cannulas to raise blood flow while lowering the insertion site's size. |
| Braile Biomédica | Its portfolio offers sterile, PVC-based, pyrogen-free cardiac cannulae for cardiopulmonary bypass (CPB). |
| Surge Cardiovascular | This primarily provides antegrade/retrograde cardioplegia sets, the ALPINE Femoral Venous Cannula, & PEAK Left Heart Vent Cannula. |
| Cardinal Health | A firm specializes in aortic perfusion cannulas, left ventricular (LV) sump vents, & vascular tourniquet kits. |
| Getinge | Their portfolio emphasizes extracorporeal membrane oxygenation (ECMO) and extracorporeal life support (ECLS). |
| EUROSETS | Its key offering encompasses aortic root, retrograde, venous (single/multi-stage), & pediatric cannulae, along with specialized ECLS femoral cannulae validated for up to 30 days. |
Cardiac Cannula Market- Supply Chain Analysis
R&D
- This prominently covers explaining clinical requirements, material selection, design & simulation, prototyping, and strong testing.
- Key Players: Medtronic, Edwards Lifesciences, Terumo Corporation, etc.
Clinical Trials & Regulatory Approvals
- Leaders are following the evaluation of safety & efficacy, typically leveraging prospective, multi-center, randomized clinical trials with substantial approvals, like FDA Class II/III, CE Mark, and PMDA in Japan.
- Key Players: IPERF SAS, Mayo Clinic, Indonesia University, etc.
Patient Support & Services
- They primarily provide 30-day CentriMag acute support, HeartMate remote monitoring, & specialized catheter training for clinicians to enhance patient results.
- Key Players: Abbott Laboratories, Boston Scientific, LivaNova PLC, etc.
Market Growth 2026-2035
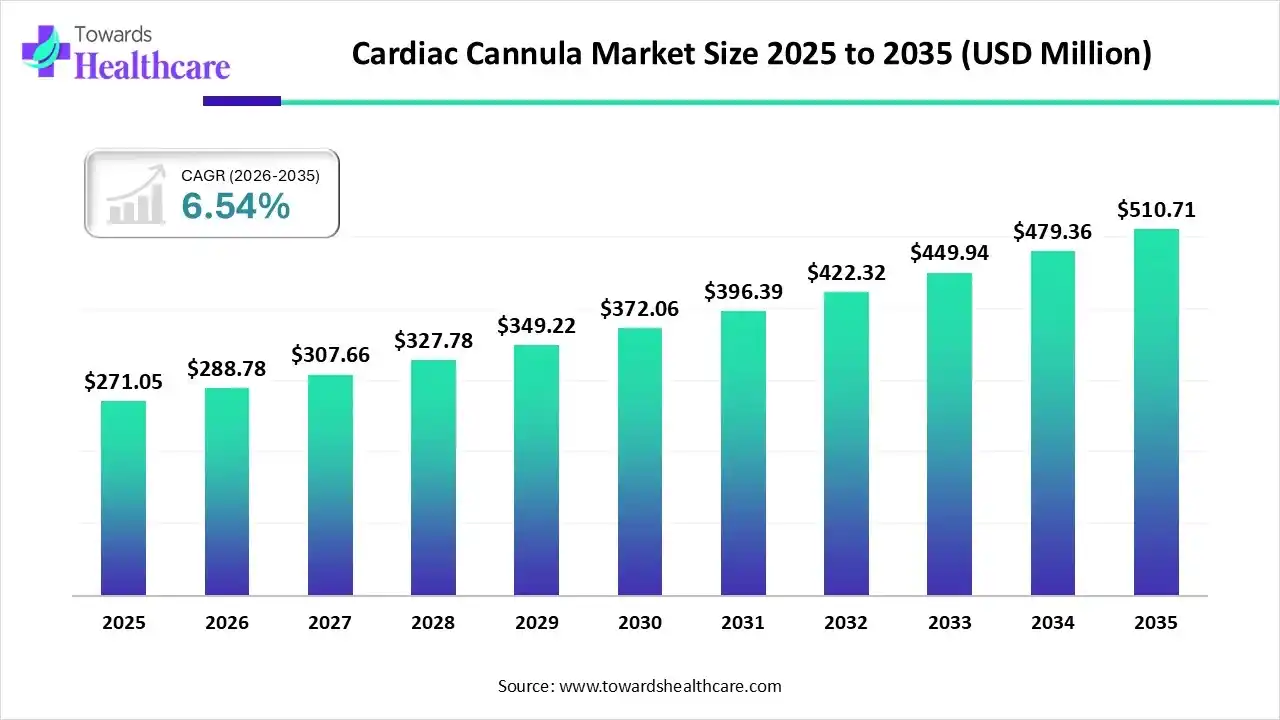
The global cardiac cannula market size was estimated at USD 271.05 million in 2025 and is predicted to increase from USD 288.78 million in 2026 to approximately USD 510.71 million by 2035, expanding at a CAGR of 6.54% from 2026 to 2035.
What are the Key Developments in the Cardiac Cannula Market?
- In October 2025, Cagent Vascular commercially launched the Serranator PTA Serration Balloon Catheter in 7.0 mm and 8.0 mm diameters.
- In August 2025, India unveiled its first motorized TAVR delivery system, the VitaFlow Liberty from MicroPort, which raises outcomes for complex bicuspid aortic valves.
- In May 2025, Abbott introduced the TactiFlex Sensor-Enabled Ablation Catheter, the world’s first ablation catheter, with a flexible tip combined with contact force technology.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
