April 2026

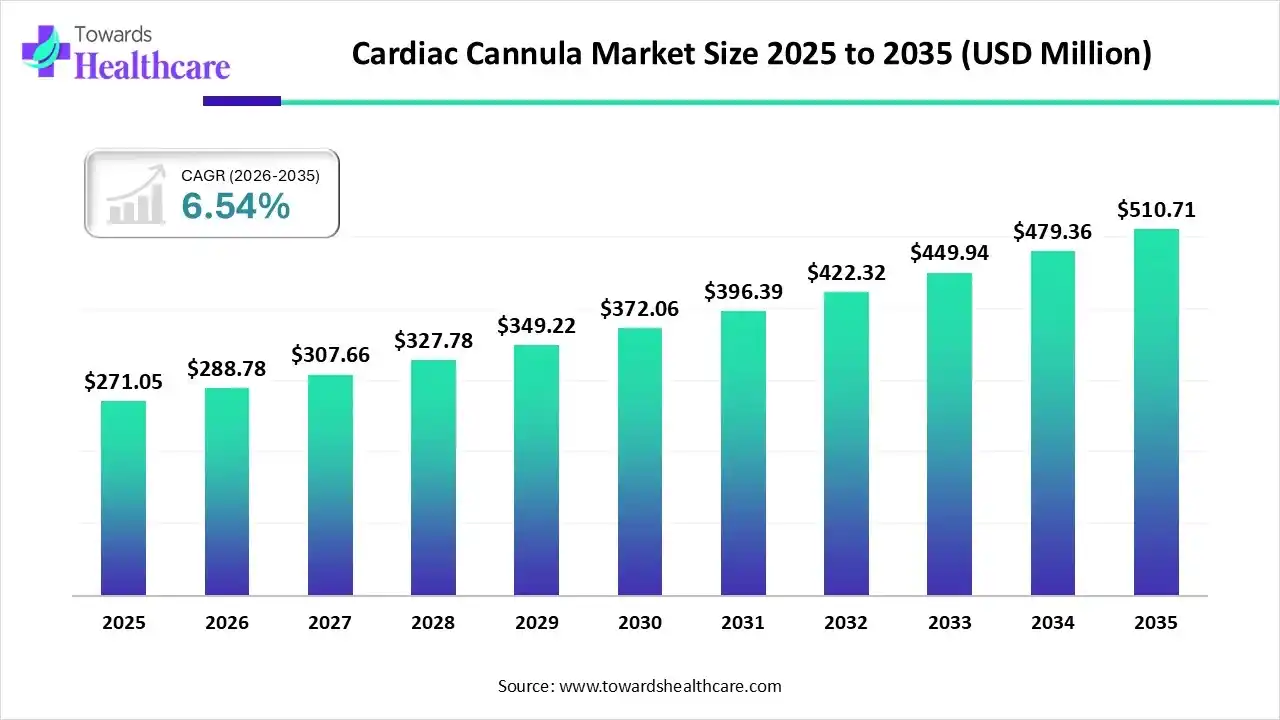
The global cardiac cannula market size was estimated at USD 271.05 million in 2025 and is predicted to increase from USD 288.78 million in 2026 to approximately USD 510.71 million by 2035, expanding at a CAGR of 6.54% from 2026 to 2035.
The worldwide rising cases of CVD, chronic lung diseases, especially in the Asia region is driving the demand for these advanced cannulas. However, researchers are actively investigating biocompatible materials to reduce the arising body issues.
Primarily, the cardiac cannula market covers a thin, flexible tube inserted into veins, arteries/heart chambers to transfer blood to a heart-lung machine or facilitate extracorporeal life support (ECMO) during surgery. To drive overall market progression, the leading firms are emphasizing minimal invasiveness, promoting self-expanding venous SmartCannula designs for robust drainage, anti-thrombogenic coatings, & also 3D-printed, patient-specific prototypes to enhance flow & lower complexity.
An immersive study has shown that a CT-driven, AI-assisted, preprocedural simulation for guiding-catheter selection lowers procedural time from 91.8 to 68.5 minutes, & reduces catheter-related events. Alongside, more sophisticated AI solutions support automatic detection of vessel lumen and boundaries, with high agreement with human experts in plaque analysis. Whereas CorPath GRX is enabling remote navigation, with features like ‘Rotate on Retract’ that improve wire rotation & reduce both procedure time and radiation exposure for clinicians.
The globe is increasingly stepping towards tiny, more flexible cannulae that allow for femoral artery access for cardiopulmonary bypass (CPB) & minimal chest incisions.
An innovative, heparin-coated venous cannulae is featuring combined inflatable balloons to gain full caval occlusion from within, which will lower the need for surgical dissection & snares.
Nowadays, researchers are putting efforts into discovering advanced bioresorbable cannulas that safely degrade within 12–18 months & focuses on omitting the need for removal surgeries & reducing long-term foreign body concerns.
| Table | Scope |
| Market Size in 2026 | USD 288.78 Million |
| Projected Market Size in 2035 | USD 510.71 Million |
| CAGR (2026 - 2035) | 6.54% |
| Leading Region | North America |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product Type, By Age Group, By Size, By Application, By Region |
| Top Key Players | Medtronic, BD (Becton, Dickinson & Company), Edwards Lifesciences Corporation, LivaNova PLC, Smartcanula LLC, Braile Biomédica, Surge Cardiovascular, Cardinal Health, Getinge, EUROSETS |
Which Product Type Led the Cardiac Cannula Market in 2025?
In 2025, the arterial cannulas segment captured a major revenue share of the market. A key driver is a massive rise in cases of cardiac, vascular, and chronic diseases, which need frequent surgeries & continuous, long-term monitoring, and finally raise the demand for reliable arterial cannulas. Prospective studies are focusing on the development of anti-thrombogenic coatings, kink-resistant, flexible polyurethane materials, & low-profile, tapered tips for vessel protection.
Specialty Cannulas
Moreover, the specialty cannulas segment is anticipated to expand fastest in the coming era. Specifically, globally increasing demand for single-use or disposable products to mitigate infections, with a growing need for specialized, kink-resistant designs in complex, long-duration procedures, such as ECMO are impacting the segmental growth. Emerging trends include the recent FDA approval for the SmartFlow Neuro Cannula for targeted, deep-brain delivery of gene therapies (KEBILIDI), and also blunt-tipped microcannulas for dermal fillers and facial volumizing.
How did the Adults Segment Dominate the Cardiac Cannula Market in 2025?
The adults segment led with the largest share of the market in 2025. Across the globe, the 60+ age group is highly affected by CVDs, with greater demand for ECMO/cardiopulmonary bypass, which fuels the demand for advanced cannula during surgeries. For the robust results, they are widely adopting smaller, high-performance, & flexible cannulas for robotic-assisted cardiac surgeries & percutaneous interventions, which further lowers trauma & recovery time.
Pediatric
On the other hand, the pediatric segment is estimated to show rapid growth As per the survey, nearly 1.35 million infants are diagnosed with CHD each year, with 90% in regions with restricted, improper, or no care. To overcome these arising risks, diverse public & private investments in specialized children’s hospitals and cardiac catheterization labs, especially in the Asia-Pacific region.
Which Size Led the Cardiac Cannula Market in 2025?
In 2025, the 11 Fr – 22 Fr segment dominated the market & is predicted to expand at the fastest CAGR. This is mainly propelled by the wider adoption in pediatric, neonatal, & minimally invasive adult procedures, which facilitate optimal venous drainage & arterial perfusion for patients with tiny vessels, typically assisting body weights ranging from 3 to 40+ kg. Further innovations are encouraging the breakthroughs in polyurethane materials, bi-directional flow designs for peripheral access, & modern, wire-reinforced structures to maintain increased flow rates.
Why did the Cardiopulmonary Bypass (CPB) Segment Dominate the Market in 2025?
In 2025, the cardiopulmonary bypass (CPB) segment captured the dominating share of the cardiac cannula market. The use of a cardiac cannula in these instances allows stable, low-resistance, & secure blood flow, with minimal vessel damage & also enables cerebral perfusion in complex cases. The market has been exploring novel designs, including the optiCAN, which possesses a helical outflow & jet-splitting dispersion tip, and emphasizes restoring physiological cerebral flow & lowering embolic concerns.
Extracorporeal Membrane Oxygenation (ECMO)
In the future, the extracorporeal membrane oxygenation (ECMO) segment is anticipated to witness rapid expansion. A particular catalyst is the accelerating incidence of chronic obstructive pulmonary disease (COPD), heart failure, & infections, such as influenza or COVID-19, which demand modern life support. However, researchers are actively focused on implementing ultrasound-guided, percutaneous insertion to minimize vascular complications & allow for awake-ECMO mobilization.
North America dominated the cardiac cannula market in 2025, due to the rising cases of CVD, the geriatric population, and a center of well-established healthcare infrastructure. The region is increasingly leveraging extracorporeal membrane oxygenation (ECMO) for vital care, specifically following the growth in respiratory & cardiac failures.
For instance,
U.S. Market Trends
However, the U.S. market held the largest share, as it is putting efforts into developing novel arterial cannulas that are being united with sensors to offer real-time monitoring of oxygen levels, pressure, & blood flow.
For instance,
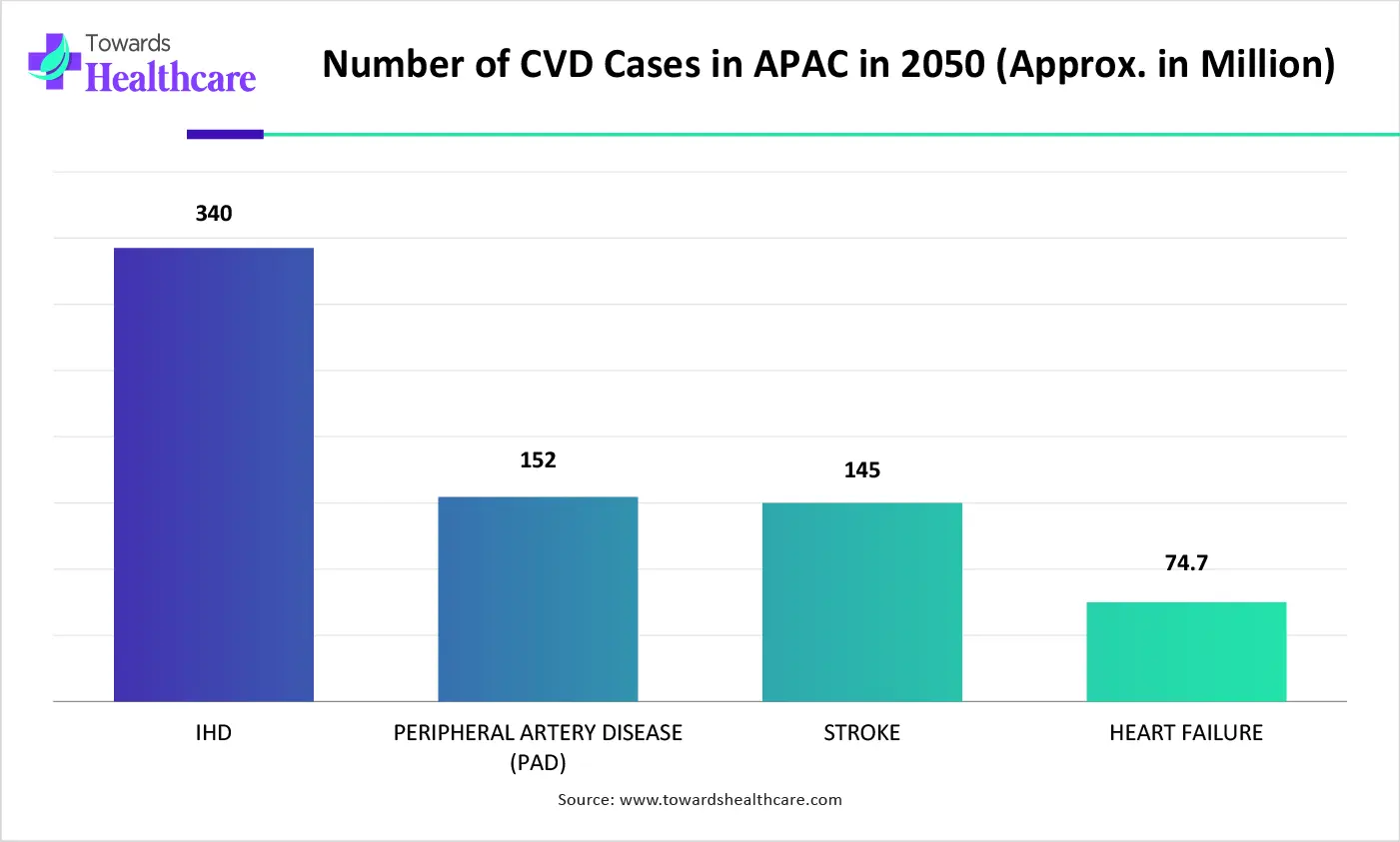
During the prospective period, the Asia Pacific is predicted to witness the fastest expansion in the cardiac cannula market. The region is highly transforming the adoption of minimally invasive techniques, like smaller, specialized cannulas for MICS-CABG & robotic-assisted procedures, with a substantial focus on domestic manufacturing of specialized catheters and valves within China & India.
Especially, many Indian manufacturers, such as Meril Life Sciences, are majorly expanding with locally established, affordable alternatives, like the MyVal THV series for transcatheter valve replacement. India is widely utilizing single-use, high-precision devices to boost patient safety & expedite recovery.

| Company | Description |
| Medtronic | It usually provides diverse cardiac cannulae for both adult and pediatric patients. |
| BD (Becton, Dickinson & Company) | A firm explores a comprehensive portfolio of vascular access & specialized cardiac devices. |
| Edwards Lifesciences Corporation | Its offerings comprise ThruPort systems for MIVS, EZ Glide aortic cannulae, Thin-Flex/Trim-Flex venous cannulae, & retrograde cardioplegia catheters. |
| LivaNova PLC | This facilitates a complete portfolio for conventional & minimally invasive cardiac surgery (MICS), coupled with extracorporeal life support (ECLS) |
| Smartcanula LLC | A company specializes in self-expandable venous cannulas to raise blood flow while lowering the insertion site's size. |
| Braile Biomédica | Its portfolio offers sterile, PVC-based, pyrogen-free cardiac cannulae for cardiopulmonary bypass (CPB). |
| Surge Cardiovascular | This primarily provides antegrade/retrograde cardioplegia sets, the ALPINE Femoral Venous Cannula, & PEAK Left Heart Vent Cannula. |
| Cardinal Health | A firm specializes in aortic perfusion cannulas, left ventricular (LV) sump vents, & vascular tourniquet kits. |
| Getinge | Their portfolio emphasizes extracorporeal membrane oxygenation (ECMO) and extracorporeal life support (ECLS). |
| EUROSETS | Its key offering encompasses aortic root, retrograde, venous (single/multi-stage), & pediatric cannulae, along with specialized ECLS femoral cannulae validated for up to 30 days. |
By Product Type
By Age Group
By Size
By Application
By Region
April 2026
April 2026
April 2026
April 2026