Explore the Top Companies in the Hematuria Treatment Market
- Jabs Biotech Pvt. Ltd.
- Vera Therapeutics
- Apotex
- Bristol Myers Squibb
- Gilead Sciences
- Ardelyx
- Janssen Pharmaceuticals Ltd.
- Shorla Oncology Ltd.
Hematuria Treatment: Redefining Urological Care
The hematuria treatment market is experiencing robust growth, driven by the increasing incidence of urinary tract disorders, biologics expansion, and advanced diagnostic technologies. It encompasses the development, manufacturing, and supply of drugs used for the treatment of hematuria. Hematuria is a condition in which blood is visible in the urine. The most common medications for hematuria treatment include antibiotics, analgesics, and hormone-related medicines.
Market Growth
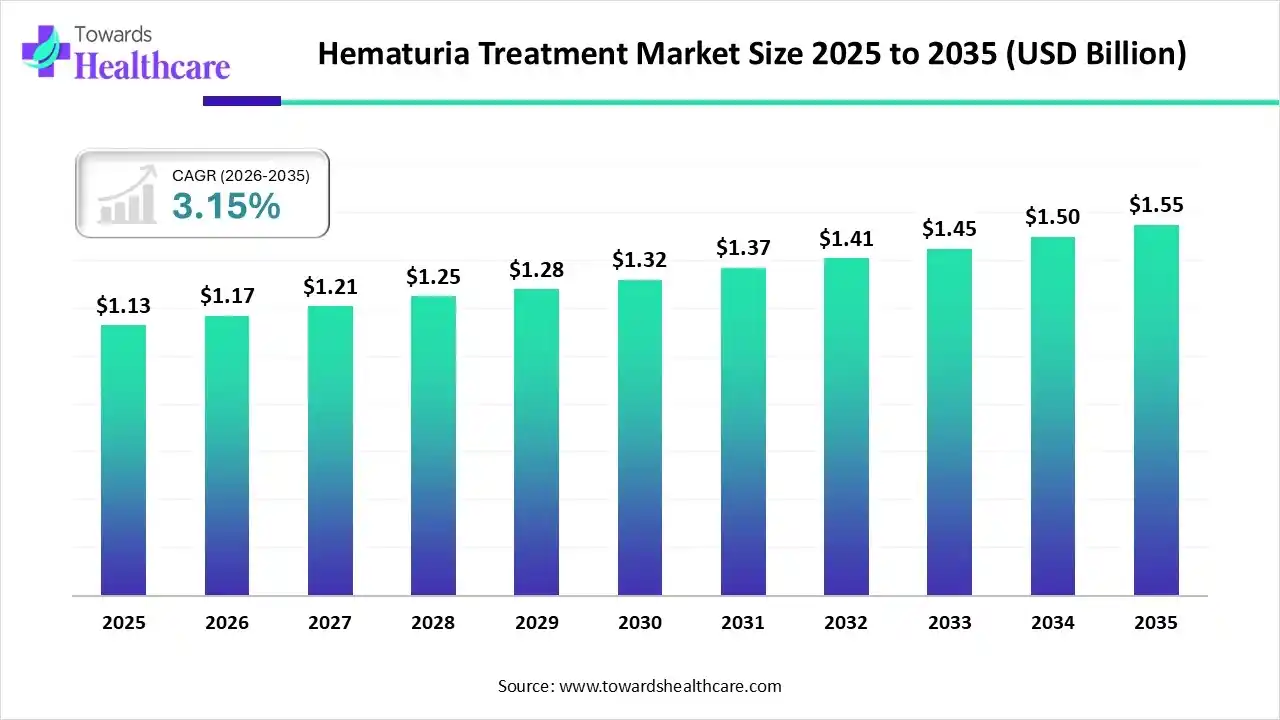
The global hematuria treatment market size is calculated at USD 1.13 billion in 2025, grew to USD 1.17 billion in 2026, and is projected to reach around USD 1.55 billion by 2035. The market is expanding at a CAGR of 3.15% between 2026 and 2035.
Hematuria Treatment Market Outlook
- Industry Growth Overview: The market is accelerating at a rapid rate, driven by the development of more targeted therapies and advanced drug delivery systems. Innovations are also made in interventional radiology techniques like embolization and ablation.
- Startup Ecosystem: The startup ecosystem is maturing, with advancements in therapeutic regimens for hematuria and venture capital investments. Vesica Health and ProArc Medical are some startups that focus on developing diagnostics and therapeutics for urinary tract disorders.
- Global Expansion: Companies expand their geographical presence by collaborating with other companies, enabling them to distribute their proprietary products across diverse geographical locations. Major companies invest in opening their manufacturing facilities in various regions.
Top Companies & Their Offerings in the Hematuria Treatment Market
Novartis AG
• Headquarters: Basel, Switzerland
• Offering: Fabhalta
Cipla Ltd.
• Headquarters: Mumbai, India
• Offering: Mesna
UroGen Pharma Ltd.
• Headquarters: New Jersey, United States
• Offering: ZUSDURI (mitomycin)
Ferring Pharmaceuticals
• Headquarters: Saint-Prex, Switzerland
• Offering: Adstiladrin
Johnson & Johnson
• Headquarters: New Jersey, United States
• Offering: INLEXZO (gemcitabine)
Company Landscape
Novartis AG
Company Overview
- Global healthcare company specializing in innovative medicines, generics, and biosimilars.
- Focus on therapeutic areas including oncology, cardiovascular, immunology, and neuroscience.
- A key player in pharmacotherapy for conditions underlying hematuria, such as kidney disease and certain cancers.
Corporate Information (Headquarters, Year Founded, Ownership Type)
- Headquarters: Basel, Switzerland
- Year Founded: 1996 (formed through the merger of Ciba-Geigy and Sandoz)
- Ownership Type: Public (SIX: NOVN, NYSE: NVS)
History and Background
- Established from a historic merger, combining decades of scientific innovation from its predecessor companies.
- Has evolved into a research-focused multinational with a vast portfolio of prescription drugs.
- Its Sandoz division provides generic drugs often used in the pharmacotherapy of hematuria-related conditions.
Key Milestones/Timeline
- 1996: Formation of Novartis AG.
- 2019: Completed spin-off of Alcon (eye care division) to focus entirely on innovative medicines.
- 2023-2024: Continued focus on strategic therapeutic areas, including R&D in kidney health (IgA Nephropathy, a cause of hematuria).
Business Overview
- Operates through two main segments: Innovative Medicines and Sandoz (Generics and Biosimilars).
- High investment in R&D to drive pipeline of novel treatments.
Business Segments/Divisions
- Innovative Medicines: Includes Pharmaceuticals (cardiovascular, renal, metabolism, oncology, immunology).
- Sandoz: Global leader in generic drugs and biosimilars.
Geographic Presence
- Operates globally in approximately 150 countries.
- Strong presence in North America, Europe, and key emerging markets (e.g., China, Brazil).
Key Offerings
- Pharmacotherapies: Drugs for chronic kidney disease (CKD), anti-infectives (for UTIs), and oncology treatments (for bladder/kidney cancer).
- Examples include drugs used to manage symptoms or underlying causes of hematuria.
End-Use Industries Served
- Hospitals and Clinics
- Retail Pharmacies
- Research Institutions
Key Developments and Strategic Initiatives
- Capacity Expansions/Investments: Continuous investment in manufacturing and R&D facilities globally.
- Regulatory Approvals: Ongoing regulatory submissions and approvals for new indications and novel therapies across its portfolio.
Mergers & Acquisitions
- Frequently divests non-core assets to focus on high-growth innovative medicines.
- Acquisitions target complementary innovative technology and pipeline assets.
Partnerships & Collaborations
Extensive collaborations with academic institutions and biotech companies for drug discovery and development, including in areas relevant to hematuria's causes (nephrology, oncology).
Product Launches/Innovations
Focus on launching innovative therapies, including those for chronic kidney disease, an underlying condition that can present with hematuria.
Distribution Channel Strategy
- Direct sales force targeting specialists (Urologists, Nephrologists, Oncologists).
- Distribution networks with wholesalers and retail pharmacies.
- Sandoz leverages a broad global network for generic drug distribution.
Technological Capabilities/R&D Focus
- R&D Focus: Focused on data science, advanced therapy platforms (gene and cell therapy), and next-generation pharmacotherapies.
- Research & Development Infrastructure: Global network of R&D centers and collaborations.
- Innovation Focus Areas: Targeted treatments for complex diseases, including IgA Nephropathy (a cause of hematuria) and renal cell carcinoma.
Competitive Positioning
- Strengths & Differentiators: Strong, diversified portfolio; global footprint; significant R&D spending; leading generics business (Sandoz).
- Market presence & ecosystem role: Dominant market player in pharmacotherapy, setting standards for treatment protocols in nephrology and oncology.
SWOT Analysis
- Strengths: Strong R&D, Diversified product portfolio, Global presence.
- Weaknesses: Patent expirations for key drugs, Regulatory scrutiny.
- Opportunities: Growth in emerging markets, Advancements in cell/gene therapies, Focus on IgAN treatment.
- Threats: Intense competition, Pricing pressures, Healthcare policy changes.
Recent News and Updates
- 2024: Sandoz, now spun off, continues to be a major generic provider impacting the affordability of pharmacotherapies.
- Press Releases: Regular announcements on Phase III trial data for various compounds, including in cardiovascular and renal health, impacting hematuria-related conditions.
- Industry Recognitions/Awards: Consistently recognized for R&D and pharmaceutical innovation.
Pfizer Inc.
Company Overview
- One of the largest global biopharmaceutical companies focused on discovering, developing, and manufacturing vaccines and medicines for humans.
- Maintains a significant portfolio across multiple therapeutic areas, including internal medicine and oncology, both relevant to hematuria etiology.
Corporate Information (Headquarters, Year Founded, Ownership Type)
- Headquarters: New York, New York, USA
- Year Founded: 1849
- Ownership Type: Public (NYSE: PFE)
History and Background
- Started as a fine chemicals business and grew rapidly through scientific innovation and strategic mergers (e.g., Warner-Lambert, Pharmacia, Wyeth).
- A century-long history of developing breakthrough medicines and vaccines.
Key Milestones/Timeline
- 1849: Founded by Charles Pfizer and Charles Erhart.
- Late 1900s/Early 2000s: Major mergers expanding its global drug portfolio.
- 2024: Continued focus on integrating key acquisitions, particularly in oncology, which includes treatments for genitourinary cancers (a cause of hematuria).
Business Overview
- Focuses on five main therapeutic areas: Oncology, Inflammation & Immunology, Rare Disease, Internal Medicine, and Vaccines.
- Internal Medicine and Oncology segments include drugs used to treat underlying causes of hematuria (e.g., UTIs, bladder cancer, kidney disease).
Business Segments/Divisions
- Pfizer Biopharmaceuticals Group (PGB): Includes all therapeutic areas and the global supply chain.
- Key Therapeutic Areas: Internal Medicine (includes anti-infectives, cardiovascular), Oncology.
Geographic Presence
- Operates in over 125 countries worldwide.
- Dominant market presence in North America and Europe.
Key Offerings
- Pharmacotherapies: Antibiotics/Anti-infectives (for UTIs, a common cause of hematuria), anticoagulants (when medically indicated), and blockbuster oncology treatments.
- Products addressing the treatment of renal cell carcinoma and urothelial carcinoma (bladder cancer).
End-Use Industries Served
- Hospitals and Health Systems
- Ambulatory Care Centers
- Government Agencies and Public Health Programs
Key Developments and Strategic Initiatives
- Mergers & Acquisitions: Completed the acquisition of Seagen in 2023, significantly enhancing its oncology portfolio, particularly in antibody-drug conjugates (ADCs) for cancers like urothelial carcinoma.
- Investments: Major investment in manufacturing capacity and R&D for next-generation medicines.
Mergers & Acquisitions
2023: Acquired Seagen for $43 billion to become a leader in protein-based cancer therapies (ADCs). These therapies target tumors in the urinary tract.
Partnerships & Collaborations
Active R&D collaborations with various biotech and academic partners to expand pipeline, especially in oncology and internal medicine.
Product Launches/Innovations
Continuous development and launch of novel oncology drugs and expanded indications for existing drugs used in internal medicine.
Capacity Expansions/Investments
Ongoing modernization and expansion of global manufacturing sites, including a focus on complex biologic therapies.
Regulatory Approvals
Frequent regulatory approvals for new drugs or expanded uses, particularly in the oncology and internal medicine segments.
Distribution Channel Strategy
- Robust global distribution network leveraging wholesalers, distributors, and direct sales to hospitals and clinics.
- Strong focus on patient access programs and reimbursement support.
Technological Capabilities/R&D Focus
- R&D Focus: Leading-edge R&D in oncology (ADCs, immunotherapies), gene therapy, and mRNA technology.
- Core Technologies/Patents: Extensive patent protection across its portfolio, including formulations and novel molecular entities in anti-infectives and oncology.
- Innovation Focus Areas: Developing precision medicines for cancer and infectious diseases that underlie hematuria.
Competitive Positioning
- Strengths & Differentiators: Massive scale, diversified global presence, one of the world's largest R&D budgets, recent strong pivot to oncology leadership.
- Market presence & ecosystem role: Highly influential global pharmaceutical leader, dictating pharmacotherapy standards in oncology and infectious disease management.
SWOT Analysis
- Strengths: Global brand recognition, Strong financial position, Deep R&D pipeline bolstered by Seagen acquisition.
- Weaknesses: Exposure to patent cliffs, High reliance on a few blockbuster drugs.
- Opportunities: Expansion in oncology and rare diseases, Growth in emerging markets, Use of AI in drug discovery.
- Threats: Generic competition, Complex regulatory landscape, Geopolitical risks.
Recent News and Updates
- Late 2024/2025: Key focus is on the integration of Seagen and leveraging its oncology assets, which directly impact the treatment landscape for cancers associated with hematuria.
- Press Releases: Continual updates on the performance of its oncology and internal medicine portfolios, showing steady revenue generation in therapeutic areas relevant to hematuria treatment.
- Industry Recognitions/Awards: Consistently recognized for its contributions to global health and pharmaceutical innovation.
Value Chain Analysis – Hematuria Treatment Market
R&D
The latest research focuses on developing novel biologics or repurposing licensed drugs to treat hematuria.
Key Players: Pfizer, Inc., Johnson & Johnson, and Novartis AG.
Clinical Trials & Regulatory Approval
Clinical trials assess the safety and efficacy of hematuria treatment drugs, which are subsequently approved by regulatory agencies.
Key Players: Vera Therapeutics, UroGen Pharma, and Ferring Pharmaceuticals.
Patient Support & Services
Healthcare professionals guide patients about the dosage regimen and lifestyle regimen.
Recent Developments in the Hematuria Treatment Market
- In November 2025, Vera Therapeutics, Inc. announced positive results of its ORIGIN Phase 3 clinical trials of atacicept in IgA nephropathy. Atacicept showed a 46% reduction in proteinuria, and hematuria was resolved in 81% of participants. The company also aims to treat other autoimmune kidney diseases using atacicept.
- In April 2024, Alpine Immune Sciences, Inc. reported positive clinical data results for povetacicept in IgA nephropathy (IgAN). Povitacicept is a potent dual antagonist of the BAFF and APRIL cytokines. It resulted in UPCR reductions of greater than 60%, associated with remission, resolution of hematuria, and stable renal function.
Partner with our experts to explore the Hematuria Treatment Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
