Top Vendors in the High Potency Active Pharmaceutical Ingredients Market & Their Offering
Company Latest Offerings
| Companies | Headquarters | Offerings |
| BASF SE | Ludwigshafen, Germany | Develops high-quality APIs and supports the production of potent pharmaceutical compounds through advanced chemical synthesis. |
| CordenPharma | Plankstadt, Germany | Provides contract development and manufacturing (CDMO) services for complex and highly potent APIs. |
| BristolMyers Squibb | New Jersey, USA | Focuses on oncology and specialty medicines that utilize HPAPIs for effective treatment at low doses. |
| CARBOGEN AMCIS AG | Bubendorf, Switzerland | Offers custom synthesis and high-containment manufacturing solutions, including for antibody–drug conjugate intermediates. |
| Pfizer, Inc. | New York, USA | Produces and develops HPAPI-based therapies, particularly in oncology and targeted drug segments. |
| Boehringer Ingelheim | Ingelheim, Germany | Manufactures HPAPIs for oncology and specialty treatments, using stringent safety and containment standards. |
| Dr. Reddy’s Laboratories | Hyderabad, India | Provides end-to-end HPAPI development and manufacturing with advanced containment to manage occupational exposure. |
Value Chain Analysis
R&D
- R&D in High-Potency Active Pharmaceutical Ingredients (HPAPIs) centers on creating highly effective compounds for oncology, hormonal, and CNS therapies. Development involves strict safety protocols and specialized containment systems, like isolators, to control occupational exposure limits (OELs).
- Key players: Lonza, Catalent, Cambrex, Siegfried, and Piramal Pharma Solutions, focusing on safe, scalable, and compliant HPAPI production.
Clinical Trials
- Clinical trials for High-Potency Active Pharmaceutical Ingredients (HPAPIs) aim to confirm the safety and effectiveness of highly potent compounds, particularly in oncology and niche therapies, while maintaining strict occupational exposure limits (OELs).
- Key players: Aenova, Cambrex, Recipharm, PCI Pharma Services, and AMRI, which provide specialized expertise in handling and testing these high-potency molecules.
Regulatory Approvals
- Regulatory approval of High-Potency Active Pharmaceutical Ingredients (HPAPIs) demands complian ce with ICH Q7 GMP standards, emphasizing containment, risk control, and thorough validation to maintain purity and avoid cross-contamination.
- Key players: Boehringer Ingelheim, WuXi AppTec, Piramal Pharma Solutions, Recipharm, and Siegfried, recognized for their expertise in safe and compliant HPAPI manufacturing.
Market Growth Story
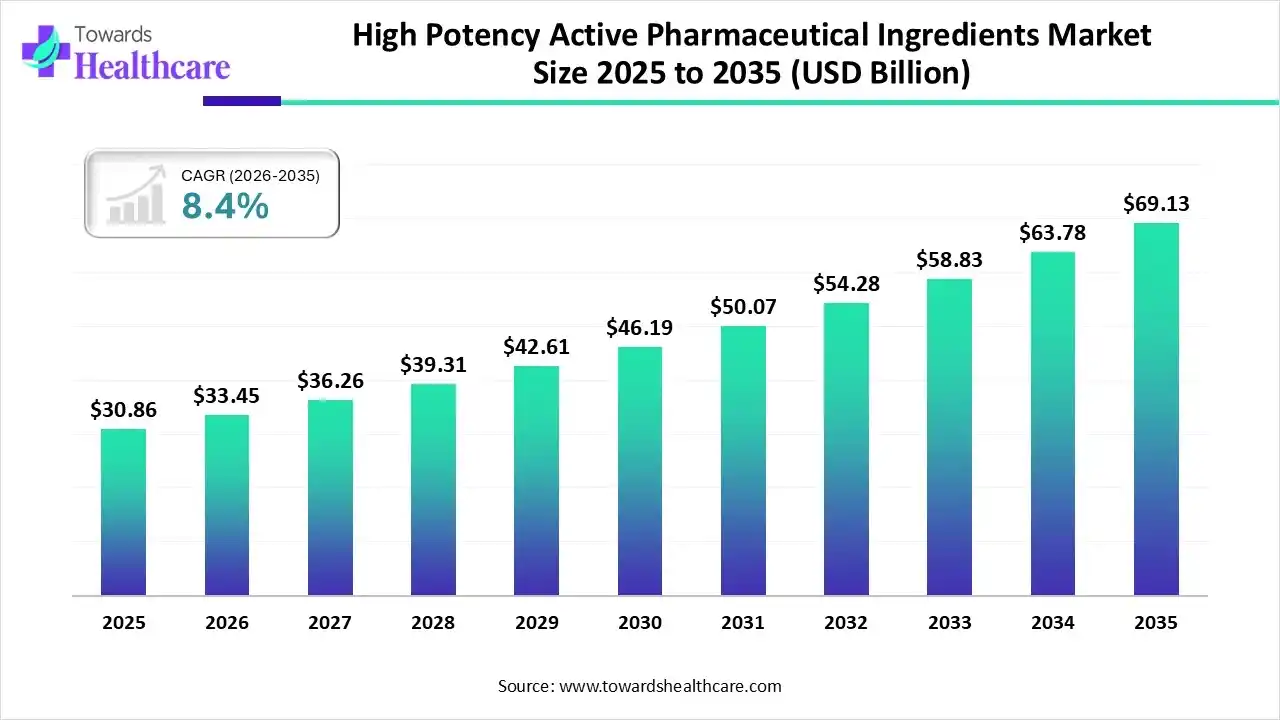
The global high potency active pharmaceutical ingredients market size was estimated at USD 30.86 billion in 2025 and is predicted to increase from USD 33.45 billion in 2026 to approximately USD 69.13 billion by 2035, expanding at a CAGR of 8.4% from 2026 to 2035.
What are the Recent Developments in the High Potency Active Pharmaceutical Ingredients Market?
- In June 2025, BASF announced the opening of a new GMP Solution Center in Wyandotte, Michigan, reinforcing its focus on the biopharma and pharmaceutical ingredients sectors. The facility enhances pharmaceutical excipient and biopharma capabilities through advanced cleanroom packaging, precise analytical testing, and collaborative technical support, enabling customized formulations and higher quality standards.
- In April 2025, the FDA approved Penpulimabkcqx for the treatment of relapsed nasopharyngeal carcinoma. The drug’s development and review were expedited through several fast-track designations, enabling faster patient access to this therapy.
Now it’s easier than ever to manage all your data in one place - Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
