Latest Updates of Key Players in the Intravenous Iron Drugs Market

Company Updates
| Company | Headquarters | Latest Update |
| CSL Vifor | Switzerland | In September 2025, CSL Vifor and Travere Therapeutics, Inc., supported the recent publication of the updated clinical practice guidelines for the treatment of IgA Nephropathy (IgAN), Kidney Disease: Improving Global Outcomes (KDIGO) 2025 healthcare practice guideline for the management of IgA Nephropathy and Immunoglobulin A vasculitis. |
| Daiichi Sankyo Company, Ltd. | Japan | Daiichi Sankyo is investing in major manufacturing expansion in the U.S. (Ohio) and China, which is primarily for ADC products. |
| Pharmacosmos A/S | Denmark | Pharmacosmos A/S is a global leader in carbohydrate chemistry and innovative treatments for iron deficiency and iron deficiency anaemia. |
| Sanofi S.A. | France | Sanofi S.A. is heavily focused on transitioning its portfolio toward high-growth immunology, rare disease, and vaccine products. |
| Fresenius Kabi AG | Germany | In December 2025, Fresenius announced the launch of its denosumab biosimilars, Conexxence and Bomyntra, in Europe |
| AbbVie Inc. | United States | In February 2026, AbbVie announced that it had submitted applications for an advanced indication to the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) for upadacitinib for the management of adult and adolescent patients living with non-segmental vitiligo. |
Value Chain Analysis
R&D:
- R&D processes in intravenous iron drugs involve preclinical formulation advancement and characterization, preclinical safety and efficacy testing, clinical trial development, and many other activities.
- Key Players: CSL Vifor and Daiichi Sankyo Company, Ltd.
Manufacturing Processes:
- Major manufacturing processes involved in clinical trial drug sourcing are highly controlled, multi-stage, sterile, and complex chemical processes.
- Key Players: Pharmacosmos A/S, and Fresenius Kabi AG
Patient Services:
- Key services involve a comprehensive, multi-stage process designed to safely and efficiently increase hemoglobin and iron levels when oral supplements are ineffective or unsuitable.
- Key Players: Sanofi, Pharmacosmos A/S, and Fresenius Kabi AG
Market Growth Story
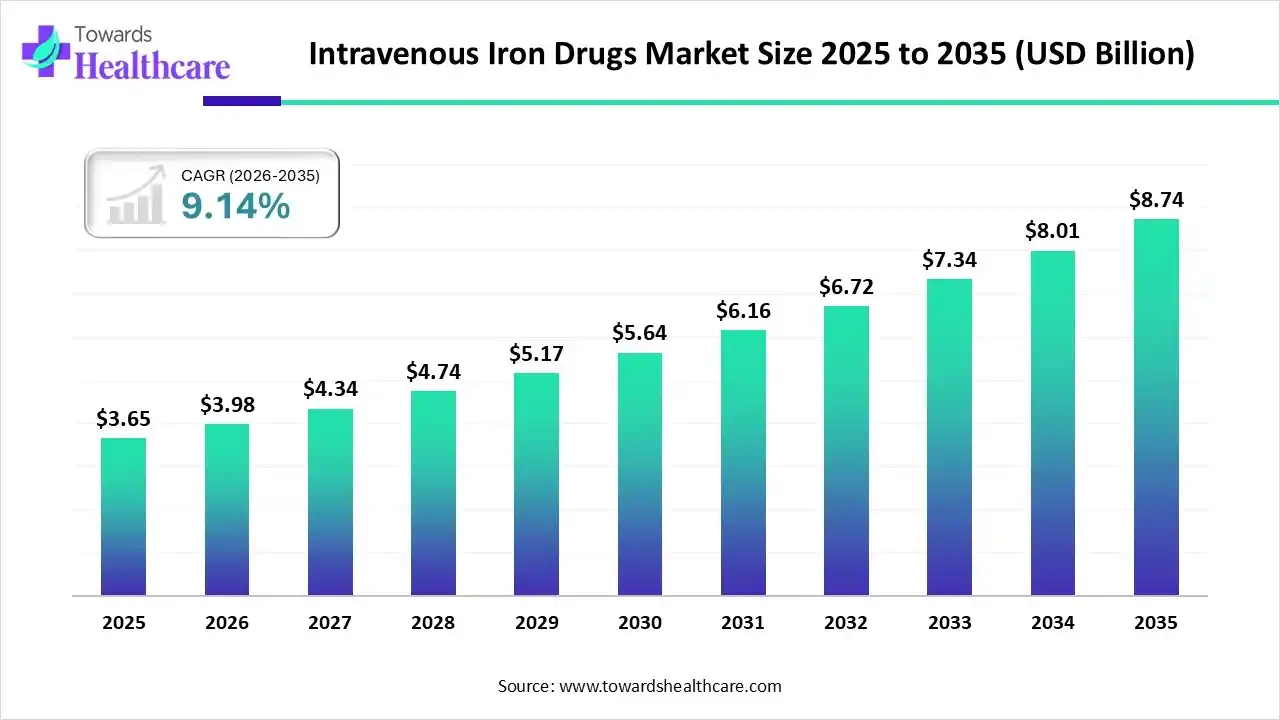
The global intravenous iron drugs market size was estimated at USD 3.65 billion in 2025 and is predicted to increase from USD 3.98 billion in 2026 to approximately USD 8.74 billion by 2035, expanding at a CAGR of 9.14% from 2026 to 2035.
Recent Developments in the Intravenous Iron Drugs Market
- In November 2025, Viatris (VTRS) announced the FDA had approved Iron Sucrose Injection, USP, an intravenous iron replacement product used to manage iron deficiency anemia in adult and pediatric patients with chronic kidney disease.
- In September 2025, Sandoz, the worldwide leader in cost-effective medicines, announced the US launch of its generic iron sucrose injection following recent approval by the US Food and Drug Administration (FDA).
- In August 2025, Viatris Inc., a global healthcare company, announced the U.S. Food and Drug Administration (FDA) had approved Iron Sucrose Injection, USP, an intravenous iron replacement product used to treat iron deficiency anemia (IDA) in adult and pediatric patients with chronic kidney disease.
No scattered data, no confusion - manage all your Intravenous Iron Drugs Market services information in one place - Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
