Explore the Companies Shaping the Future of Oral Mucositis Market
- Amgen Inc.
- Basic Pharma Life Science Pvt Ltd.
- CANbridge Pharmaceuticals Inc.
- Enzychem Lifesciences Corporation
- EpicentRx, Inc.
- Galera Therapeutics, Inc.
- Helsinn Healthcare SA.
- Innovation Pharmaceuticals Inc.
- Monopar Therapeutics, Inc.
- Pfizer Inc.
- Soleva Pharma, LLC.
- Swedish Orphan Biovitrum AB
What is Oral Mucositis?
The main factor driving the oral mucositis market is the rising incidence of cancer. An acute inflammation of the oral mucosa that occurs after systemic cancer therapy, especially chemotherapy and/or radiation, is known as oral mucositis (OM). An estimated 40% of patients are thought to be affected by this issue.
Market Growth
The oral mucositis market size touched US$ 1.64 billion in 2025, with expectations of climbing to US$ 1.76 billion in 2026 and hitting US$ 3.35 billion by 2035, driven by a CAGR of 7.38% over the forecast period from 2026 to 2035.
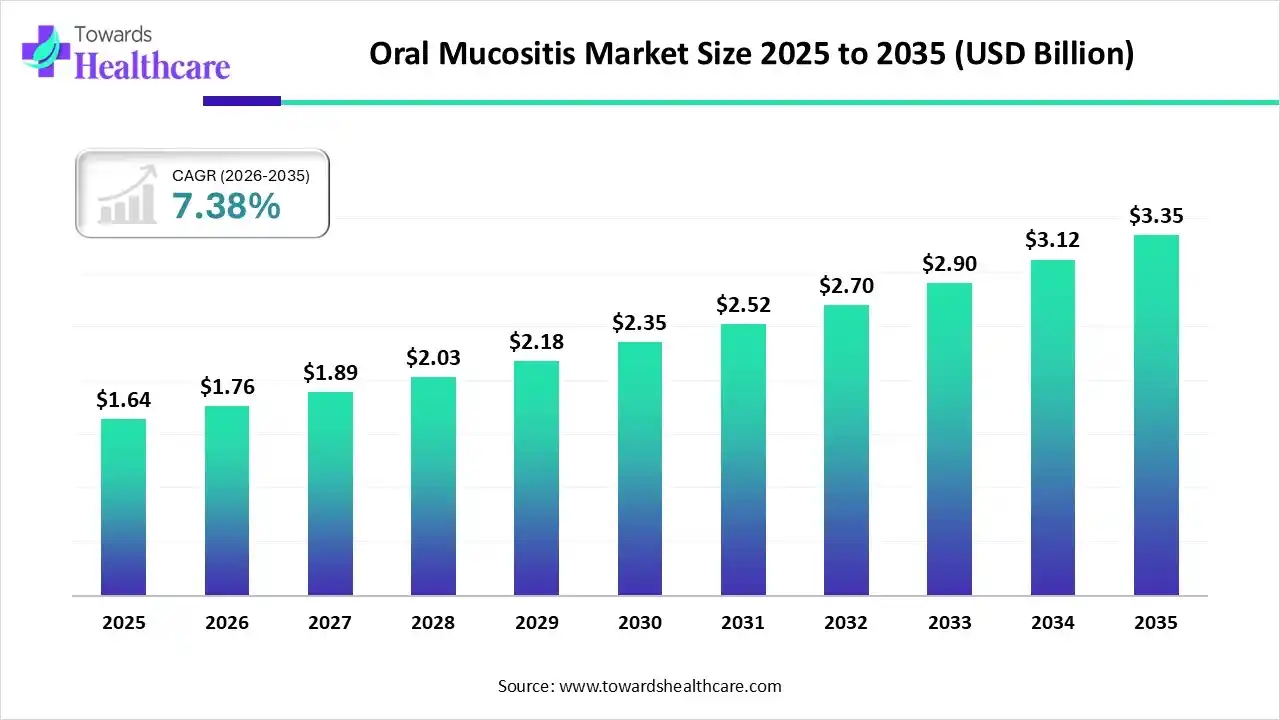
Oral Mucositis Market Outlook
- Industry Growth Overview: The market is anticipated to grow significantly between 2025 and 2034 as a result of rising cancer rates worldwide and an increase in chemotherapy treatments. Laser therapies and novel bioadhesive gels are the main forces behind growth.
- Major Investors: Pharmaceutical behemoths Amgen, Pfizer, and Soligenix are major investors in the oral mucositis market. To increase their product offerings and market penetration, they prioritize significant investments in R&D, partnerships, and acquisitions.
- Startup Ecosystem: Innovative businesses like Innovation Pharma, Monopar, and Galera are part of the startup ecosystem. They develop cutting-edge treatments and medication-delivery methods, fostering a vibrant atmosphere for patient-centered care and preventive measures.
Top Vendors in the Oral Mucositis Market & Their Offerings
1. Basic Pharma Life Science Pvt Ltd
-
Role / Offering: API supplier
-
Key Contribution: Manufactures chlorhexidine base, used in mouthwashes for oral care/disinfection
-
Current Status / Highlights: Supplies GMP-grade chlorhexidine for global customers
-
Mechanism / Technology: Antiseptic, broad-spectrum disinfectant for mucosal cleaning
2. CANbridge Pharmaceuticals Inc.
-
Role / Offering: Commercialization
-
Key Contribution: Licensed Caphosol® (CAN002) for China to treat oral mucositis
-
Current Status / Highlights: CFDA approval obtained; first commercial OM therapy in China
-
Mechanism / Technology: Supersaturated calcium phosphate oral rinse that lubricates and protects the mucosa
3. Enzychem Lifesciences Corporation
-
Role / Offering: Drug developer
-
Key Contribution: Developing EC-18 (mosedipimod) for chemoradiation-induced oral mucositis
-
Current Status / Highlights: Completed Phase 2, database lock; positive safety/efficacy; Fast Track from FDA for CRIOM
-
Mechanism / Technology: Oral immunomodulator that reduces inflammatory neutrophil recruitment
4. EpicentRx, Inc.
-
Role / Offering: Clinical developer
-
Key Contribution: Developing RRx-001 to prevent/reduce severe oral mucositis
-
Current Status / Highlights: FDA granted Fast Track; IND accepted for Phase 2b trial (KEVLARx) in head & neck cancer
-
Mechanism / Technology: NLRP3 inhibitor & Nrf2 activator; anti-inflammatory, antioxidant effects
5. Galera Therapeutics, Inc.
-
Role / Offering: Therapeutic innovator
-
Key Contribution: Developing avasopasem manganese (GC4419) for radiotherapy-induced severe oral mucositis
-
Current Status / Highlights: Submitted NDA; completed Phase 3 ROMAN trial; Fast Track / Breakthrough from FDA
-
Mechanism / Technology: Small-molecule dismutase mimetic (superoxide dismutase mimic) to reduce oxidative damage
Company Landscape
Amgen Inc.
Company Overview: Amgen is a leading global biotechnology company dedicated to unlocking the potential of biology for patients suffering from serious illnesses by discovering, developing, manufacturing, and delivering innovative human therapeutics.
Corporate Information
- Headquarters: Thousand Oaks, California, United States
- Year Founded: 1980
- Ownership Type: Public (NASDAQ: AMGN)
History and Background:
Founded as Applied Molecular Genetics Inc., Amgen pioneered the biotechnology industry. It has a history of developing transformative medicines, including some of the first successful recombinant human proteins, focusing on areas like oncology, inflammation, and nephrology.
Key Milestones/Timeline
- 1980: Founded.
- 2003: Pivotal Phase 3 data for palifermin (later Kepivance) showed a significant reduction in severe OM incidence/duration.
- 2004: Submitted Biologics License Application (BLA) for palifermin.
- 2004/2005: FDA and EMA approvals for palifermin (Kepivance) for use in specific high-risk cancer patients.
Business Overview: Focuses on six therapeutic areas: Cardiovascular Disease, Oncology, Bone Health, Neuroscience, Nephrology, and Inflammation. The company is committed to science-based innovation.
Business Segments/Divisions
- Human Therapeutics (Primary division for product sales).
- Research and Development.
- Manufacturing.
Geographic Presence: Global presence, with operations across North America (largest market share), Europe, and the Asia-Pacific regions.
Key Offerings (Relevant to OM Market)
Kepivance (palifermin): A recombinant human keratinocyte growth factor (KGF) indicated to decrease the incidence and duration of severe oral mucositis in patients with hematologic malignancies receiving high-dose chemotherapy and/or radiotherapy followed by hematopoietic stem cell transplant (HSCT).
End-Use Industries Served
- Hospitals and Cancer Treatment Centers (Primary users for Kepivance).
- Specialty Clinics.
- Healthcare Systems.
Key Developments and Strategic Initiatives (Latest Info)
- Mergers & Acquisitions: Continues to pursue strategic acquisitions; e.g., the planned acquisition of Horizon Therapeutics.
- Partnerships & Collaborations: Ongoing R&D collaborations with academic institutions and biotechs focused on advancing novel therapeutics.
- Product Launches/Innovations: Ongoing development and commercialization of new biologics and biosimilars.
- Technological Capabilities/R&D Focus: Strong focus on biologics, human genetics, and novel drug delivery systems, with a significant R&D budget ($4.9 billion in 2023).
Competitive Positioning
- Strengths & Differentiators: Only FDA-approved recombinant growth factor (Kepivance) for a specific OM indication; strong global oncology presence; deep expertise in biologics.
- Market presence & ecosystem role: A market leader in supportive oncology care through its high-value biologic, primarily serving the high-risk, inpatient HSCT segment.
SWOT Analysis
- Strengths: Proprietary, FDA-approved biologic; strong financial base; robust R&D pipeline.
- Weaknesses: Kepivance use is limited to a specific high-risk patient population; high product cost limits broader access.
- Opportunities: Expanding indications for KGF analogs; developing biosimilars for supportive care.
- Threats: Competition from emerging non-drug therapies (LLLT); patent expiration pressure; changing reimbursement models.
Recent News and Updates (2024/2025)
- Press Releases: Recent focus on new oncology launches (not directly OM-related) and general portfolio growth in Q3 2025.
- Industry Recognitions/Awards: Frequently recognized as a top R&D spender and innovator in the biopharma sector.
Bausch Health Companies Inc. (via Salix Pharmaceuticals)
Company Overview: Bausch Health is a multinational pharmaceutical company that develops, manufactures, and markets a range of pharmaceutical, medical device, and over-the-counter products, primarily in gastroenterology (GI), ophthalmology, and dermatology. Its OM presence is driven by the Salix Pharmaceuticals segment.
Corporate Information (Bausch Health Companies Inc.)
- Headquarters: Laval, Quebec, Canada (Bausch Health); Bridgewater, New Jersey, United States (Salix)
- Year Founded: 2013 (BHC, post-rebranding); 1989 (Salix)
- Ownership Type: Public (NYSE/TSX: BHC)
History and Background:
Salix Pharmaceuticals was acquired by Bausch Health (then Valeant Pharmaceuticals) in 2015. Salix is a leading specialty pharmaceutical company focused on GI disorders. It markets Gelclair, an FDA-approved prescription rinse/gel for oral mucositis.
Key Milestones/Timeline (Relevant to OM Market)
- 2001: Gelclair receives FDA clearance (510(k)).
- 2015: Salix Pharmaceuticals acquired by Bausch Health.
- 2023/2024: Jaguar Health Inc. (Napo Pharmaceuticals subsidiary) acquired US rights to Gelclair from Salix, though Salix/Bausch retains historical market impact and may have ongoing distribution ties in certain markets/regions. Note: Market reports often still cite Bausch/Salix as a top player due to the product's market establishment.
Business Overview: Bausch Health operates three segments: Salix (Gastrointestinal), International, and Bausch + Lomb (Eye Health). Salix focuses on developing and commercializing proprietary GI products.
Business Segments/Divisions (BHC)
- Salix Pharmaceuticals (Gastrointestinal, includes OM product).
- Bausch + Lomb (Spun-off entity).
- International (Pharmaceuticals outside the US).
- Geographic Presence: Strong presence in the United States (via Salix) and a broad international footprint.
Key Offerings (Relevant to OM Market)
Gelclair (formerly Salix): An oral rinse/gel that forms a protective barrier over the oral mucosa, providing pain relief and promoting healing of oral lesions caused by chemotherapy and/or radiation therapy. Note: US commercial rights now belong to Jaguar Health as of April 2023/2024.
End-Use Industries Served
- Hospital Pharmacies and Clinics (for oncology supportive care).
- Specialty and Retail Pharmacies.
- Gastroenterologists and Oncologists.
Key Developments and Strategic Initiatives (Latest Info)
- Mergers & Acquisitions: Bausch Health has focused on divestitures (like the Bausch + Lomb IPO) to simplify its structure and reduce debt.
- Partnerships & Collaborations: Focus on licensing and distribution agreements for specialty GI products.
- Product Launches/Innovations: Salix continues to launch and expand indications for GI products, indirectly supporting its hospital/oncology presence.
- Distribution channel strategy: Highly dependent on hospital pharmacy and specialty distribution networks for its specialty GI and supportive care products.
Competitive Positioning
- Strengths & Differentiators: Strong hospital presence via the Salix GI portfolio; Gelclair is a recognized and widely adopted barrier agent (a dominant treatment type).
- Market presence & ecosystem role: A significant player in the high-volume barrier/coating segment, leveraging its established pharmaceutical distribution channels in supportive care.
SWOT Analysis
- Strengths: Diversified revenue streams beyond OM; established distribution networks; strong brand recognition for key OM product (Gelclair).
- Weaknesses: Gelclair ownership change (to Jaguar Health) may impact future OM market share metrics for BHC; high debt load.
- Opportunities: Growth in the global supportive oncology care market; utilizing Salix's GI focus for related mucosal injury treatments.
- Threats: Intense competition in barrier agents; generic competition; shift towards non-drug options like Photobiomodulation Therapy (PBMT).
Recent News and Updates (2024/2025)
- Press Releases: Bausch Health's recent news centers on financial restructuring, debt management, and the performance of its core GI portfolio. OM-specific news is limited post-Gelclair rights transfer.
- Industry Recognitions/Awards: Focus on its overall GI portfolio leadership.
Oral Mucositis Market Value Chain Analysis
R&D
- Finding novel therapeutic agents, creating biologics, improving mucosal healing mechanisms, and developing radioprotective and anti-inflammatory candidates are the main goals of the oral mucositis research and development stage.
- Key companies involved include Amgen, Helsinn, Soligenix, Galera Therapeutics, Enzychem Lifesciences, Innovation Pharmaceuticals, and Monopar Therapeutics.
Clinical Trials and Regulatory Approvals
- This phase entails preparing regulatory submissions for major international markets while assessing safety, efficacy, dosage optimization, and long-term patient benefit.
- Key companies conducting trials or pursuing approvals include Galera Therapeutics, Soligenix, Amgen, Bausch Health, EpicentRx, Monopar Therapeutics, and Onxeo.
Distribution to Hospitals and Pharmacies
- Distribution focuses on using international logistics partners and specialized medical distributors to guarantee uniform product availability across oncology centers, clinics, and pharmacies.
- Key companies involved include 3M, Bausch Health, Colgate-Palmolive, Alliance Pharma, Helsinn, Napo Pharmaceuticals, and OraPharma.
Recent Developments in the Oral Mucositis Market
- In March 2025, the official launch of GenSci's flagship product, Episil®, which is currently offered in China, was announced. Solasia Pharma K.K. granted GenSci exclusive commercialization rights for Episil® in China through a strategic alliance. Cancer patients facing treatment-related oral complications have new hope thanks to Episil®, the only clinically validated mucosal protectant in China with a registered clinical trial.
- In October 2024, Jaguar Health, Inc. declared that Napo Pharmaceuticals ("Napo"), a member of the Jaguar family, had started the commercial introduction of Gelclair, a prescription medication for oral mucositis that had received FDA approval in the U.S.
Collaborate with our experts to explore the Oral Mucositis Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
