Top Companies in the Orphan Oncology Market & Their Offerings

Centralize your Orphan Oncology Market intelligence and stay ahead - Buy Now.
| Companies | Headquarters | Offerings |
| Agios Pharmaceuticals | Massachusetts, U.S. | Develops targeted therapies for rare blood cancers, especially AML, MDS, and genetically defined oncology conditions. |
| Incyte Corporation | Delaware, U.S. | Focuses on precision oncology therapies for rare hematologic malignancies and biomarker-driven cancer indications. |
| Blueprint Medicines | Massachusetts, U.S. | Specializes in mutation-targeted therapies for rare cancers, including mast cell disorders and genomically driven tumors. |
| Deciphera Pharmaceuticals | Massachusetts, U.S. | Offers kinase inhibitors and targeted treatments for rare tumors such as GIST and other orphan oncology indications. |
| Karyopharm Therapeutics | Massachusetts, U.S. | Provides therapies for multiple myeloma and rare hematologic cancer treatments with novel export inhibition mechanisms. |
| SpringWorks Therapeutics | Connecticut, U.S | Focuses on therapies for rare tumors and oncology conditions, including desmoid tumors and NF1-associated cancers. |
What are the Recent Developments in the Orphan Oncology Market?
- In June 2025, a newly developed oral therapy candidate received orphan drug recognition from the U.S. FDA for sickle cell disease, a rare blood disorder affecting a limited patient group. This milestone is expected to support accelerated clinical development, regulatory benefits, and improved focus on innovative treatment options for rare disease management.
- In January 2025, a specialized orphan therapy for adults with non-dystrophic myotonic disorders was introduced in the European market to support symptomatic relief of myotonia. This launch strengthened treatment accessibility for patients with rare neuromuscular conditions and marked an important step in expanding orphan drug availability for underserved patient groups.
Supply Chain Analysis
R&D
- R&D in orphan oncology is primarily focused on creating advanced targeted therapies for rare cancer indications, especially blood-related malignancies such as AML and MDS. Strong regulatory benefits, including orphan drug incentives, exclusivity periods, and tax support, continue to encourage innovation. Although development expenses are high, the segment remains commercially attractive due to strong revenue potential.
- Key players: Roche, Novartis, Bristol Myers Squibb, Pfizer, and Johnson & Johnson.
Formulation and Final Dosage Preparation
Formulation and final dosage preparation in orphan oncology involve highly specialized processes to improve drug stability, address low solubility challenges, and ensure precise dosing for small patient groups, including pediatric cases. These steps are essential for maintaining treatment efficacy and safety in rare cancer therapies. Key players: Lonza Group, Catalent, Thermo Fisher Scientific, Recipharm, and Samsung Biologics.
Patient Support and Services
- Patient support in orphan oncology mainly focuses on easing the financial and emotional burden associated with rare cancer treatment. Assistance is provided through NGOs, charitable foundations, specialty cancer centers, and palliative care organizations that help with funding, patient counseling, awareness programs, and supportive care services.
- Key players: CanSupport, ORDI, Pallium India, Indian Cancer Society, and Tata Memorial Centre.
Market Trends
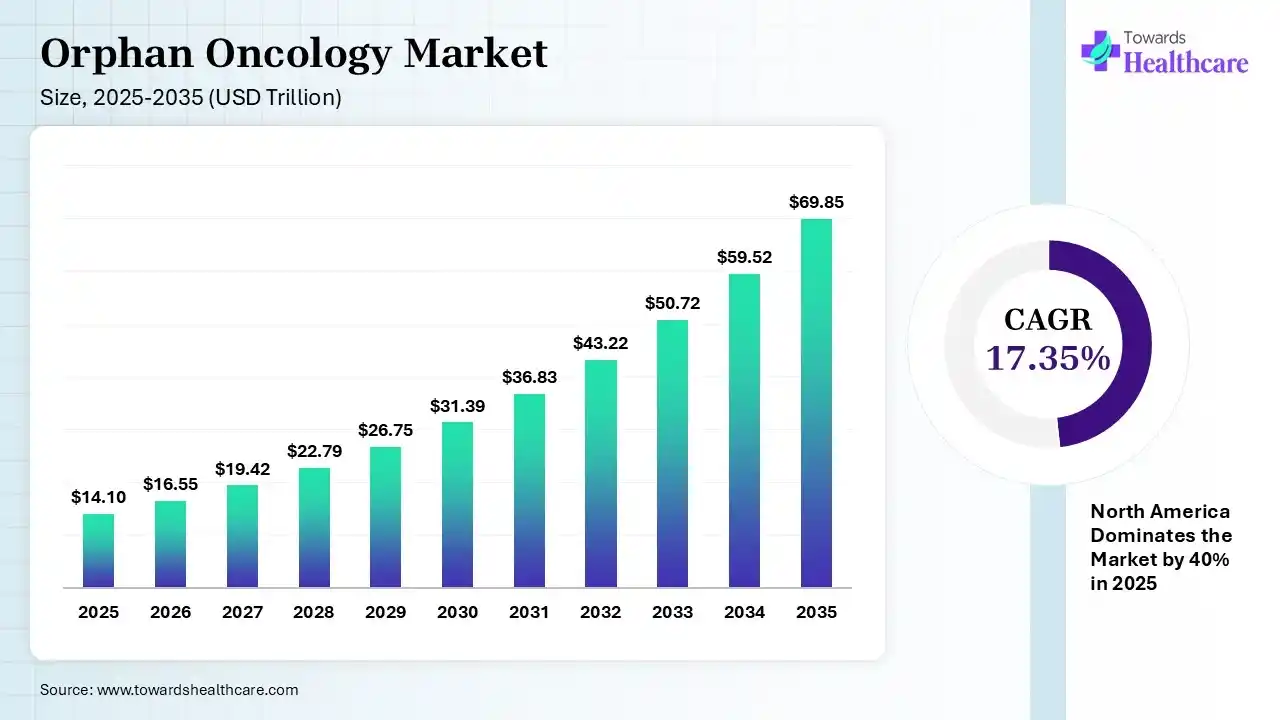
The global orphan oncology market size was estimated at USD 16.55 trillion in 2025 and is predicted to increase from USD 16.55 trillion in 2026 to approximately USD 69.85 trillion by 2035, expanding at a CAGR of 17.35% from 2026 to 2035. The market is growing due to rising focus on rare cancer treatments and advancements in precision medicine and targeted biologics. Strong regulatory support, faster approvals, and increasing R&D investments are driving new drug development and market expansion.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
