April 2026

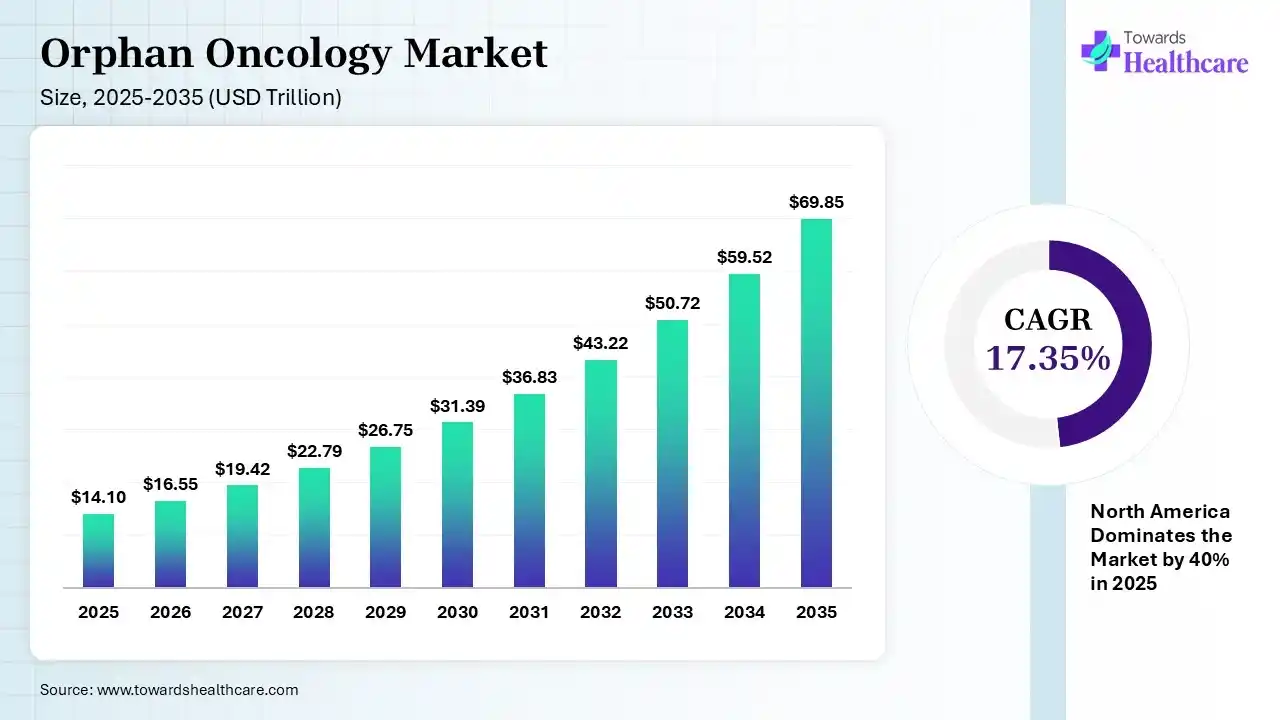
The global orphan oncology market size was estimated at USD 16.55 trillion in 2025 and is predicted to increase from USD 16.55 trillion in 2026 to approximately USD 69.85 trillion by 2035, expanding at a CAGR of 17.35% from 2026 to 2035. The market is growing due to rising focus on rare cancer treatments and advancements in precision medicine and targeted biologics. Strong regulatory support, faster approvals, and increasing R&D investments are driving new drug development and market expansion.
Orphan oncology refers to the development and treatment of drugs for rare cancers that affect a small patient population. It focuses on targeted, high-precision therapies supported by special regulatory incentives to address unmet medical needs. The orphan oncology market is expanding due to the increasing incidence of rare and complex cancers that require specialized treatment options. Advances in genomics, precision medicine, and targeted biologics are improving therapy effectiveness. Strong regulatory incentives, including orphan drug designations and faster approvals, are encouraging pharma companies to invest heavily. Rising funding and unmet clinical needs further accelerate innovation and global market growth.
AI is transforming the market by enabling faster drug discovery, identifying rare cancer biomarkers, and improving patient stratification through advanced data analysis. It supports more accurate diagnosis and personalized treatment planning while reducing research time and costs. AI-driven predictive modeling also helps accelerate clinical trials and improve success rates for targeted therapies.
| Table | Scope |
| Market Size in 2026 | USD 16.55 Trillion |
| Projected Market Size in 2035 | USD 69.85 Trillion |
| CAGR (2026 - 2035) | 17.35% |
| Leading Region | North America by 40% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Therapy Type, By Application, By Drug Modality, By Distribution Channel, By Region |
| Top Key Players | Agios Pharmaceuticals, Incyte Corporation, Blueprint Medicines, Deciphera Pharmaceuticals, Karyopharm Therapeutics, SpringWorks Therapeutics |
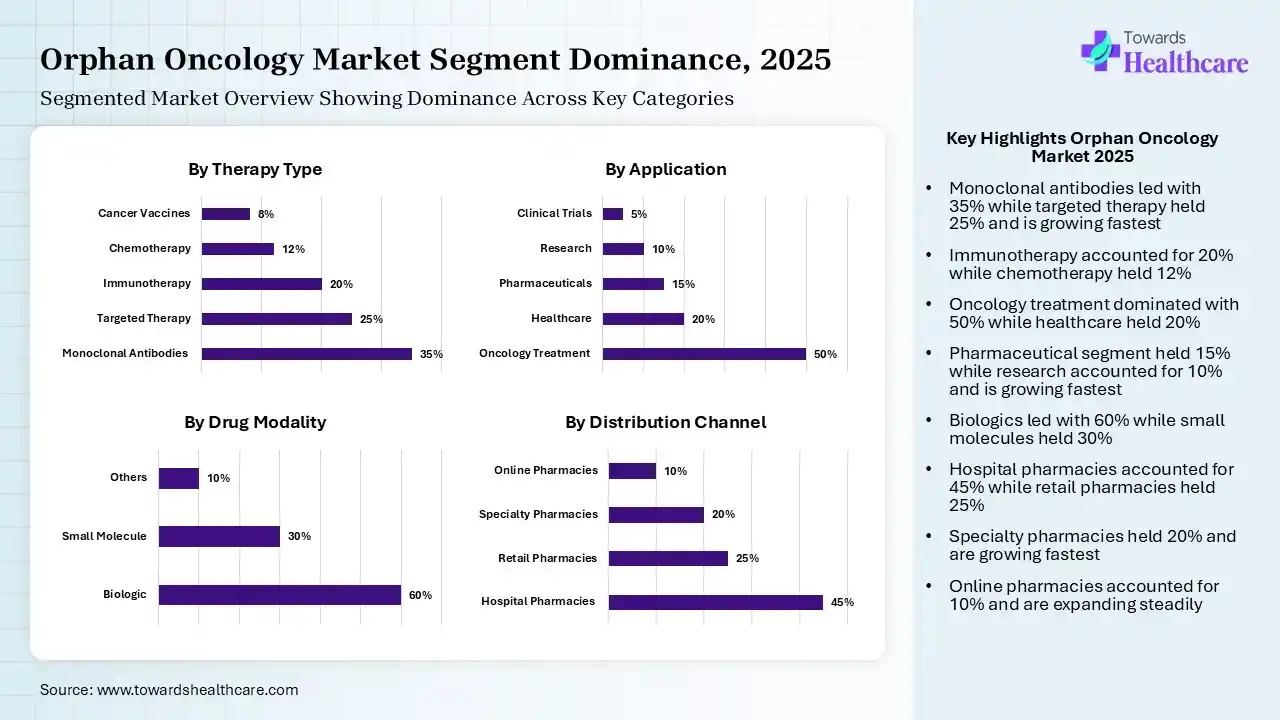
By Therapy Type Insights
| Segment | Share 2025 (%) |
| Monoclonal Antibodies | 35% |
| Targeted Therapy | 25% |
| Immunotherapy | 20% |
| Chemotherapy | 12% |
| Cancer Vaccines | 8% |
The Monoclonal Antibodies Segment Dominated the Market in 2025
The monoclonal antibodies segment dominated the orphan oncology market with a share of 35% in 2025 because of its strong effectiveness in treating rare cancer types with high target precision. These therapies offer better response rates and fewer side effects compared to conventional treatments. Growing adoption in personalized cancer care, along with continuous innovation in next-generation antibody therapies, further supported its dominant market position.
The targeted therapy segment held the second-largest share of 25% of the market in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to its strong adoption in treating rare cancers through biomarker-specific mechanisms and improved treatment precision. It is expected to grow at the fastest CSGR during the forecast period because of rising advances in molecular diagnostics, expanding precision oncology pipelines, and increasing demand for personalized therapies with better efficacy and reduced systemic toxicity.
The immunotherapy segment held 20% of the orphan oncology market share in 2025 due to its ability to enhance the body’s immune response against rare and difficult-to -treat cancers. Increasing clinical success in hematologic malignancies and rare solid tumours, along with advances in checkpoint inhibitors, CAR-T therapies, and combination regimens, is driving adoption. Rising approvals and strong pipeline activity are further supporting sustained segment growth.
The chemotherapy segment held 12% of the market share in 2025 due to its continued use as a foundational treatment for several rare cancers, especially where targeted options remain several rare cancers, especially where targeted option remain limited. It is widely adopted in combination with immunotherapies and targeted drugs to improve response rates. Increasing diagnosis of rare hematologic malignancies and broader access in emerging markets are also supporting steady segment growth.
| Segment | Share 2025 (%) |
| Oncology Treatment | 50% |
| Healthcare | 20% |
| Pharmaceuticals | 15% |
| Research | 10% |
| Clinical Trials | 5% |
The Oncology Treatment Segment Led the Market in 2025 with the Largest Share
The oncology treatment segment led the orphan oncology market with shares of 50% in 2025 due to the rising burden of rare cancers and growing demand for advanced treatment options. Increased use of targeted therapies, monoclonal antibodies, and immunotherapies significantly improved patient outcomes. Strong investments in clinical research, higher treatment adoption rates, and supportive regulatory pathways further reinforced the segment’s dominant market position.
The healthcare segment held the second-largest share of 20% of the market in 2025 due to the growing involvement of hospitals, speciality oncology centers, and diagnostic laboratories in rare cancer management. Rising investments in advanced diagnostic infrastructure, personalized treatment services, and supportive care programs have strengthened this segment. Increased patient access to specialized healthcare facilities and multidisciplinary treatment further contributed to its significant market presence.
The pharmaceutical segment held 15% of the orphan oncology market share in 2025 due to increasing focus on developing innovative therapies for rare cancers and strong investment in research and clinical trials. Companies are expanding pipelines in targeted therapy, biologics, and immunotherapy to address unmet patient needs. Supportive orphan drug incentives, faster approval pathways, and higher commercial value per therapy are further driving this segment growth.
The research segment held 10% share in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to rising investment in rare cancer drug discovery and biomarker studies. Increasing focus on precision oncology, AI-driven clinical research, and expanding collaborations between pharmaceutical companies and research institutes are accelerating innovation, pipeline development, and future market growth.
| Segment | Share 2025 (%) |
| Biologic | 60% |
| Small Molecule | 30% |
| Others | 10% |
The Biologics Segment Led the Market in 2025 with the Largest Share
The biologics segment led the orphan oncology market with shares of 60% in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to its high effectiveness in targeting rare cancer pathways with greater precision and improved clinical outcomes. Strong adoption of monoclonal antibodies, cell-based therapies, and recombinant proteins, along with rising approvals and pipeline expansion, supported its leading market share.
The small molecule segment held the second-largest share of 30% of the market in 2025 due to its strong use in targeted cancer treatment, oral administration convenience, and cost-effective manufacturing. These therapies are widely adopted for rare cancers with specific genetic mutations. Continuous innovation in kinase inhibitors and other precision drugs has further strengthened the segment position.
| Segment | Share 2025 (%) |
| Hospital Pharmacies | 45% |
| Retail Pharmacies | 25% |
| Specialty Pharmacies | 20% |
| Online Pharmacies | 10% |
The Hospital Pharmacies Segment held a dominant position in the Market in 2025
The hospital pharmacies segment held a dominant position in the orphan oncology market with a share of 45% in 2025 due to the high administration of orphan oncology drugs within a hospital setting, especially biologics, chemotherapies, and immunotherapies that require specialist supervision. Hospitals remain the primary point of diagnosis, treatment, and follow-up for rare cancer patients. Better drug availability, reimbursement support, and access to multidisciplinary oncology care further strengthened the segment’s leading share.
The retail pharmacies segment second-largest share of 25% of the market in 2025 due to increasing availability of oral oncology drugs, supportive care medication, and follow-up prescriptions for rare cancer patients. Growing access to speciality pharmacy networks and improved distribution partnerships have made these therapies more accessible outside the hospital. Rising patient preference for convenient refill options and broader geographic reach further supported segment growth.
The specialty pharmacies segment held 20% share in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to rising demand for high-cost complex orphan oncology drugs that require personalized dispensing and patient support services. These pharmacies offer therapy management, reimbursement assistance, and cold-chain handling for biologics and advanced therapies, making them increasingly preferred for rare cancer treatment distribution.
The online pharmacies segment held 10% of the orphan oncology market share in 2025 due to increasing demand for convenient home delivery of oral oncology drugs and supportive care medications. Improved digital healthcare adoption, easy prescription refills, and broader access to specialty medicine in remote areas are driving growth. Rising patient preference for contactless services and competitive pricing options are further supporting expansion during the forecast period.
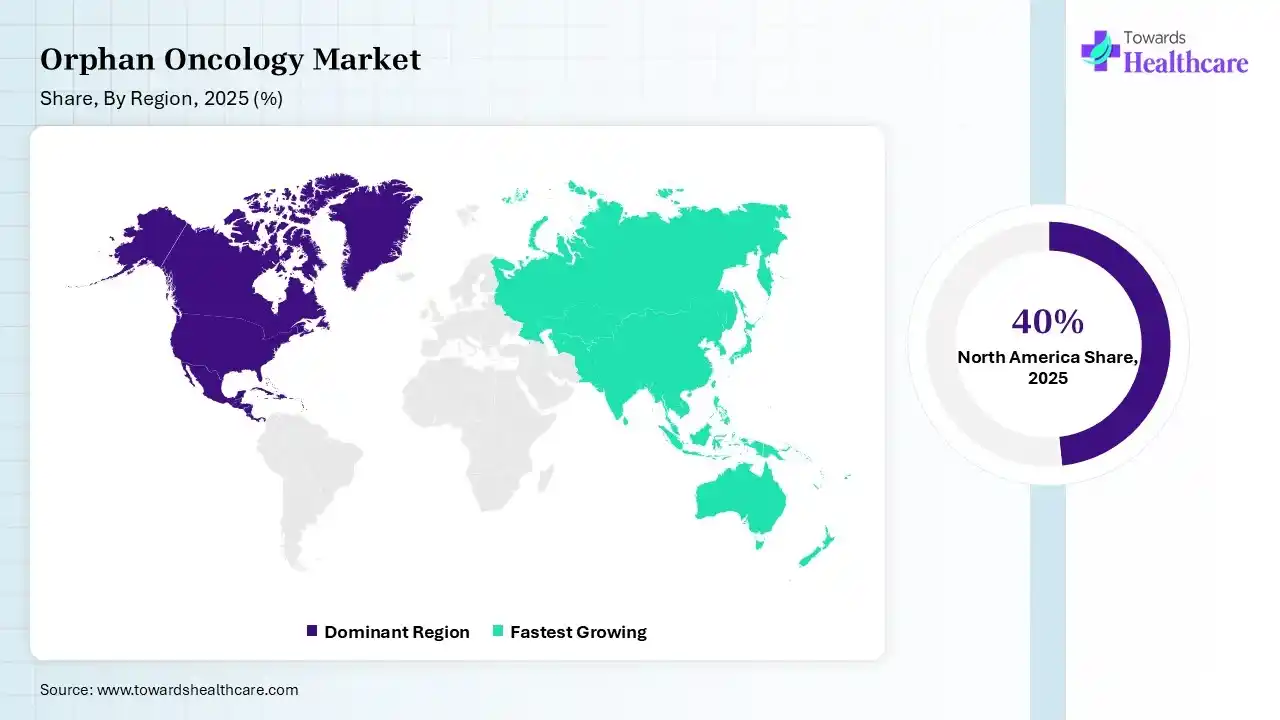
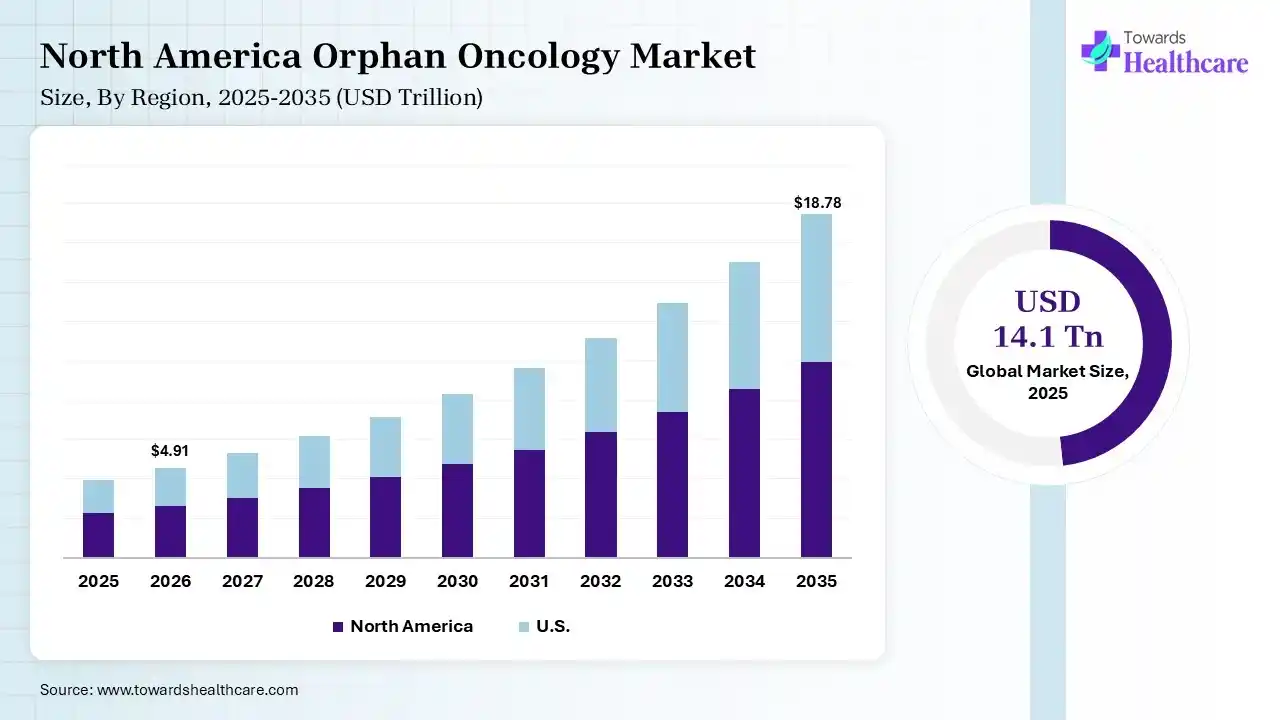
North America dominated the market with a revenue share of 40% in 2025 due to strong healthcare infrastructure, high investment in rare cancer research, and early adoption of advanced therapies. The region benefits from supportive regulatory frameworks, strong clinical trial activity, and the presence of major pharmaceutical innovators, which together accelerate drug development, approvals, and patient access to specialized treatments.
U.S. Market Trends
The U.S. market is growing due to increasing cases of rare cancers, strong funding for oncology research, and rapid development of targeted therapies. Supportive regulatory incentives, faster approval timelines, and high adoption of precision medicine are boosting innovation. In addition, the presence of leading pharmaceutical companies and advanced treatment facilities continues to support steady market growth.
Asia Pacific held 20% of the market share in 2025 and is anticipated to grow at the fastest CAGR in the orphan oncology market due to improving healthcare infrastructure, rising awareness of rare cancers, and increasing investment in oncology research. Expanding patient populations, growing access to advanced therapies, and supportive government initiatives are accelerating market expansion. In addition, pharmaceutical companies are increasing their presence and clinical trial activities across emerging economies in the region.
India Market Trends
India is expected to grow at the fastest CAGR in the orphan oncology market due to the increasing awareness of rare cancers and rapid improvements in oncology care infrastructure. Rising healthcare investments, expanding access to advanced therapies, and growing participation in clinical research are driving market growth. In addition, government support and the presence of cost-efficient pharmaceutical manufacturing further accelerate expansion.
R&D
Formulation and Final Dosage Preparation
Formulation and final dosage preparation in orphan oncology involve highly specialized processes to improve drug stability, address low solubility challenges, and ensure precise dosing for small patient groups, including pediatric cases. These steps are essential for maintaining treatment efficacy and safety in rare cancer therapies. Key players: Lonza Group, Catalent, Thermo Fisher Scientific, Recipharm, and Samsung Biologics.
Patient Support and Services

| Companies | Headquarters | Offerings |
| Agios Pharmaceuticals | Massachusetts, U.S. | Develops targeted therapies for rare blood cancers, especially AML, MDS, and genetically defined oncology conditions. |
| Incyte Corporation | Delaware, U.S. | Focuses on precision oncology therapies for rare hematologic malignancies and biomarker-driven cancer indications. |
| Blueprint Medicines | Massachusetts, U.S. | Specializes in mutation-targeted therapies for rare cancers, including mast cell disorders and genomically driven tumors. |
| Deciphera Pharmaceuticals | Massachusetts, U.S. | Offers kinase inhibitors and targeted treatments for rare tumors such as GIST and other orphan oncology indications. |
| Karyopharm Therapeutics | Massachusetts, U.S. | Provides therapies for multiple myeloma and rare hematologic cancer treatments with novel export inhibition mechanisms. |
| SpringWorks Therapeutics | Connecticut, U.S | Focuses on therapies for rare tumors and oncology conditions, including desmoid tumors and NF1-associated cancers. |
Strengths
Weaknesses
Opportunities
Threats
By Therapy Type
By Application
By Drug Modality
By Distribution Channel
By Region
April 2026
April 2026
April 2026
April 2026