Top Companies in the Positive Airway Pressure Devices Market and Their Offerings

Company Offerings
| Companies | Headquarters | Positive Airway Pressure Devices |
| ResMed Inc. | San Diego, U.S. | AirSense 10, AirSense 11, AirCurve 10, and AirMini |
| Koninklijke Philips N.V. | Amsterdam, Netherlands | DreamStation 2, DreamStation GO, and SystemOne |
| Fisher & Paykel Healthcare | Auckland, New Zealand | SleepStyle CPAP, F&P Icon, Eson 2, and Evora |
| BMC Medical Co., Ltd. | Beijing, China | RESmart G2S A20, RESmart Y30T, and G3 Series |
| Drive DeVilbiss Healthcare | Port Washington, U.S. | DeVilbiss Blue AutoPlus, IntelliPAP series, and SleepCube |
| Lowenstein, Medical | Hamburg, Germany | Prisma 20A, Prisma SMART, Prisma 25S |
| Wellell Inc. | New Taipei City, Taiwan | iXT series, XT Auto, and XT Fit |
| React Health | Winter Haven, U.S. | Luna II, G3 BiLevel ST 30VT, G3 CPAP/APAP, and Travel PAP |
| Somnetics International, Inc. | Eagan, U.S. | Transcend Micro and Transcend 3 |
| Invacare Corporation | Elyria, U.S. | Polaris EX |
Market Value Chain Analysis
R&D
- The R&D of the positive airway pressure devices focuses on the integration of AI-driven diagnostic algorithms, silent micro-blower technology, and cloud-based remote algorithms.
- Key players: ResMed Inc., Fisher & Paykel Healthcare, and Lowenstein Medical.
Clinical Trials and Regulatory Approvals
- The safety, pressure accuracy, apnea-hypopnea index reduction, and subjective patient comfort are evaluated in the clinical trials and regulatory approvals of the positive airway pressure devices.
- Key players: ResMed Inc., Fisher & Paykel Healthcare, Koninklijke Philips N.V.
Packaging and Serialization
- The packaging and serialization of the positive airway pressure devices involve the use of tamper-evident packaging and unique device identification (UDI) via GS1-compliant 2D data matrix codes for real-time tracking and authentication of each unit.
- Key players: ResMed Inc., Fisher & Paykel Healthcare, Koninklijke Philips N.V.
Patient Support and Services
- The patient-facing mobile apps, automated supply replenishment services, and telehealth coaching are provided in the patient support and services of the positive airway pressure devices.
- Key players: ResMed Inc., React Health, Koninklijke Philips N.V.
Market Growth
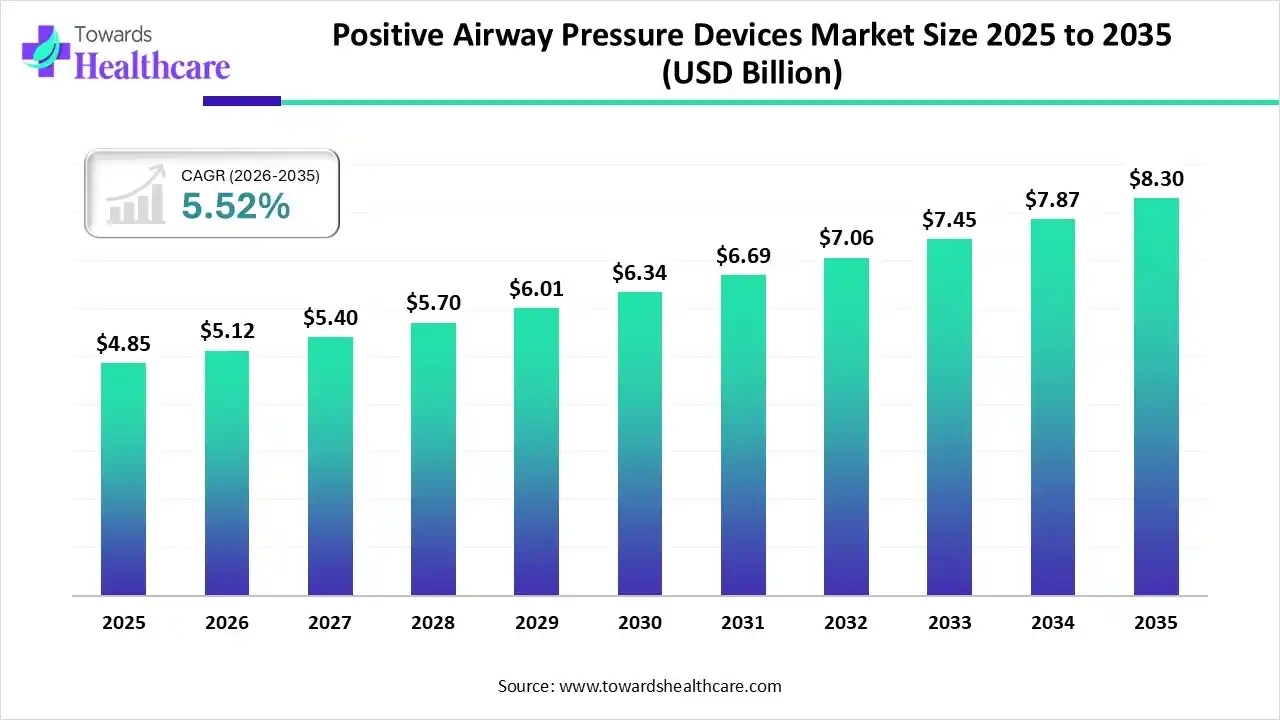
The global positive airway pressure devices market size was estimated at USD 4.85 billion in 2025 and is predicted to increase from USD 5.12 billion in 2026 to approximately USD 8.3 billion by 2035, expanding at a CAGR of 5.52% from 2026 to 2035.
Recent Developments in the Market
- In January 2026, the F&P Nova Nasal mask, a nasal positive airway pressure (PAP) mask for the treatment of obstructive sleep apnea, was launched by Fisher & Paykel Healthcare in the U.S.
- In December 2025, the Kricket PAP device, a new positive airway pressure device used for the treatment of obstructive sleep apnea, was announced to have received U.S. FDA 510(k) clearance for its use across hospitals, sleep centers, and home settings.
Download a customized sample of the Positive Airway Pressure Devices Market report based on your specific needs: Download Now
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
