Inside the Small Molecule CDMO Market: Top Innovators

- Lonza
- Catalent, Inc
- Thermo Fisher Scientific Inc.
- Cambrex Corporation
- Bellen Chemistry
- Siegfried Holding AG
- Recipharm AB
- Eurofins Scientific
- Aurigene Pharmaceutical Services Ltd.
- CordenPharma International
Market Growth
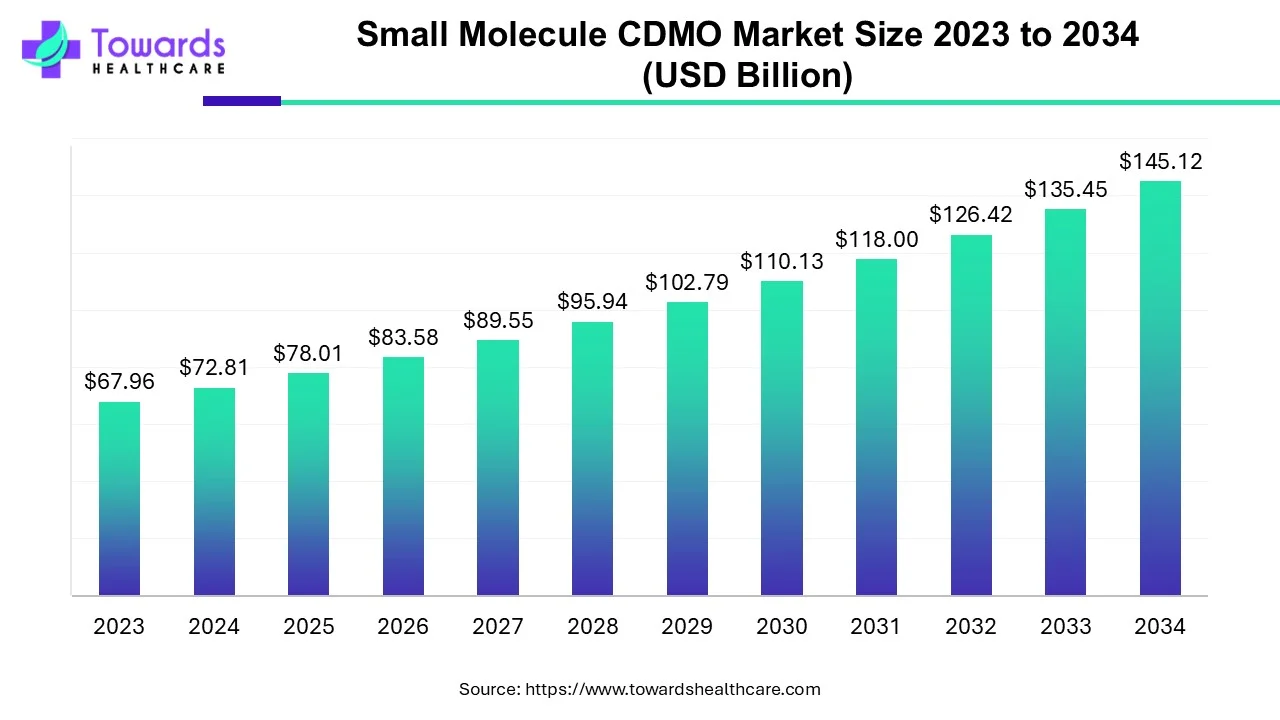
The global small molecule CDMO market size is calculated at US$ 72.81 in 2024, grew to US$ 78.01 billion in 2025, and is projected to reach around US$ 145.12 billion by 2034. The market is expanding at a CAGR of 7.14% between 2025 and 2034. The rise in chronic conditions and demand for generic medicines is driving the small molecule CDMO market.
Market Overview
For almost a century, small-molecule medications have formed the backbone of the pharmaceutical sector. Any low molecular weight organic substance is referred to by this definition. New and inventive methods for creating small-molecule medications are made possible by the quick development of biopharmaceutical research and technology. In order to successfully navigate small molecule API development and manufacturing from start to finish, biotechnology and pharmaceutical companies seeking to outsource critical components of small molecule drug development and manufacturing should look to a top-tier CDMO with extensive industry experience. Technology breakthroughs, shifting consumer needs, and stricter regulations are all contributing to the quick changes in the small molecule contract manufacturing industry.
Small Molecule CDMO Market Trends
In November 2024, Arcturus Therapeutics, Inc., a commercial mRNA medication and vaccine business, and Axcelead, Inc., which oversees a collection of top pharmaceutical and healthcare platform firms, partnered to develop ARCALIS. In addition to contract development and manufacturing organization (CDMO) services, ARCALIS is involved in the development of mRNA medicines and vaccines. In order to produce mRNA vaccines domestically, the business intends to set up a whole system that includes making formulations and active medicinal components as well as developing manufacturing methods.
- In April 2024, Biologics, Curida, and small molecules Signet Healthcare Partners made a private equity investment in CDMO.
Statistical Data
In 2024, the FDA approved 50 new drugs, slightly down from 55 in 2023, but this raised the ten-year average to 46.5 approvals per year, the highest in over 20 years. According to Nature Reviews Drug Discovery, 64% of these were small molecules (32 drugs), 32% were protein-based (16 drugs), and 4% were oligonucleotides (2 drugs). Oncology saw the most approvals, with 15 drugs (30%), followed by dermatology and hematology with 6 drugs each (12%), and cardiovascular treatments with 5 drugs (10%). Although the total number of approvals and small molecules increased and decreased since 2022, the percentage of small molecules has consistently risen, from 57% in 2022 to 62% in 2023 and 64% in 2024. Over the last five years, small molecules have accounted for 64.6% of FDA approvals, underscoring their major role in pharmaceutical innovation.
Growth Factor
- Rising Demand for Generic and Specialty Drugs: As patent expiration increases, the need for generic small-molecule drugs grows. Additionally, specialty small molecules, particularly in areas like oncology, are in high demand due to their targeted treatment and lower cost compared to biologics.
- Cost-Effective Manufacturing: Small molecules are cheaper to manufacture than biologics, making them an attractive option for pharmaceutical companies. Their simpler structure and production processes drive demand for contract manufacturing services as companies seek to optimize production costs and efficiency.
- Outsourcing and Biotech Growth: Biotech companies and pharma firms are increasingly outsourcing small-molecule development and manufacturing to CDMOs. This trend is fueled by the need for specialized services, flexibility, and cost savings, as companies focus on research and development while leaving manufacturing to experts.
Top Companies’ Revenue Info.
Recipharm is a world-leading CDMO that focuses on the development and manufacturing of biologics and small molecules. The full-year 2024 revenue was €827 million, an increase of 7% from 2023. In 2025, the company focuses on expanding continuous manufacturing, high-potency product development and manufacturing, and further investments in sterile fill-finish.
Catalent, Inc. is a leading global CDMO company that provides CDMO services, delivery technologies, and manufacturing solutions to develop pharmaceuticals, biologics, and consumer health products. The net revenue of the company in the fourth quarter of 2024 was $1.3 billion, an increase of 24%. The net revenue in the fiscal year 2024 was $4.38 billion.
Latest Announcements by Industry Leaders
In October 2024, according to Thermo Fisher Scientific, Accelerator Drug Development is being launched. Michael Shafer, executive vice president and president of biopharma services at Thermo Fisher Scientific, stated in a press release that the company is revolutionizing drug development and manufacturing procedures to help clients advance vital programs to quickly, effectively, and efficiently address global health concerns.
Recent Developments in the Small Molecule CDMO Market
- In January 2025, BioCina and NovaCina, two global contract development and manufacturing organizations (CDMOs) announced a strategic combination that would establish a strong brand in small molecule and biopharmaceutical contract manufacturing.
- In September 2024, by investing $260 million to build a new, state-of-the-art facility in Sejong, South Korea, SK Pharmteco, a worldwide contract development and manufacturing organization (CDMO), announced a significant increase in its small molecule and peptide production capabilities.
Partner with our experts to explore the Small Molecule CDMO Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
