May 2026

The global biopharmaceuticals market size was calculated at USD 474.28 billion in 2025, to reach USD 533.57 billion in 2026 is expected to be worth USD 1540.14 billion by 2035, expanding at a CAGR of 12.5% from 2026 to 2035.
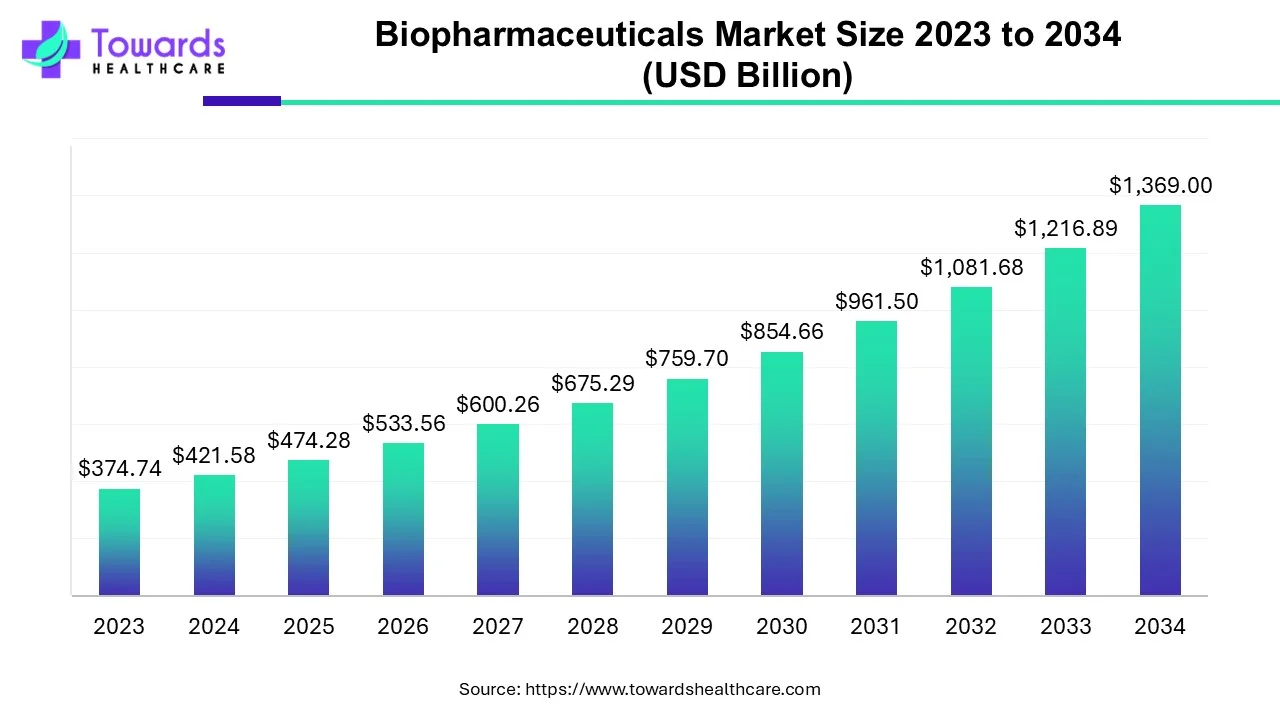
| Key Elements | Scope |
| Market Size in 2026 | USD 533.57 Billion |
| Projected Market Size in 2035 | USD 1540.14 Billion |
| CAGR (2026 - 2035) | 12.5% |
| Leading Region | North America |
| Market Segmentation | By Type, By End-user, By Application, By Region |
| Top Key Players | Abbott Laboratories, Amgen, Inc., Biogen, Inc., Eli Lilly and Company, F. Hoffmann-La Roche, Ltd., Johnson & Johnson, Merck & Co., Inc., Novo Nordisk A/S, Pfizer, Inc., Sanofi |
A biopharmaceutical, also known as a "biologic," is a medical therapeutic (or drug) derived from a biological source, such as a cell. Biotechnology is used to directly extract proteins and nucleic acids from biological sources to produce biopharmaceuticals, which are drugs used in medicine. Transgenic organisms are genetically modified plants and animals that are used in the production of biopharmaceuticals, which are pharmaceuticals made primarily from living organisms. This controversial technology is still in the trial stage.
The AI in biopharmaceuticals market size is calculated at US$ 1.55 billion in 2024, grew to US$ 2.05 billion in 2025, and is projected to reach around US$ 24.49 billion by 2034. The market is expanding at a CAGR of 32.27% between 2025 and 2034.
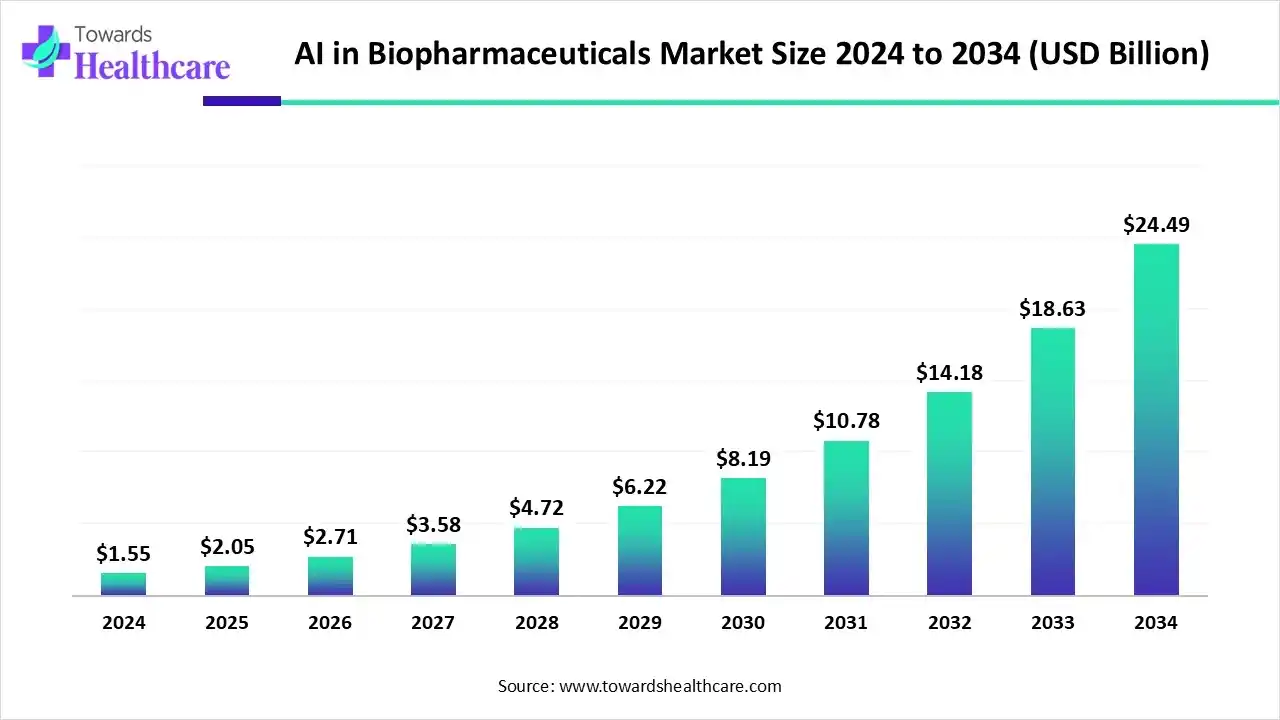
Artificial intelligence (AI) and machine learning (ML) algorithms have become an integral part of biopharmaceutical research and manufacturing processes. AI and ML help to design novel biopharmaceuticals based on a patient’s medical conditions, thereby developing personalized treatment regimens. They can also be used to assess the interactions or effects of a biopharmaceutical after administering in the body.
They can predict the pharmacokinetic and pharmacodynamic properties of a newly developed biopharmaceutical at an early stage of drug development. Moreover, AI and ML can be integrated into manufacturing, enhancing efficiency, accuracy, and reproducibility. They eliminate human intervention in manufacturing, reducing manual errors. Additionally, AI and ML algorithms can streamline supply chain management, leading to timely delivery to different geographical locations.
The biopharma industry is experiencing a golden era of innovation, marked by its largest and most diverse clinical pipeline in history—a culmination of decades of groundbreaking research. The biopharma research and innovation is driven by increasing funding from government and private institutions. This leads to the development of an expansive pipeline of drugs by various pharmaceutical and biotechnology companies.
Government funding includes an estimated $18 billion for Operation Warp Speed and $48 billion annually from the National Institutes of Health. Additionally, the private sector has contributed significantly, with an estimated $146 billion in life sciences venture capital funding over the past three years.
The primary factor propelling the growth of this market is the enormous demand for biopharmaceuticals brought on by the increase in chronic diseases and the aging population. Additionally, it is anticipated that the ability of biopharmaceuticals to treat untreatable conditions will speed up the market's overall growth.
Additionally, the population's growing awareness of the efficacy and accessibility of biopharmaceuticals as well as the expanding healthcare sector is anticipated to temper market growth during the anticipated time frame. The market is expected to grow as a result of the increased efforts being made to develop the biopharmaceutical sector.
The risk of failure and the high cost of drug development are the main things preventing the global biopharmaceutical industry from expanding. Furthermore, the market is unable to grow internationally due to strict regulatory requirements, high development costs, and the cost of biopharmaceutical drugs. Currently, biopharmaceutical companies, health professionals, and stakeholders face numerous challenges and struggles during the development of new bio drugs and prior to the commercialization of these novel products. For example, according to pharmaceutical protocols, every bio-drug undergoes numerous steps and procedures prior to marketing, including animal and clinical studies. Indeed, this new medicine would be tested on laboratory animals during the development process; however, there may be a scarcity of animal samples or restrictions on their use because there are associations and animal rights organizations that consider the use of some animals in scientific tests or other scientific purposes to be a form of torture and unethical practice.
Moreover, it is predicted that the market will benefit greatly from the increased emphasis on research and associated investments. Numerous opportunities for market expansion will also be provided by the increased government funding for the development of the biopharmaceutical industry as well as the increased investments made by market participants in the research and development of new drugs and their clinical trials.
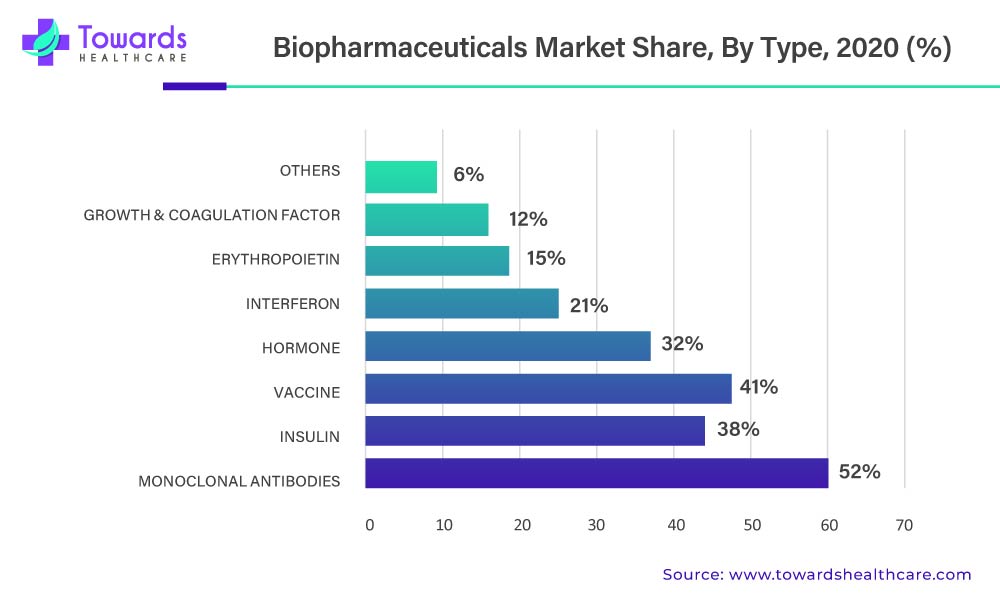
By type, the vaccine segment is expected to grow at the fastest rate in the market during the forecast period. The rising prevalence of infectious diseases, including bacterial and viral diseases, promotes the use and development of vaccines. Favorable government policies encourage increasing manufacturing of vaccines globally due to a surge in demand. The COVID-19 pandemic potentiated the demand for vaccines due to the lack of effective medications for its treatment. Vaccines are generally safe and effective and help protect people from diseases by stimulating the body’s natural defenses.
The successful application of monoclonal antibodies and antibody derivatives in therapeutics is the main factor influencing the segment's quick expansion. Monoclonal antibodies have been used as treatments for cardiovascular disease, cancer, rheumatoid arthritis, and multiple sclerosis. Healthcare systems are being heavily burdened by the COVID-19 outbreak. Researchers from all over the world are constantly working to address this growing burden. As a result, the market for monoclonal antibodies that target COVID-19 is anticipated to grow significantly.
Additionally, the demand for effective options for disease prevention, treatment, and complete eradication is growing, which is contributing to an increase in pharma R&D spending. Many pharmaceutical firms are concentrating on creating more potent medicines to treat fatal illnesses and enable patients to lead healthier lives with fewer disabilities. Global healthcare spending is essentially driving up R&D activities, which is boosting the number of bio-based pharmaceutical products on the market.
A new phase of biological therapies for neoplastic disorders, infectious diseases, and multiple sclerosis has also been made possible by cytokines and interferons (IFNs). These agents mediate their biological effects indirectly, through several immunological and other cells, or accessory proteins.
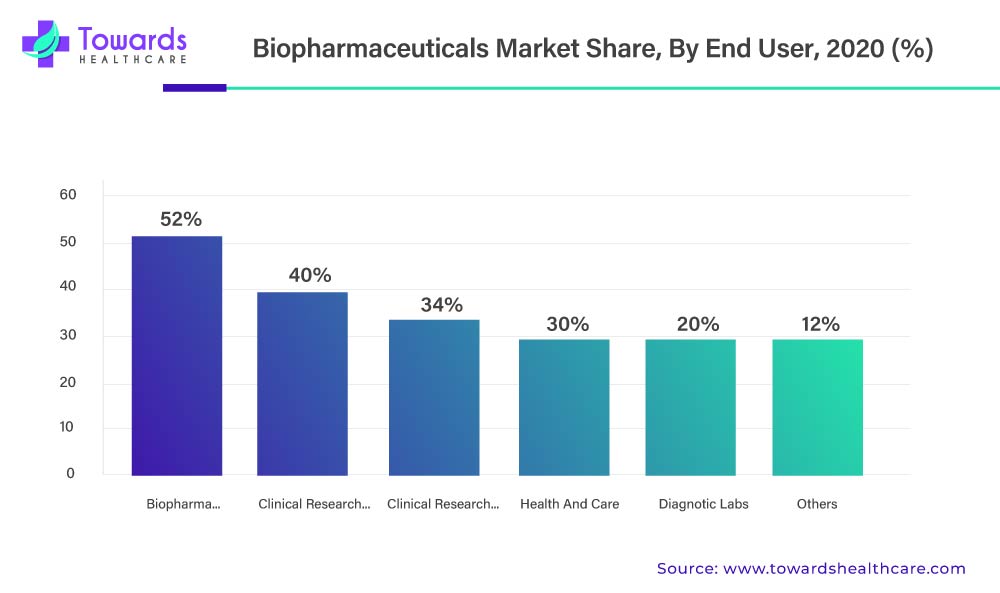
Depending on the end-use sector, the biopharmaceuticals market can be further divided into the following categories: health and care, biopharma companies, diagnostic labs, clinical research institutes, clinical research organizations (CROs), and others. The segment representing biopharma companies held the largest market share. This expansion is mainly due to the significant advancements in modern medicine and their use in treating complex diseases. This segment's growth is currently being further fueled by the recent improvements in molecular medicines and R&D made by numerous top industry players and governmental organizations.
Additionally, the Clinical Research Organizations segment is anticipated to expand at the fastest CAGR of 9.1% from as a result of an increase in research into the creation of new molecular drugs using molecular biology. Clinical research organizations (CROs) provide outsourcing research services to pharmaceutical and biotechnology companies. The availability of skilled professionals, suitable infrastructure, and the presence of specialized equipment are anticipated to drive the segment’s growth. The increasing number of CROs also potentiates the segment’s growth. There are a total of 4,232 CROs in the U.S. as of 2023.
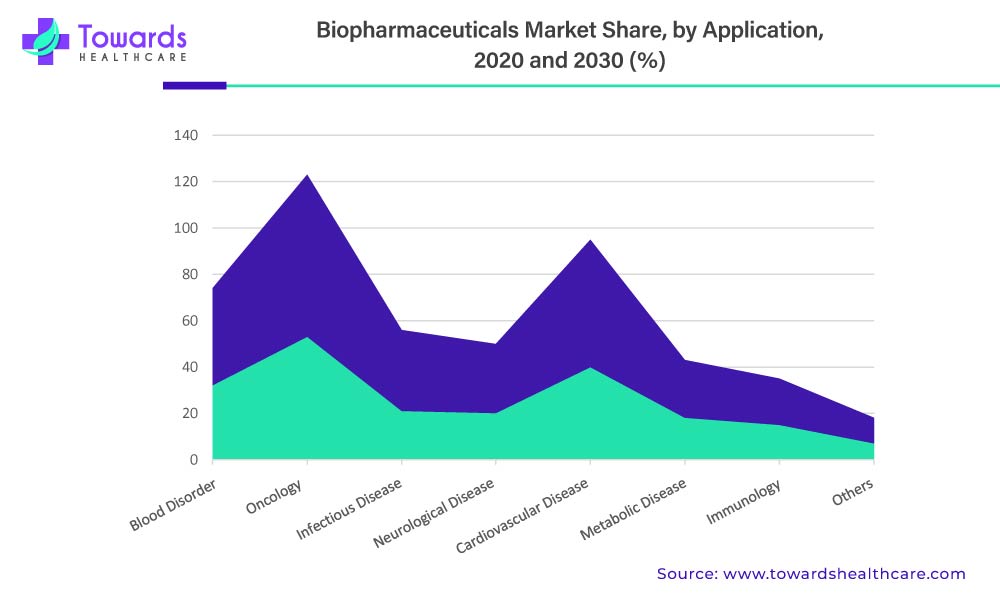
In terms of revenue, the oncology segment will dominate the global biopharmaceutical market in 2020. This is simply due to the increased use of biopharmaceuticals in the treatment of cancers such as lung cancer, breast cancer, colorectal cancer, and prostate cancer. Global cancer cases are increasing at an alarming rate. According to the International Agency for Research on Cancer, the global cancer burden is expected to reach 28.4 million by 2040, up 47% from 2020.
Cardiovascular disease, on the other hand, is expected to be the most opportunistic segment during the forecast period. According to World Health Organization, approximately 17.9 million people die each year as a result of cardiovascular diseases, accounting for approximately 32% of all global deaths. As a result, the increased prevalence of CVDs around the world, as well as rising investments by biopharmaceutical manufacturers in developing innovative CVD drugs, are expected to drive market growth during the forecast period.
The primary drivers of the biopharmaceuticals market in North America are the rising burden of chronic diseases and rising investments in research and development activities in the United States. The world’s center of life sciences innovation has long been acknowledged to be in the United States. It participates in international capital investments in biopharmaceutical startups.
The North American biopharmaceutical market is anticipated to grow as a result of the increase in clinical trials. The market is also driven by new product launches and favorable regulatory frameworks. The U.S. Food and Drug Administration approved a total of 55 new drugs, including biologicals and small molecules, in 2024. The increasing collaboration, partnerships, and mergers & acquisition activities support the development of biopharmaceuticals in North America.
Asia-Pacific is anticipated to grow at the fastest rate in the market during the forecast period. The rising prevalence of chronic disorders and the growing geriatric population drive the market. Favorable government policies to promote the pharmaceutical sector and provide better treatment regimens boost the market. The burgeoning pharmaceutical and biotechnology sector in Asia-Pacific, owing to the increasing number of startups and increasing investments and collaborations, augment market growth. Countries like China, Japan, Thailand, and India have suitable manufacturing infrastructure, which encourages foreign manufacturers to set up their manufacturing facilities in these countries.
Ray Mercier, President of Thermo Fisher Scientific’s single-use technologies business, commented on the launch of bio-based films that biopharma manufacturers are focused on maximizing productivity, accelerating time-to-market, and generating efficiencies along with improving the overall sustainability of their operations. The bio-based films developed by the company allow customers to reduce their environmental impact.
One of the most important goals of biopharmaceutical innovation is to contribute to lower mortality and premature death rates. The Manhattan Institute recently published a research study on the reasons why the average life expectancy and longevity vary from country to country. The effective contribution of new bio drugs demonstrates that the more we use new bio drugs, the more we gain longevity and provide welfare to the population. Furthermore, tailoring drug formulations to specific patients and cases leads to improved cures and reduce premature mortality, both of which are important public health goals. Since the biopharmaceutical field was further revolutionized and initiated—to develop and discover new bio drugs—we have observed that this contributes to an increase in average life expectancy6, indicating a significant impact.
Bio-pharma firms can create drugs from biological components of substances known as biologics, which include sugars, proteins, nucleic acids such as DNA and RNA, and complex combinations of components. Biologics, or their precursors or components, are isolated from living sources such as humans, animals, plants, fungi, or microbes. Because they are structurally identical, these drugs are frequently more effective and have fewer side effects.
The majority of biopharmaceuticals are derived from naturally occurring proteins. Biopharmaceuticals are also derived from recombinant proteins, which are special proteins that contain genes that scientists have manipulated. The changes cause the proteins to behave in a specific manner; because the changes are encoded in the genes, each subsequent generation of proteins carrying the modified genes will behave in the same manner.

Explore further to see how top companies are transforming the Biopharmaceuticals Market: https://www.towardshealthcare.com/companies/biopharmaceuticals-companies
By Type
By End-user
By Application
By Region
May 2026
May 2026
May 2026
May 2026