Top Companies in Sterile Injectables CDMO Market & Their Offerings

Take a closer look at the market; download your free sample customized for your business: Download for FREE
| Companies | Headquarters | Offerings |
| Boehringer Ingelheim GmbH | Ingelheim am Rhein, Germany | End-to-end CDMO services, including sterile fill-finish, biologics manufacturing, formulation development, and large-scale commercial production. |
| Baxter BioPharma Solutions | Deerfield, Illinois, USA | Sterile injectable manufacturing, aseptic fill-finish, formulation development, and support for clinical to commercial-scale production. |
| Vetter Pharma | Ravensburg, Germany | Specialized in aseptic fill-finish, prefilled syringes, cartridges, and packaging solutions for biologics and complex injectables. |
| Recipharm AB | Stockholm, Sweden | Sterile manufacturing, formulation development, clinical trial supply, and commercial production of injectable drugs. |
| Aenova Group | Starnberg, Germany | Contract manufacturing, including sterile injectables, formulation development, and packaging services for pharmaceutical companies. |
| Fresenius Kabi | Bad Homburg, Germany | Production of sterile injectables, infusion therapies, and contract manufacturing services with strong expertise in hospital-based products. |
| FAMAR Health Care Services | Athens, Greece | Sterile manufacturing, aseptic fill-finish, packaging, and supply chain solutions for pharmaceutical and biotech clients. |
Supply Chain Analysis
Clinical Trials
- Sterile injectable CDMOs play a vital role in supporting clinical trials by offering specialized services such as aseptic fill-finish, formulation development, stability studies, and compliance with regulatory standards set by organizations like FDA and EMA. These services ensure safe and efficient production of trial-ready injectables.
- Key players: Lonza Group, Catalent, Recipharm, Samsung Biologics.
Regulatory Approvals
- Sterile injectable CDMOs offer end-to-end packaging and serialization solutions to maintain product safety, sterility, and compliance with regulations like the DSCSA and EU FMD. Their services cover primary packaging such as vials, syringes, cartridges, and ampoules, along with secondary packaging like labeling, cartoning, and device assembly. They also enable unit-level serialization with full supply chain traceability. Key players: West Pharmaceutical Services, Gerresheimer, SCHOTT Pharma, and Stevanato Group.
Packaging and Serialization
- Sterile injectable CDMOs are required to follow strict quality and safety standards, including adherence to cGMP guidelines enforced by authorities such as the FDA, EMA, and CDSCO. Compliance ensures consistent product quality, sterility, and regulatory approval across global markets.
- Key Players: Pfizer CentreOne, Baxter BioPharma Solutions, Vetter Pharma, and WuXi Biologics.
The Pharmaceutical Companies Segment held a dominant position in the Market in 2025
| Segment | Share 2025 (%) |
| Pharmaceutical Companies | 60% |
| Biotech Companies | 25% |
| Contract Research Organizations (CROs) | 10% |
| Others | 5% |
The pharmaceutical companies segment dominated the sterile injectables CDMO market with shares of 60% in 2025 due to their large-scale production needs and expanding injectable drug pipelines. They increasingly rely on CDMOs to handle complex sterile manufacturing, reduce operational costs, and meet strict regulatory standards while focusing on core activities like drug discovery and commercialization.
The biotech companies segment held the second-largest market share of 25% in 2025 and is estimated to achieve the fastest CAGR of 10% due to their growing focus on biologics, biosimilars, and advanced therapies requiring sterile injectable formats. Limited in-house manufacturing capabilities and high development costs drive them to partner with CDMOs for specialized expertise, scalable production, and regulatory compliance, supporting their market expansion.
The contract research organizations (CROs) segment captured a 10% share of the market in 2025 and is growing due to increasing outsourcing of early-stage drug development and clinical trials services. CROs collaborate with CDMOs for a seamless transition from research to sterile manufacturing, reducing timelines and costs. Their expanding role in integrated development services and rising demand for complex injectable therapies are driving steady growth in this segment.
Market Growth
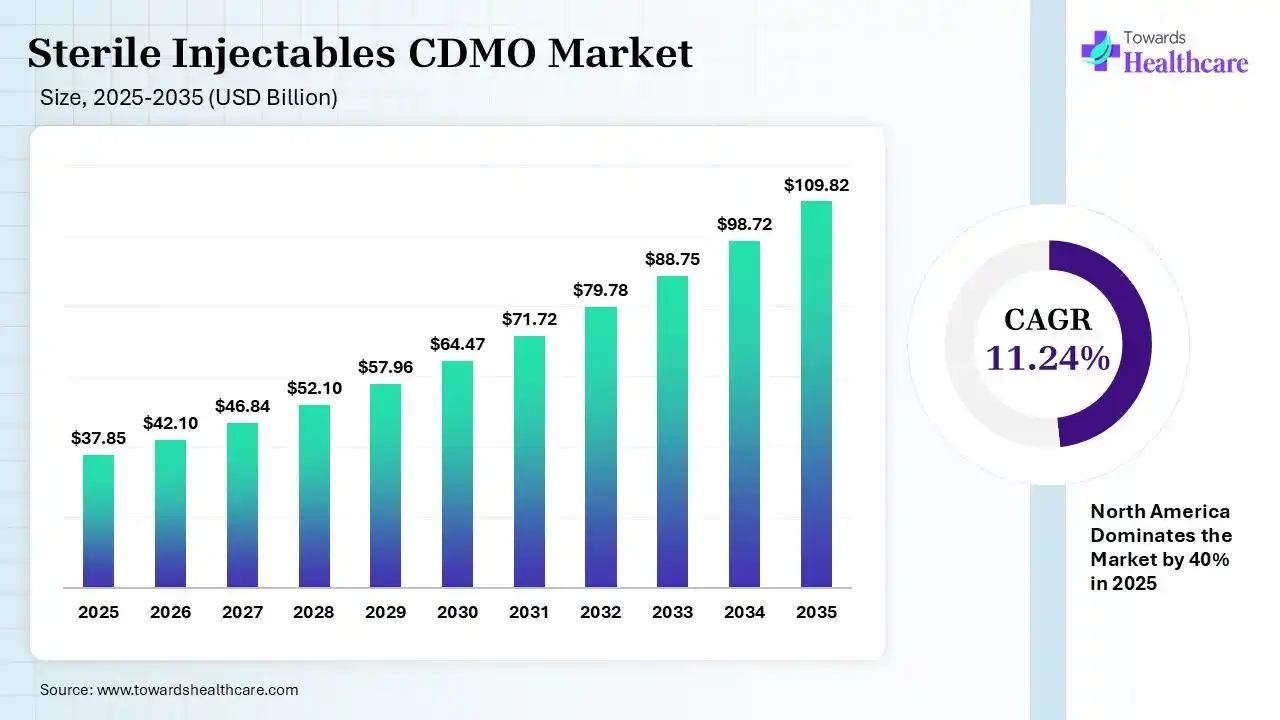
The global sterile injectables CDMO market size was estimated at USD 37.85 billion in 2025 and is predicted to increase from USD 42.1 billion in 2026 to approximately USD 109.82 billion by 2035, expanding at a CAGR of 11.24% from 2026 to 2035. The market is growing strongly as drugmakers outsource complex sterile drug manufacturing, driven by rising demand for biologics, cost optimization, and stricter quality/regulatory requirements.
What are the Recent Developments in the Sterile Injectables CDMO Market?
- In January 2026, Vetter Pharma revealed plans to expand its operations by setting up a new manufacturing facility in Saarland, Germany. The first phase involves an investment of around USD 568 million (EUR 480 million), aimed at enhancing its production capacity and strengthening its presence across the European sterile injectables market.
- In May 2025, FAMAR Health Care Services expanded its capabilities by acquiring a sterile manufacturing facility from MiP Pharma in Homburg, Germany. This move strengthens its regional presence and enhances expertise in high-value dosage forms, particularly lyophilization and aseptic fill-finish services.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
