Who are the U.S. Exoskeleton Market Top Vendors and What are Their Offerings?
Company Offerings
| U.S. Companies | Headquarters | Exoskeletons |
| Ekso Bionics Holdings, Inc. | San Rafael, California | EksoNR, Ekso Indego, and Ekso EVO |
| Lifeward Ltd. | Marlborough, Massachusetts | ReWalk Personal 7, MyoCycle FES, and ReStore |
| Sarcos Technology & Robotics Corp. | Salt Lake City, Utah | Guardian XO and Guardian XT |
| SuitX | Berkeley, California | Phoenix, Shoulder LegX, and BackX |
| Myomo, Inc. | Boston, Massachusetts | Myopro |
| Lockheed Martin Corporation | Bethesda, Maryland | ONYX and HULC |
| Levitate Technologies Inc. | San Diego, California | AIRFRAME |
| Harmonic Bionics, Inc. | Austin, Texas | Harmony SHR |
| Seismic Powered Clothing | Menlo Park, California | Seismic Suit |
| HeroWear, LLC | Nashville, Tennessee | Apex 2 |
U.S. Exoskeleton Market Value Chain Analysis
R&D
- The R&D of the U.S. exoskeleton focuses on the long-life battery integration, AI-driven intent recognition, soft robotic textile, and the development of more intuitive and lightweight wearable systems.
- Key players: Ekso Bionics Holdings, Inc., Sarcos Technology & Robotics Corp., HeroWear, LLC.
Clinical Trials and Regulatory Approvals
- The safety, skin integrity, joint alignment, risk of falls, functional independence, and diagnostic efficacy through the 10-meter walk test are evaluated in the clinical trials and regulatory approvals of the U.S. exoskeleton.
- Key players: Ekso Bionics Holdings, Inc., Myomo, Inc., Lifeward Ltd.
Packaging and Serialization
- The packaging and serialization of the U.S. exoskeleton involves the use of custom-molded protective cases for medical units and heavy-duty crating for industrial systems, along with the unique device identification (UDI) and GS1-standards serialization.
- Key players: Ekso Bionics Holdings, Inc., Lifeward Ltd.
Distribution to Hospitals, Pharmacies
- The specialized medical equipment distributors and direct sales forces are responsible for the distribution of the exoskeletons across the U.S. hospitals and pharmacies.
- Key players: Ekso Bionics Holdings, Inc., Lifeward Ltd.
Patient Support and Services
- The gait training programs, 24/7 technical support, and insurance reimbursement assistance are provided in the patient support and services of the U.S. exoskeletons.
- Key players: Ekso Bionics Holdings, Inc., Myomo, Inc., Lifeward Ltd.
Market Growth
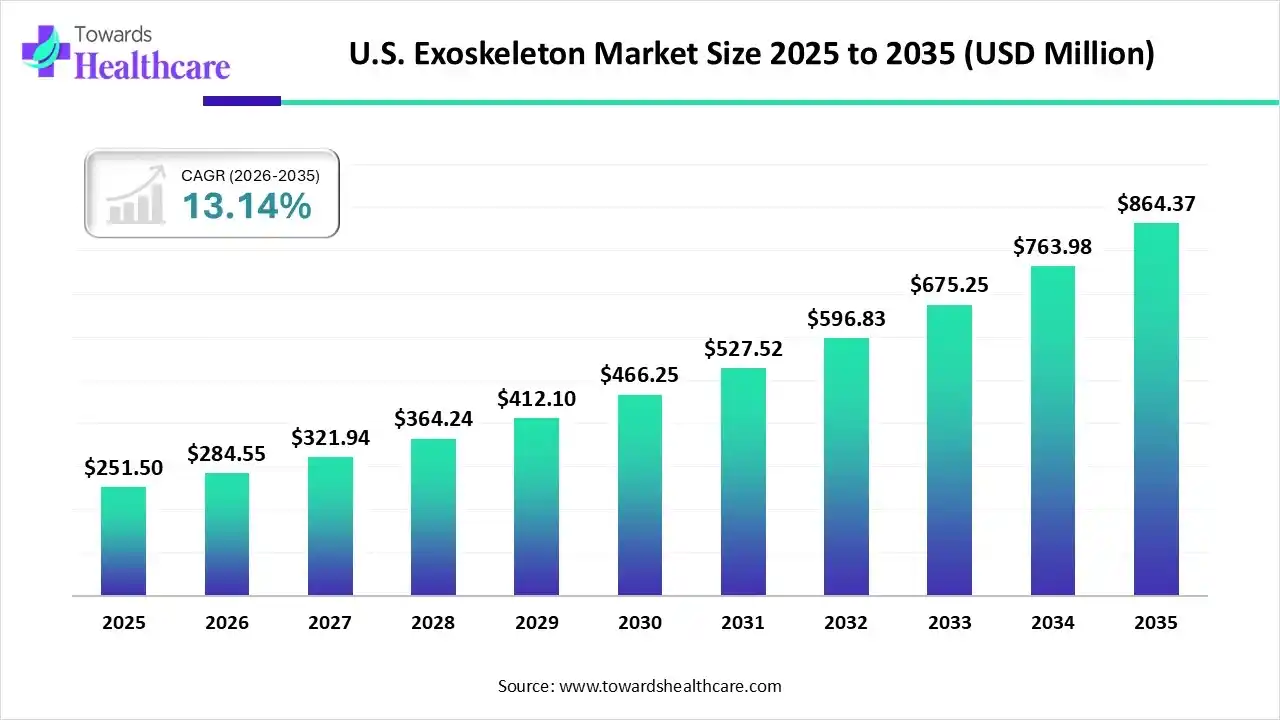
The U.S. exoskeleton market size was estimated at USD 251.5 million in 2025 and is predicted to increase from USD 284.55 million in 2026 to approximately USD 864.37 million by 2035, expanding at a CAGR of 13.14% from 2026 to 2035.
Recent Developments in the U.S. Exoskeleton Market
- In February 2026, a definitive agreement with an aim to acquire certain technology assets was announced between Lifeward Ltd. and Skelable Ltd., where the key engineering team of Skelable Ltd. will offer industrial, electrical, mechanical, and software design expertise supporting the launch of Lifeward's new upper body exoskeleton.
- In February 2026, to enhance the access and evaluation of the ReWalk Personal Exoskeleton for people with spinal cord injury (SCI), a collaboration between Lifeward and Shirley Ryan AbilityLab was announced, where they will combine clinical evaluation, patient support, and training with ReWalk.
Turn complex U.S. Exoskeleton Market data into clear, confident decisions - Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
