Top Pharmaceutical Companies in USA
- Bristol Myers Squibb
- Amgen, Inc.
- Regeneron Pharmaceuticals, Inc.
- Biogen Inc.
- Gilead Sciences
- Vertex Pharmaceuticals
- Moderna Inc.
- Sanofi U.S.
- GlaxoSmithKline (GSK)
- AstraZeneca Pharmaceuticals LP
- Viatris Inc.
- Boehringer Ingelheim USA
U.S. Pharmaceutical Sector: Fueling America’s Health
The U.S. pharmaceutical market is fueled by rising chronic disease prevalence, aging demographics, advanced healthcare infrastructure, and the dominance of global pharma leaders headquartered or heavily operating in the U.S. It represents the largest and most advanced pharmaceutical ecosystem globally, encompassing research, manufacturing, distribution, and commercialization of prescription and over-the-counter (OTC) drugs. It is characterized by high R&D spending, rapid biologics and biosimilars adoption, a strong generics market, and increasing digital integration in drug discovery and patient care.
Market Growth
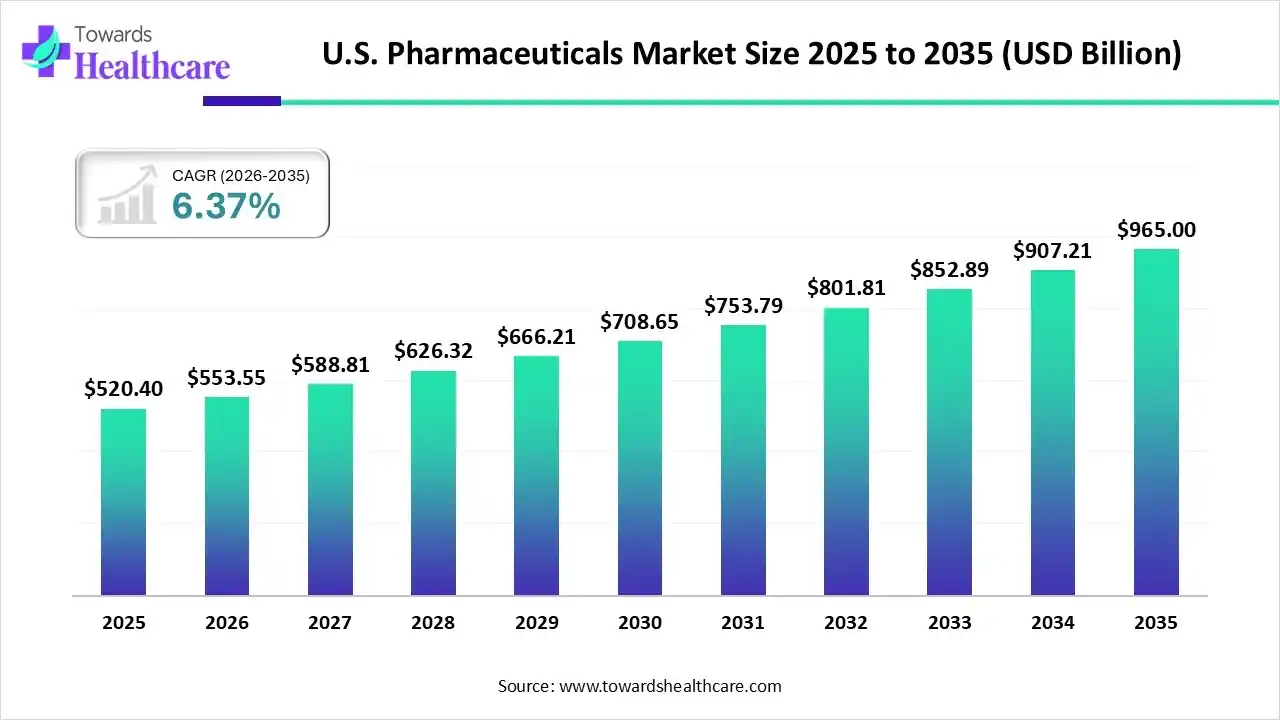
The U.S. pharmaceutical market size is calculated at USD 520.4 billion in 2025, grew to USD 553.55 billion in 2026, and is projected to reach around USD 965 billion by 2035. The market is expanding at a CAGR of 6.37% between 2026 and 2035.
U.S. Pharmaceutical Market Outlook
- Industry Growth Overview: The market is expected to expand rapidly, due to increasing investments, rising collaboration among key players, and favorable trade policies. Government bodies are encouraging companies to move more of their manufacturing and production facilities back to U.S. soil.
- Major Investors: Government and private organizations provide funding for various purposes, such as research and manufacturing. Alta Partners, Advent International, and Carlyle Group are some private equity firms that invest in the pharmaceutical industry in the U.S.
- Startup Ecosystem: The startup ecosystem is maturing, driven by venture capital investments and technological innovations. Funding helps develop novel medical technologies and a support system of specialized vendors and service providers. A well-functioning patent system secures property rights in inventions, supporting the launch of new ideas to the marketplace.
Company Landscape
Johnson & Johnson
Corporate Information:
- Headquarters: New Brunswick, New Jersey, USA
- Year Founded: 1886
- Ownership Type: Publicly traded company (NYSE: JNJ)
History and Background
- Founded by the Johnson brothers in 1886 to produce sterile surgical dressings.
- Over time, expanded through acquisitions into pharmaceuticals, medical devices, and consumer health.
- Notable recent development: separation of consumer-health business (Kenvue) and focus on “Innovative Medicine” and MedTech.
Key Milestones/Timeline
- 1886: Company founded.
- 2021–2023: Announced and completed spin-off of consumer-health business (Kenvue).
- 2024: Reported consolidated sales of approx. US $88.82 billion; Innovative Medicine sales US $57 billion.
- 2025: Announced investments of US$50 billion (since Jan 2024) in R&D and inorganic growth.
Business Overview
- Business Segments/Divisions:
- Innovative Medicine (pharmaceuticals) – largest segment.
- MedTech (medical devices & surgical) – major division.
- (Previously) Consumer Health – now largely separated into Kenvue.
Geographic Presence:
Operates worldwide. In 2024, U.S. sales were ~US$50.3 billion, and International ~US$38.5 billion.
Key Offerings:
- Pharmaceutical drugs covering oncology, immunology, neuroscience, cardiovascular/metabolism.
- Medical devices for orthopaedics, surgery, cardiovascular, and vision.
End-Use Industries Served:
- Healthcare providers (hospitals, clinics)
- Pharmaceutical/biotech markets
- Medical devices for surgical/implantable markets
- Global public health programmes
- Key Developments and Strategic Initiatives
Mergers & Acquisitions:
- 2024: Announced planned acquisition of Intra-Cellular Therapies (clinical-stage biopharma).
- Numerous smaller collaborations and licences (40+ in 2024) as part of inorganic growth.
Partnerships & Collaborations:
Partnership/licensing agreements, external R&D collaborations to accelerate pipeline growth.
Product Launches/Innovations:
- In 2024, J&J reported 26 platforms/products each generating over US$1 billion annually.
- Example: DARZALEX first brand to exceed US$3 billion sales in a single quarter.
Capacity Expansions/Investments:
- Announced US$2 billion advanced-technology facility in North Carolina, expanding biologic production.
- Strengthened manufacturing capacity globally.
Regulatory Approvals:
Numerous approvals globally through its Innovative Medicine business; detailed approvals not enumerated here.
Distribution Channel Strategy:
Global direct sales, partnerships, distribution via wholesalers, hospitals, and clinics; major wholesalers account for a significant proportion of revenue.
Technological Capabilities / R&D Focus:
- Core Technologies / Patents:
Strength in biologics, small molecules, and antibody-drug conjugates (via acquisitions like Ambrx Biopharma).
Research & Development Infrastructure:
Multiple global R&D sites; heavy investment (~US$50 billion since Jan 2024 for R&D and inorganic).
Innovation Focus Areas:
- Oncology, immunology, neuroscience, and advanced medical devices.
- Competitive Positioning
Strengths & Differentiators:
- Large-scale global presence; diversified across pharma, devices.
- Strong financials and track record of dividend growth (62nd consecutive year of increases).
- Broad therapeutic portfolio and pipeline in higher-growth areas.
Market Presence & Ecosystem Role:
One of the largest healthcare companies globally plays a key role in innovation-ecosystem partnerships, manufacturing, and the global supply chain of pharmaceuticals and medical devices.
SWOT Analysis:
- Strengths: scale, diversification, strong R&D/financial resources, global presence
- Weaknesses: legacy business complexity, regulatory/legal risks
- Opportunities: growth in biologics, cell therapies, MedTech innovation, emerging markets
- Threats: patent expirations, regulatory changes, competitive pressure in key therapeutic areas
Recent News and Updates
- Press Releases / Industry Recognitions / Awards:
2024 Annual Report: operational sales growth 7% excluding COVID vaccine impact.
Recent News:
In October 2025, announced plan to spin off its orthopaedics business (DePuy Synthes) into a standalone company — refocusing on pharma and MedTech.
AbbVie Inc.
Corporate Information:
- Headquarters: North Chicago, Illinois, USA
- Year Founded: January 1, 2013 (as a spin-out from Abbott Laboratories)
- Ownership Type: Publicly traded (NYSE: ABBV)
History and Background
- Created as a separation from Abbott Laboratories to focus on research-based pharmaceuticals.
- Over time, built a strong immunology and oncology portfolio (e.g., Humira, Skyrizi, Rinvoq) to drive growth and compensate for biosimilar competition.
Key Milestones/Timeline
- 2013: AbbVie is independent from Abbott.
- 2024: Reported revenue ~US$56.3 billion.
- 2025: Announced licensing deal for obesity-treatment drug GUB014295 valued up to US$2.2 billion.
- 2025: Received U.S. FDA approval for Emblaveo (intra-abdominal infection drug) in conjunction with Pfizer.
Business Overview
- Business Segments/Divisions:
Focused biopharmaceutical company (no large device or consumer business).
Geographic Presence:
Operates globally in 170+ countries.
Key Offerings:
Medicines treating immunology, oncology, neuroscience, aesthetics, and eye care.
End-Use Industries Served:
Healthcare providers/hospitals, specialty clinics, and global markets with pharmaceuticals.
Key Developments and Strategic Initiatives
- Mergers & Acquisitions:
Acquired or licensed drugs and pipelines (e.g., recent up-to US$2.2 billion licensing deal).
- Partnerships & Collaborations:
Collaboration with Pfizer on Emblaveo drug.
- Product Launches/Innovations:
Skyrizi & Rinvoq showing strong growth: Skyrizi up 58% and Rinvoq up 46% in Q4 2024.
- Capacity Expansions/Investments:
Focus on pipeline and internal innovation investment.
- Regulatory Approvals:
2025: U.S. FDA approval of Emblaveo for complicated intra-abdominal infections.
Distribution Channel Strategy:
Global commercial infrastructure delivering branded therapies across developed/emerging markets; patient assistance programmes (e.g., myAbbVie Assist).
Technological Capabilities / R&D Focus:
- Core Technologies / Patents:
Strong biologics/immunology franchise; extended exclusivity for Rinvoq until ~2037 gives competitive edge.
- Research & Development Infrastructure:
Pipeline in 75+ conditions, ~55,000 employees.
- Innovation Focus Areas:
Immunology, oncology, neuroscience, and emerging obesity treatment (via amylin-analog licensing).
Competitive Positioning
- Strengths & Differentiators:
- Specialty focus in high-growth therapeutic areas; extended patent protection on key products (Rinvoq).
- Leaner platform compared to conglomerates, enabling sharper focus.
- Market Presence & Ecosystem Role:
- Among the top biopharmaceutical companies globally, it plays a significant role in the specialty drug market and pipeline innovation.
SWOT Analysis:
- Strengths: focused specialty platform, strong product growth (Skyrizi/Rinvoq), global footprint
- Weaknesses: reliance on fewer blockbuster products, biosimilar risk (e.g., Humira) remains
- Opportunities: obesity treatment entry, expansion of pipeline, extended exclusivity of key drugs
- Threats: patent expirations, generic/biosimilar competition, regulatory pricing pressures
Recent News and Updates
- Press Releases / Industry Recognitions / Awards:
Recognized for strong growth and innovation in 2024–25.
- Recent News:
- 2025: Shares hit record high after Rinvoq exclusivity extended until 2037.
- 2025: Licensing deal for GUB014295 entry into obesity market valued up to US$2.2 billion.
- 2025: Emblaveo FDA approval for difficult intra-abdominal infections.
Value Chain Analysis – U.S. Pharmaceutical Market
R&D
The pharmaceutical research is driven by advances in genomic technologies and innovations in nanotechnology.
Key Players: WuXi AppTec, PPD, Fortrea, Gilead Sciences, and Johnson & Johnson.
Clinical Trials & Regulatory Approval
Pharmaceutical clinical trials assess the safety and efficacy of new drugs and medical devices.
Key Players: Nona Biosciences, IQVIA, Parexel, and Syneos Health.
Distribution to Hospitals, Pharmacies
Pharmaceuticals are either distributed by manufacturers or by distributors to wholesalers to U.S. hospitals and retail pharmacies.
Key Players: McKesson Corporation, Allied Medical Products, Inc., and Kingworld Medicines Group Ltd.
Recent Developments in the U.S. Pharmaceutical Market
- In November 2025, Senores Pharmaceuticals acquired a 51% stake in Zoraya Pharmaceuticals, LLC to strengthen its presence in the U.S. pharmaceutical market. Senores also benefits from expanded distribution and manufacturing capabilities and enhanced ability to navigate the U.S. regulatory landscape.
- In September 2025, AstraZeneca launched an online direct-to-consumer (DTC) medicine platform in the U.S. to sell diabetes and asthma drugs directly to patients at a step discount of 70%. Patients can purchase Airsupra (albuterol/budesonide) or Farxiga (dapaglifloxin) directly from a DTC pharmacy.
Partner with our experts to explore the U.S. Pharmaceutical Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
