RNAi becomes more widely applied in gene silencing and drug development because of its high level of specificity, lower side effects, and ease of synthesis, which drive the growth of the market.
What is RNA Interference Therapy?
RNAi is a significant tool in functional genomics; it allows basic research on all genes, which is vital to understand gene function and genomic interaction among genes, DNA, and RNA sequences. The results and uses are from low to full downregulation of single genes or gene families, in order to change plant features and enhance plant diversity while protecting natural resources. RNAi improves plant protection against diseases, pests, and environmental stresses. RNA interference is a biological process in which double-stranded RNA molecules sequence-specifically suppress gene expression. An RNA-interfering therapeutic is a gene-silencing medicine that inhibits the production of unwanted proteins, which cause disease.
Global Market Growth
The global RNA interference therapy market size was estimated at USD 4.85 billion in 2025 and is predicted to increase from USD 5.71 billion in 2026 to approximately USD 24.96 billion by 2035, expanding at a CAGR of 17.8% from 2026 to 2035.
RNA-driven therapeutics have transformed modern medicine, offering adaptable and precise modalities to moderate gene expression for a broad range of diseases, involving infectious diseases, genetic disorders, and cancer. Evolution and existing landscape of RNA therapeutics, encompassing major classes like small interfering RNAs (siRNAs), mRNA vaccines, antisense oligonucleotides (ASOs), and emerging RNA editing technologies such as CRISPR-Cas13.The integration of targeted RNA therapeutics, accurate RNA editing, and artificial intelligence-based design and clinical decision support heralds a novel era of individualized and adaptive therapies.
Therapy Area for RNA Therapeutic
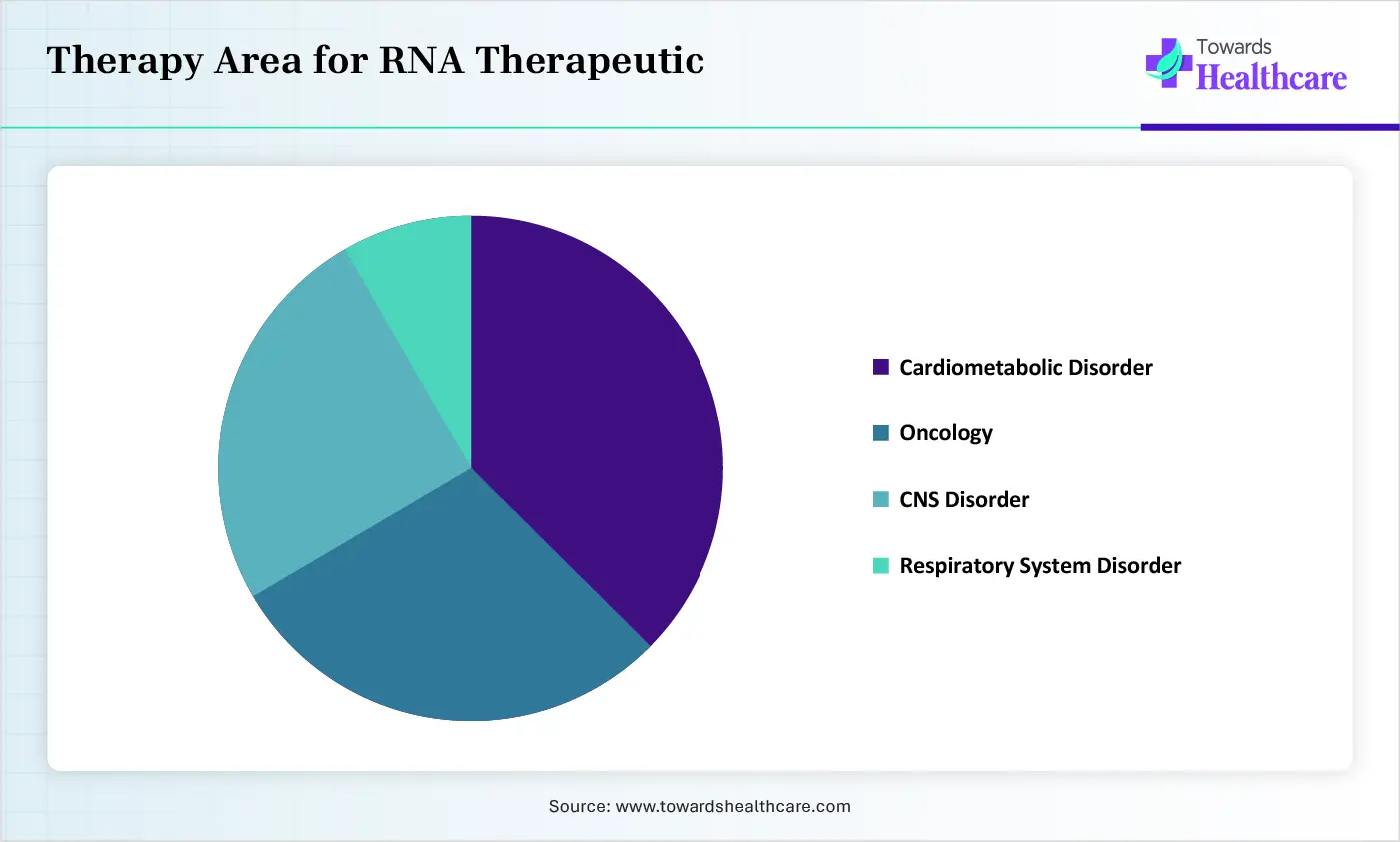
| Areas |
Percentages |
| Cardiometabolic Disorder |
32% |
| Oncology |
25% |
| CNS Disorder |
21% |
| Respiratory System Disorder |
8% |
| Ophthalmic Disorder |
6% |
| Other |
8% |
Market Segmentation Overview
- By molecule type, siRNA segment contributed the largest U.S. RNA interference therapy market share of 68% in 2025, as siRNA shows an increasing level of safety. As it acts on the post-translational stage of gene expression, it does not interact with DNA, therefore evading the common challenges of mutations and teratogenic effects related to gene therapy. siRNA gene silencing is highly effective. siRNA therapy is used jointly with other medications to improve therapeutic effectiveness and overcome drug resistance.
- By delivery method, the lipid nanoparticles (LNPs) segment contributed the largest market share of 52%, as LNPs allow for effective delivery of drugs, resulting in improved targeted delivery to specific cells or tissues. These LNPs are categorized into several types, including liposomes, solid lipid nanoparticles, nanostructured lipid carriers, and lipid-polymer hybrid nanoparticles.
- By application, the oncology segment contributed the largest market share of 34%, as RNA interference (RNAi) is a mechanism for gene silencing. Such a mechanism possesses uncanny capability in targeting cancer-based genes. RNAi is proposed as a strong cancer treatment substitute by allowing targeted silencing of genes, which disrupts the expression of oncogenes and restores the function of cancer suppressor genes.
- By route of administration, the intravenous segment contributed the largest U.S. RNA interference therapy market share of 46%, as intravenous injection is the most common parental route of medicine administration and avoids the liver's first-pass metabolism. Intravenous medications transport an immediate, fast-acting side effect, which is noteworthy in developing situations, such as cardiac arrest or narcotic overdose. They are significant for managing pain and nausea by quickly achieving therapeutic levels.
Top Companies in the U.S. RNA Interference Therapy Market
The top five organizations in this group dominate the U.S. RNA interference therapy market. Meanwhile, Alnylam Pharmaceuticals, it is the leader in RNAi, having pioneered the technology with major approved products. Arrowhead Pharmaceuticals focuses on broad RNAi applications, using its proprietary TRiM platform to target diseases. Dicerna Pharmaceuticals, known for its GalXC RNAi technology platform, specializes in chronic liver and genetic diseases. Arcturus Therapeutics Holdings, Inc. specializes in lipid nanoparticle (LNP) delivery systems for RNA-driven medicines. Intellia Therapeutics heavily focuses on CRISPR/Cas9; it utilizes LNP delivery systems for RNA-driven gene editing.
Segments Covered in the Report
By Molecule Type
- siRNA (Small Interfering RNA)
- Synthetic siRNA
- Dicer-substrate siRNA
- miRNA (MicroRNA)
- miRNA Mimics
- miRNA Inhibitors (Antagomirs)
- shRNA (Short Hairpin RNA)
- Viral Vector-based shRNA
- Plasmid-based shRNA
By Delivery Method
- Lipid Nanoparticles (LNPs)
- Ionizable Lipid-based LNPs
- Cationic Lipid-based LNPs
- Conjugate-based Delivery
- GalNAc Conjugates
- Peptide Conjugates
- Antibody Conjugates
- Viral Vector Delivery
- Lentiviral Vectors
- Adenoviral Vectors
- Polymer-based Nanoparticles
- PEGylated Polymers
- Dendrimers
By Application
- Oncology
- Solid Tumors
- Hematological Cancers
- Cardiovascular Diseases
- Hypercholesterolemia
- Hypertension-related Disorders
- Neurological Disorders
- Alzheimer’s Disease
- Parkinson’s Disease
- Infectious Diseases
- Viral Infections
- Rare Genetic Infections
- Metabolic Disorders
- Diabetes
- Obesity-related Disorders
- Rare Genetic Disorders
By Route of Administration
- Intravenous
- Subcutaneous (SC)
- Intrathecal
- Inhalation
- Oral
By End User
- Hospitals
- Specialty Clinics
- Research Institutes
- Pharmaceutical & Biotechnology Companies

