Top Companies in the Antibody-Drug Conjugates (ADCs) CMO and CDMO Market & Their Offering

Company Offerings
| Companies | Headquarters | Offerings |
| Sterling Pharma Solutions | United Kingdom | Provides ADC process development, bioconjugation, HPAPI and linker handling, analytical services, and GMP manufacturing for early- to mid-stage clinical programs |
| Recipharm | Sweden | Supports complex biologics manufacturing, sterile fill–finish, analytical development, and clinical supply, enabling ADC production through advanced formulation and aseptic capabilities. |
| Lonza | Switzerland | Offers end-to-end ADC solutions, including monoclonal antibody production, linker and payload synthesis, conjugation, analytical testing, and commercial-scale manufacturing. |
| Catalent | United States | Delivers biologics development, clinical and commercial manufacturing, sterile fill–finish, and advanced drug delivery support for ADCs. |
| Sartorius | Germany | Supplies critical bioprocessing technologies, single-use systems, and purification solutions that enable efficient and scalable ADC manufacturing for CMOs and CDMOs. |
| WuXi Biologics | China | Provides integrated ADC CRDMO services covering antibody development, payload and linker production, conjugation, analytical characterization, and commercial manufacturing. |
| Piramal Pharma Solutions | India | Offers HPAPI manufacturing, ADC development, formulation, and global clinical-to-commercial manufacturing through its multi-site CDMO network. |
What’s Driving the Rapid Growth of the Market?
Antibody-drug conjugates (ADCs) CMO and CDMO are specialized contract partners that provide outsourced manufacturing, development, and scale-up services for ADCs, supporting pharmaceutical companies across process development, clinical production, and commercial supply. The antibody-drug conjugates (ADCs) CMO and CDMO market is growing due to rising oncology drug pipelines, increasing preference for outsourcing complex biology, and the need for specialized conjugation and cytotoxic handling expertise. High development costs, strict regulatory requirements, and demand for faster clinical-to-commercial transition are pushing pharma companies to rely on experienced contract partners.
Antibody-drug Conjugates (ADCs) CMO and CDMO Market Growth
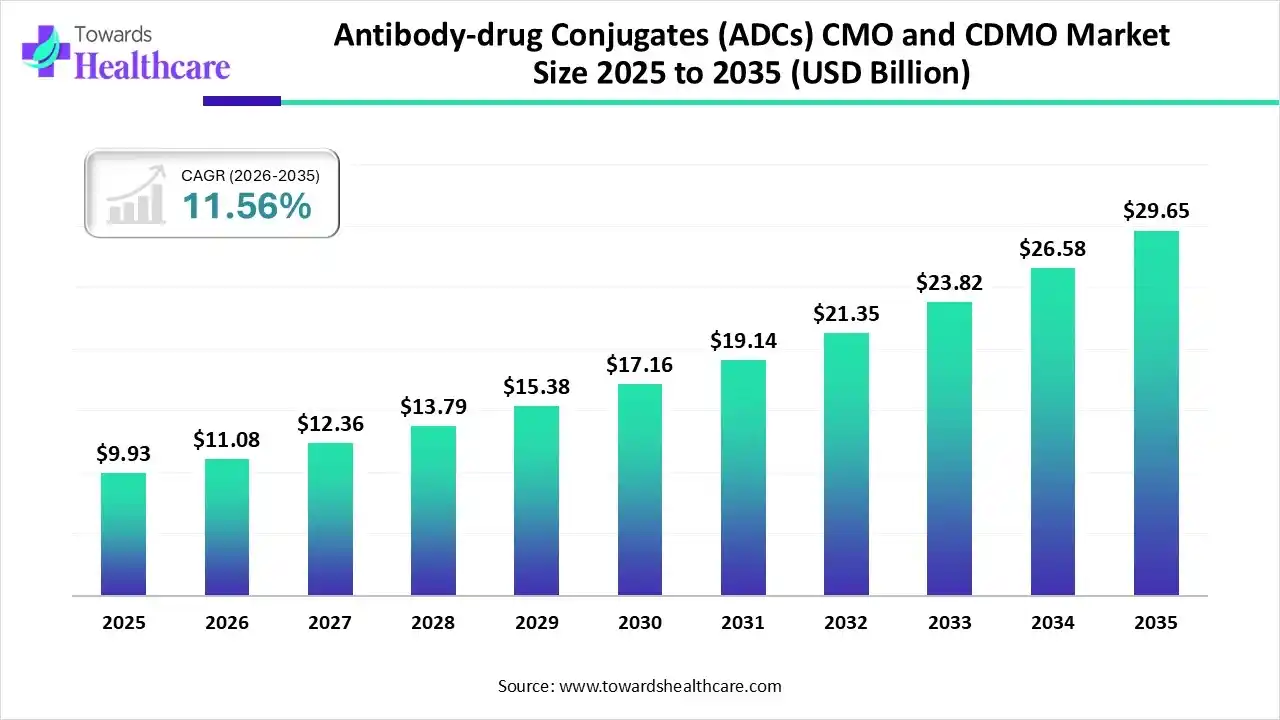
The global antibody-drug conjugates (ADCs) CMO and CDMO market size was estimated at USD 9.93 billion in 2025 and is predicted to increase from USD 11.08 billion in 2026 to approximately USD 29.65 billion by 2035, expanding at a CAGR of 11.56% from 2026 to 2035.
What are the Recent Developments in the Antibody-Drug Conjugates (ADCs) CMO and CDMO Market?
- In June 2025, Simtra BioPharma Solutions partnered with Merck KGaA to strengthen ADC development and manufacturing capabilities. The collaboration focuses on expanding expertise in bioconjugation, linker and payload production, formulation development, and sterile fill–finish to support advanced ADC programs.
- In March 2025, HAS Healthcare Advanced Synthesis acquired Cerbios-Pharma to strengthen innovation and expand high-quality manufacturing capabilities. The acquisition combines chemical and biological expertise with strong technical know-how, enabling the group to deliver a broader, integrated service portfolio tailored to the evolving needs of the global pharmaceutical industry.
Stop switching between reports - manage your entire Market data in one smart hub. Access the Dashboard
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
