Who are the Contract Pharmaceutical Manufacturing Market Top Vendors and What are Their Offerings?

One place for the Market insights, stats, strategies and 30+ more features for your business. Access the dashboard today
| Companies | Headquarters | Products Under Contract Pharmaceutical Manufacturing Services |
| Lonza Group | Basel, Switzerland | Biologics, small molecule API, antibody-drug conjugates, and cell and gene therapies |
| Thermo Fisher Scientific (Patheon) | Waltham, U.S. | Sterile injectable, biologics, and oral solid dosage forms |
| Catalent, Inc. | Somerset, U.S. | Advanced drug delivery, gene therapy, oral solids, and biologics |
| Samsung Biologics | Incheon, South Korea | Monoclonal antibodies, ADCs, mRNA vaccines |
| WuXi AppTec | Shanghai, China | Small molecules, peptides, cell and gene therapies, and oligonucleotides |
| Recipharm AB | Stockholm, Sweden | Sterile injectables, advanced therapeutic medicinal products, and oral solids |
| Boehringer Ingelheim (BioXcellence) | Ingelheim, Germany | Monoclonal antibodies, recombinant proteins, plasmid DNA, and fusion proteins. |
| Piramal Pharma Solutions | Mumbai, India | ADCs, potent APIs, sterile injectables, and complex oral solids |
| Akums Drugs & Pharmaceuticals | Haridwar, India | Tablets, capsules, injectables, and syrups |
| Siegfried Holding AG | Zofingen, Switzerland | Tablet, capsules, sterile vials, and ointments |
Contract Pharmaceutical Manufacturing Market Value Chain Analysis
R&D
- The R&D of the contract pharmaceutical manufacturing focuses on the development of advanced drug delivery systems, cell and gene therapy platforms, and AI-driven process optimization.
- Key players: Lonza Group, Catalent, Inc., WuXi AppTec.
Clinical Trials and Regulatory Approvals
- The manufacturing consistency, safety, and efficacy are evaluated in the clinical trials and regulatory approvals of the contract pharmaceutical manufacturing.
- Key players: Samsung Biologics, Thermo Fisher Scientific (Patheon), Lonza Group.
Formulation and Final Dosage Preparation
- The formulation and final dosage preparation of the contract pharmaceutical manufacturing focuses on optimization of drug stability, delivery formats, and solubility.
- Key players: Recipharm AB, Catalent, Inc., Thermo Fisher Scientific (Patheon).
Packaging and Serialization
- The packaging and serialization of the contract pharmaceutical manufacturing involves the use of secure primary and secondary containers, tamper-evident labelling, and unique track-and-trace coding.
- Key players: Catalent, Inc., Thermo Fisher Scientific (Patheon), Akums Drugs & Pharmaceuticals.
Distribution to Hospitals, Pharmacies
- The cold-chain logistics, last-mile delivery, and secure wholesale supply chains are responsible for the distribution of contract-manufactured pharmaceuticals to the hospitals and pharmacies.
- Key players: McKesson, Cardinal Health, and AmerisourceBergen.
Patient Support and Services
- The reimbursement assistance, specialized home-delivery, and adherence programs are provided in the patient support and services by the contract pharmaceutical manufacturing facilities.
- Key players: McKesson, Cardinal Health, and AmerisourceBergen.
Market Growth 2026-2035
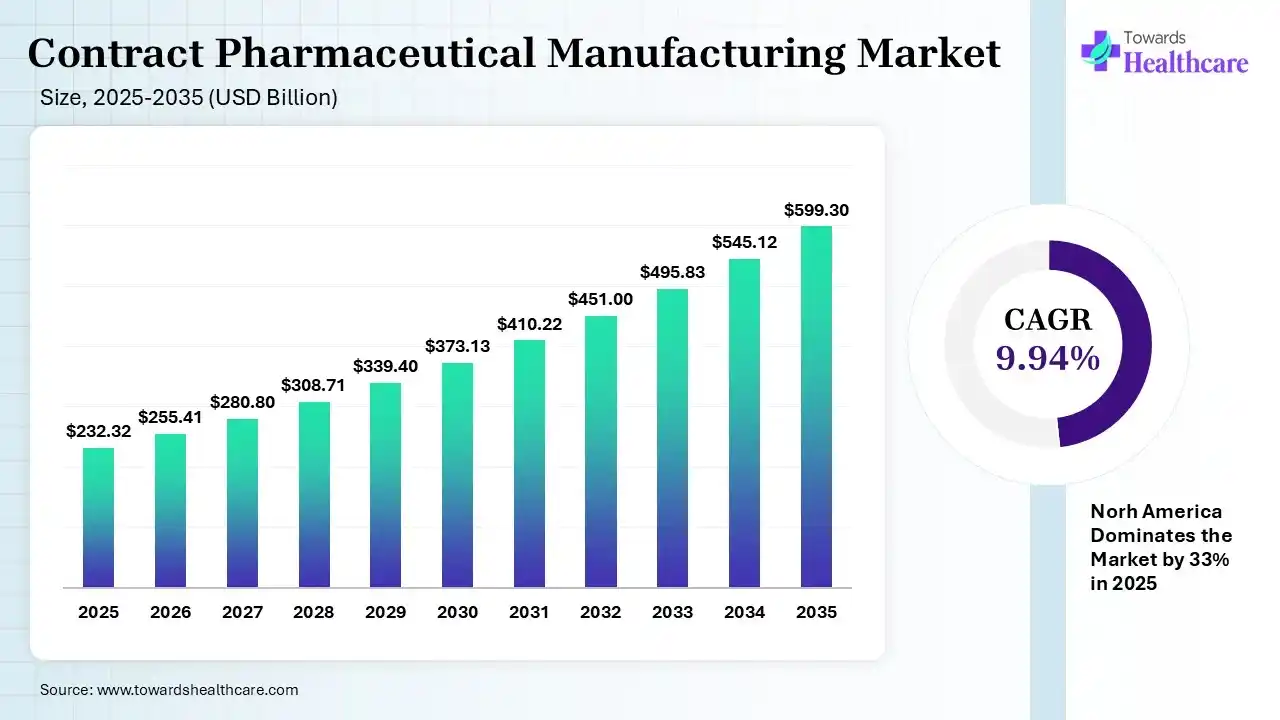
The global contract pharmaceutical manufacturing market size was estimated at USD 232.32 billion in 2025 and is predicted to increase from USD 255.41 billion in 2026 to approximately USD 599.3 billion by 2035, expanding at a CAGR of 9.94% from 2026 to 2035.
Recent Developments in the Contract Pharmaceutical Manufacturing Market
- In March 2026, a strategic collaboration between Suzhou Langrui Biopharmaceutical Co., Ltd. (LexBio) and Wuhan Hiteck Biopharmaceutical Co., Ltd. was announced, where they will integrate preclinical development and commercial manufacturing capabilities of Hiteck with LexBio's 1.2 billion-compound AI-powered discovery platform to enhance the small-molecule therapies for the treatment of inflammatory diseases.
- In March 2026, to support the pharmaceutical, diagnostics, and biotechnology organizations across the U.S., Canada, and Europe, and other markets by providing contract research organization ("CRO") services for custom aptamer discovery, machine-learning ("ML") classification, and biosensor enablement, was announced by the wholly-owned subsidiary, Triera Biosciences Ltd. of Zentek.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
