April 2026

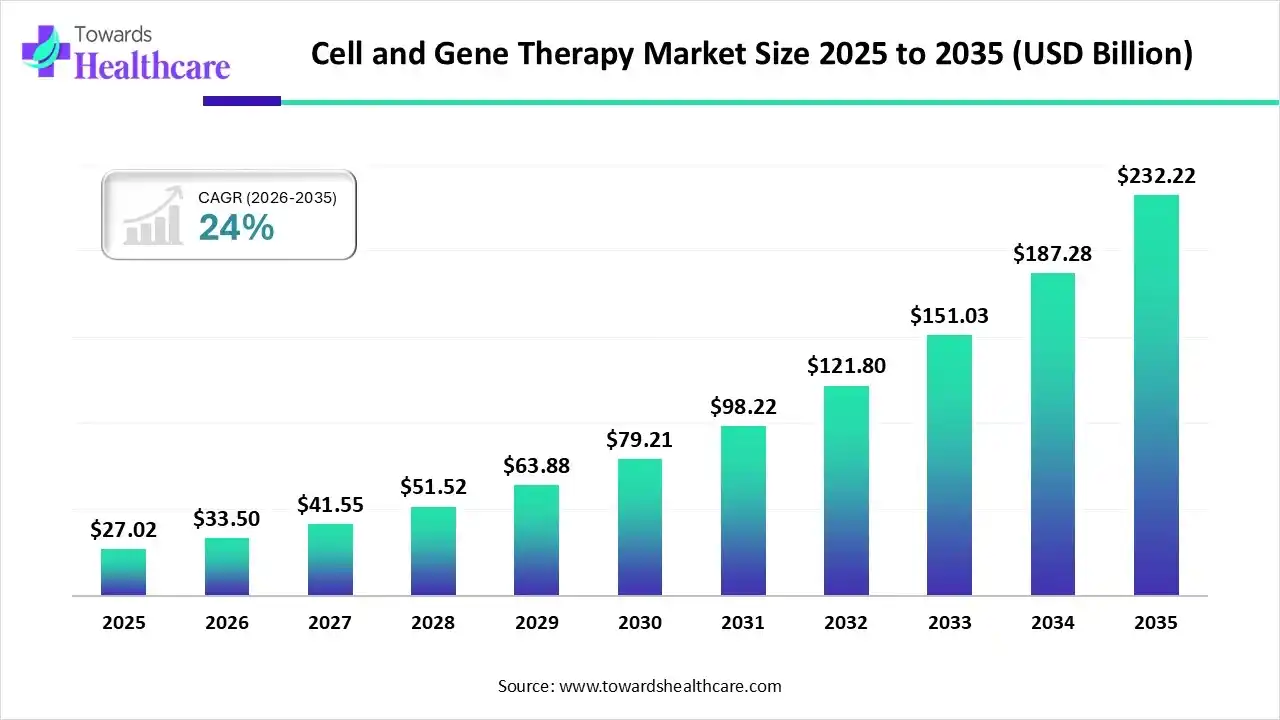
The cell and gene therapy market size touched US$ 8.94 billion in 2025, with expectations of climbing to US$ 10.44 billion in 2026 and hitting US$ 47.18 billion by 2035, driven by a CAGR of 18.1% over the forecast period.
| Key Elements | Scope |
| Market Size in 2026 | USD 10.44 Billion |
| Projected Market Size in 2035 | USD 47.18 Billion |
| CAGR (2026 - 2035) | 18.1% |
| Leading Region | North America by 49.88% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Therapy Type, By Therapeutic Class, By Delivery Method, By End-Users, By Region |
| Top Key Players | Alnylam Pharmaceuticals Inc., Amgen Inc., Biogen Inc., CORESTEM Inc., Dendreon Pharmaceuticals LLC., Helixmith Co. Ltd., JCR Pharmaceuticals Co. Ltd., Kolon TissueGene Inc., Novartis AG, and Pfizer Inc. |
Oncology Accounts for the Most Active Therapeutic Segment Through All the Stages of Development.
The cell and gene therapy market is a rapidly growing sector within the healthcare industry that involves the use of cellular and genetic materials to treat various diseases and medical conditions. Cell therapy involves the transplantation or administration of live cells into a patient's body to restore or improve cellular function, while gene therapy involves the introduction or alteration of genetic material to treat genetic disorders or modify the function of cells.
Cell and gene therapies have the potential to revolutionize the treatment of a wide range of diseases. They can be used for conditions such as cancer, genetic disorders, cardiovascular diseases, neurodegenerative diseases, and autoimmune disorders, among others. This broad applicability enhances the market's growth potential.
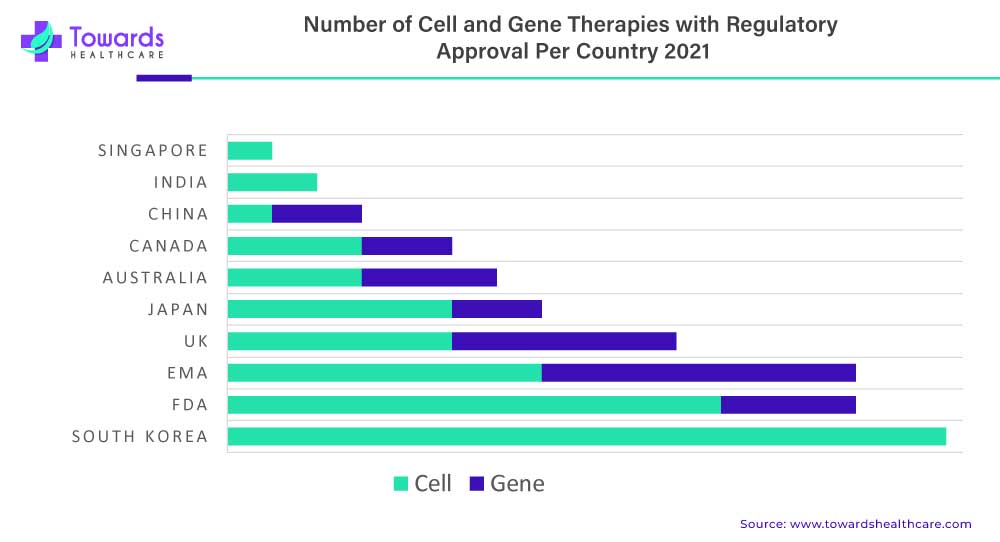
Oncology continued to be the most active therapeutic area of research across all stages of development, including preclinical through preregistration. In the field of gene and cell therapy, there were more than 1,300 candidates reported in development specifically for oncology in 2021. This highlights the significant focus and investment in developing innovative therapies to address cancer and improve patient outcomes.
Explanations:
Rising Prevalence of Chronic Diseases
The increasing number of people suffering from chronic illnesses such as cancer, genetic disorders, and autoimmune diseases is significantly driving the growth of the cell and gene therapy market. These advanced therapies offer targeted and potentially curative solutions by addressing the underlying genetic or cellular causes of disease, unlike traditional treatments that often only manage symptoms. With a rising global patient population and growing demand for more effective, long-term treatments, cell and gene therapies are becoming essential tools in modern medicine, offering hope where conventional methods have limited success.
High-Cost Associated Therapies
The high cost of cell and gene therapies is a major factor limiting market growth. These treatments often involve complex development, personalized manufacturing, and rigorous clinical testing, all of which drive up costs. As a result, many patients and healthcare systems face affordability challenges. Limited insurance coverage and high out-of-pocket expenses further restrict access, especially in low- and middle-income regions. This financial barrier slows widespread adoption, despite the therapies' medical potential.
Expansion of Cell and Gene Therapy Manufacturing
The expansion of manufacturing capacities presents a key opportunity for the cell and gene therapy market because it helps meet growing demand, lowers production costs, and improves accessibility. With more facilities and advanced technologies, companies can produce therapies at a larger scale, more efficiently, and with consistent quality. This also supports faster delivery of treatments, better patient access, and smoother commercialization. As manufacturing capabilities grow, the market can expand into new regions and treat more patients globally.
The biopharma sector is seeing a surge in investment and innovation, with both major global companies and emerging start-ups pouring resources into advanced therapies like cell, gene, and gene-modifying treatments. In 2018 alone, global investment in these advanced therapies reached about $13 billion. The following year, 19 major mergers and acquisitions in the cell and gene therapy (CGT) space were completed, totaling over $156 billion.
With more than 900 companies worldwide dedicated to CGTs and over 1,000 clinical trials in progress, the industry is poised for significant growth. Starting in 2025, we could see 10 to 20 new advanced therapies approved each year. Europe is a key player in this field, with 33% of these clinical trials taking place there.
Next-generation cell and gene therapies are making groundbreaking strides, offering potential cures for patients with few other treatment options, especially for rare and ultra-rare diseases. The primary focus areas for these therapies are cancer, musculoskeletal conditions, and eye diseases. Numerous products have already been approved and launched globally, and the number of clinical trials continues to increase.
In Europe, these therapies are classified as Advanced Therapeutic Medicinal Products (ATMPs). They are driven by a range of innovative scientific advancements, including CAR-T and TCR-T therapies, stem cells, siRNA, oligonucleotides, gene editing technologies (such as CRISPR, Zinc Fingers, and TALENs), and viral transfection methods.
| Segment | Share 2025 (%) |
| Cell Therapy | 64.78% |
| Gene Therapy | 35.22% |
Which Therapy Type Segment Dominated the Cell and Gene Therapy Market?
The cell therapy segment registered its dominance by 64.78% over the global cell and gene therapy market. Cell therapy is the transplantation of human cells to replace or repair damaged tissues. The therapy is used to prevent or treat several chronic disorders. Stem cell-based therapies are one of the most common types of cell therapy used clinically. The segment’s growth is attributed to the growing research and development activities, technological advancements, innovative products, and the increasing number of clinical trials. As of November 2024, 21,746 cell therapy-based clinical trials have been registered on clinicaltrials.gov.
The gene therapy segment captured a revenue share of 35.22% of the market in 2025 and is expected to grow at the fastest CAGR during the forecast period. Gene therapy grows rapidly due to technological breakthroughs like CRISPR, advancing from experimental science to curative, one-time treatments for previously untreatable genetic disorders and cancers. Increased regulatory approvals, heavy R&D investment, and precision medicine advancements are accelerating commercialization and clinical adoption worldwide.
| Segment | Share 2025 (%) |
| Oncology | 39.48% |
| Genetic Disorders | 12.05% |
| Neurological Disorders | 4.24% |
| Cardiovascular Diseases | 4.68% |
| Ophthalmology | 5.22% |
| Musculoskeletal / Orthopedic Disorders | 21.24% |
| Infectious Diseases | 5.39% |
| Metabolic Disorders | 3.44% |
| Others | 4.26% |
Which Therapeutic Area Led the Market in 2025?
The oncology segment led the cell and gene therapy market with 39.48% share in 2025. The rising prevalence of cancer, the latest innovations, and favorable government initiatives to effectively treat cancer drive the segment’s growth. Furthermore, government organizations and private companies are collaborating to develop new cell and gene therapies for tackling the rising cases.
The musculoskeletal/orthopedic disorders segment held 21.24% of the market share in 2025. It drives market growth by creating high demand for regenerative treatments of debilitating, chronic conditions. These therapies offer targeted repair of damaged tissues, replacing traditional surgery. Rising aging populations and injuries significantly boost research investments.
The genetic disorders segment held 12.05% share of the cell and gene therapy market in 2025 and is expected to grow at the fastest CAGR during the forecast period. Genetic disorders drive the market by offering curative potential for previously untreatable, rare conditions. High unmet needs, advancements in gene editing (CRISPR), and increasing regulatory approvals for single-dose treatments boost investment and development in this space.
The infectious diseases segment held 5.39% share of the market in 2025. The rising incidence of infectious diseases and the COVID-19 pandemic increase the demand for cell and gene therapy for infectious diseases. The rising demand for genome editing methods like CRISPR/Cas for infectious diseases augments the market.
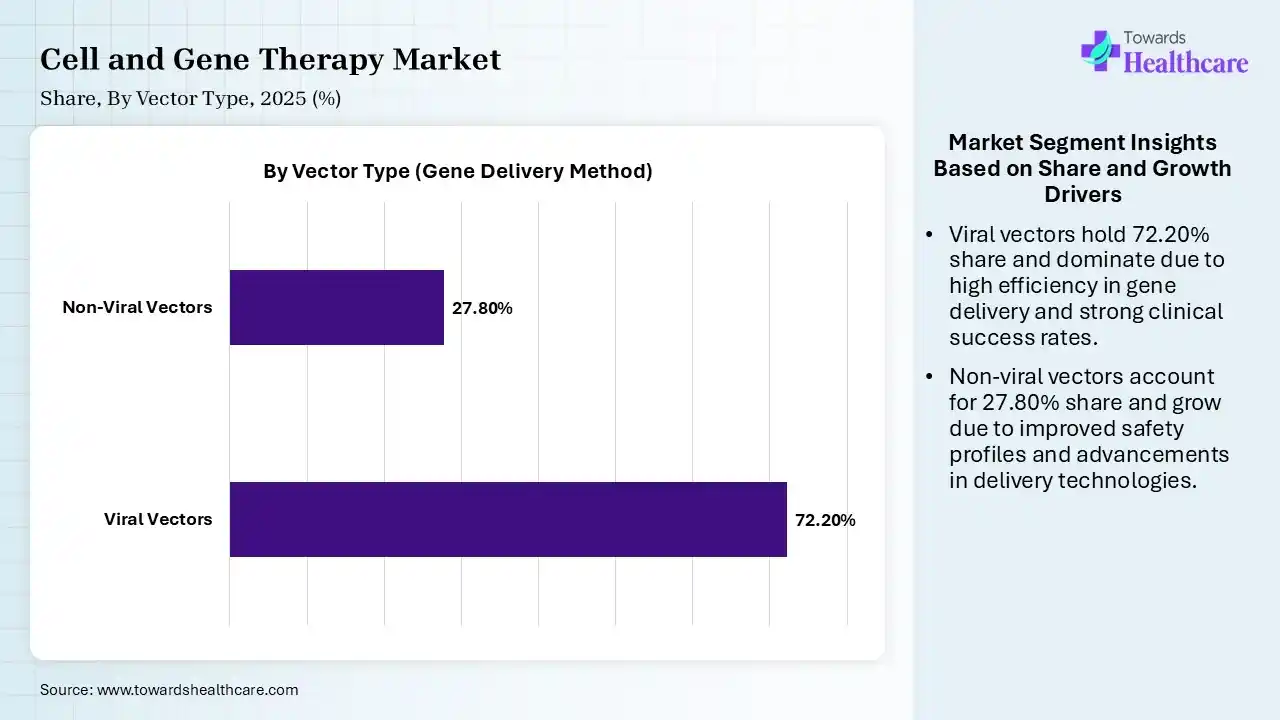
| Segment | 2025 Market Share (%) |
| Viral Vectors | 72.20% |
| Non-Viral Vectors | 27.80% |
The Viral Vectors Segment Led the Market in 2025
The viral vectors segment dominated the cell and gene therapy market with 72.20% share in 2025. Viral vectors are favored for gene/cell therapy because they efficiently enter cells, deliver genetic cargo, and offer stable expression. Modified for safety, they exploit natural transduction mechanisms to target specific tissues, providing long-term, one-time treatment options for inherited diseases.
The non-viral vectors segment held 27.80% share of the market in 2025 and is expected to grow at the fastest CAGR during the forecast period. Non-viral vectors, particularly lipid nanoparticles and polymers, are experiencing rapid usage growth in gene/cell therapies, driven by improved safety, lower immunogenicity, scalable manufacturing, and versatility in delivering large genetic payloads.
The In-House Segment Dominated the Market in 2025
The in-house segment led the cell and gene therapy market with 45.50% share in 2025. In-house facilities dominate gene/cell therapies by offering superior control over complex processes, safeguarding proprietary intellectual property, accelerating clinical-to-commercial speed, ensuring sterile quality, and improving flexibility to meet personalized patient needs.
The contract development & manufacturing organizations (CDMOs) segment held 38.69% share of the market in 2025 and is expected to grow at the fastest CAGR during the forecast period. CDMOs accelerate gene/cell therapy growth by providing specialized expertise, manufacturing capacity, regulatory compliance, and scalable platforms, allowing developers to advance complex, high-cost, and innovative treatments from clinical trials to commercialization.
The hybrid models segment captured 15.81% share of the market in 2025. Hybrid models grow in cell/gene therapy by balancing in-house control over proprietary IP during early stages with outsourced scale-up expertise, reducing high capital costs, mitigating risk, and enhancing manufacturing flexibility.
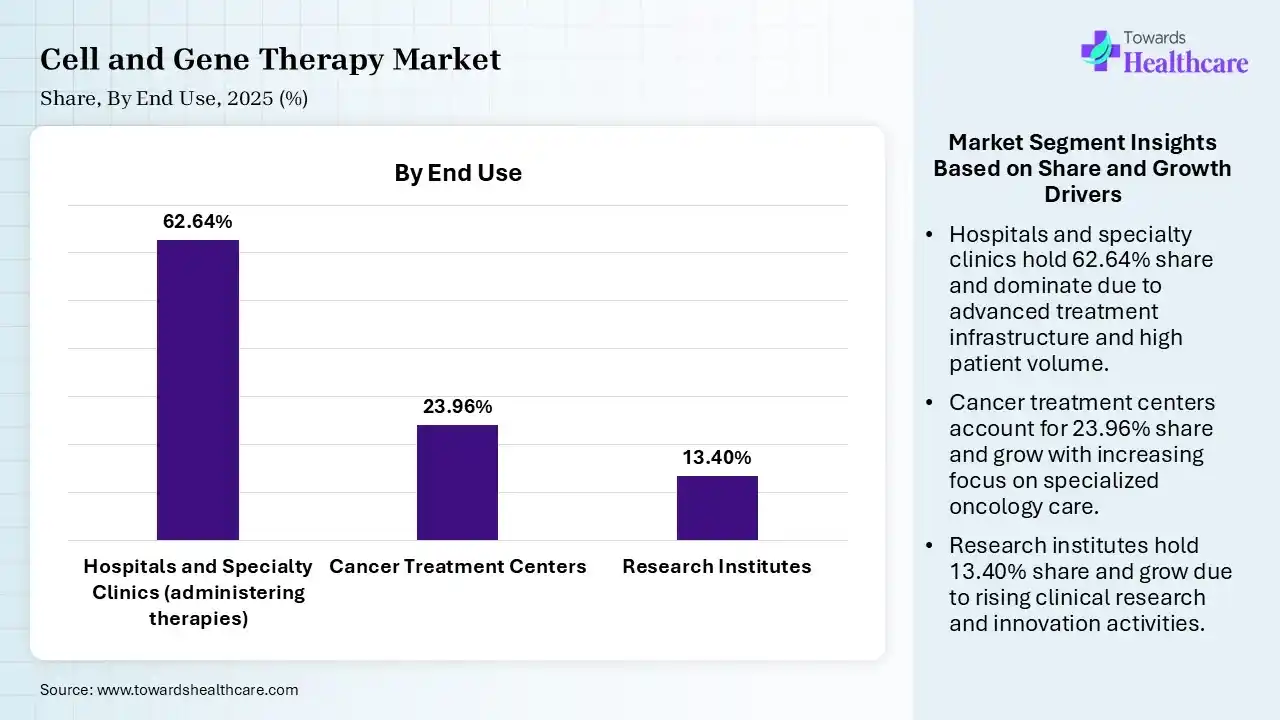
| Segment | Share 2025 (%) |
| Hospitals and Specialty Clinics (administering therapies) | 62.64% |
| Cancer Treatment Centers | 23.96% |
| Research Institutes | 13.40% |
Which End-Use Dominated the Market in 2025?
The hospitals and specialty clinics segment led the cell and gene therapy market with 62.64% share in 2025 due to the need for specialized infrastructure, stringent regulatory compliance, and multidisciplinary care required to administer these complex treatments. They act as the primary hubs for clinical trials, patient administration, and high-cost treatment reimbursement.
The cancer treatment centers segment held 23.96% share of the market in 2025 and is expected to grow at the fastest CAGR during the forecast period, driven by the curative potential of CAR-T cell therapies for relapsed cancers, rising cancer prevalence, and high demand for personalized medicine. The growth is accelerated by FDA approvals, technological advancements in CRISPR gene editing, and the move toward community-based treatment delivery.
The research institutes captured 13.40% share in 2025. Research institutes and academic centers play a significant role in the cell and gene therapy market, particularly within the clinical trials and early-stage research segments, often driving innovation alongside biotech companies.
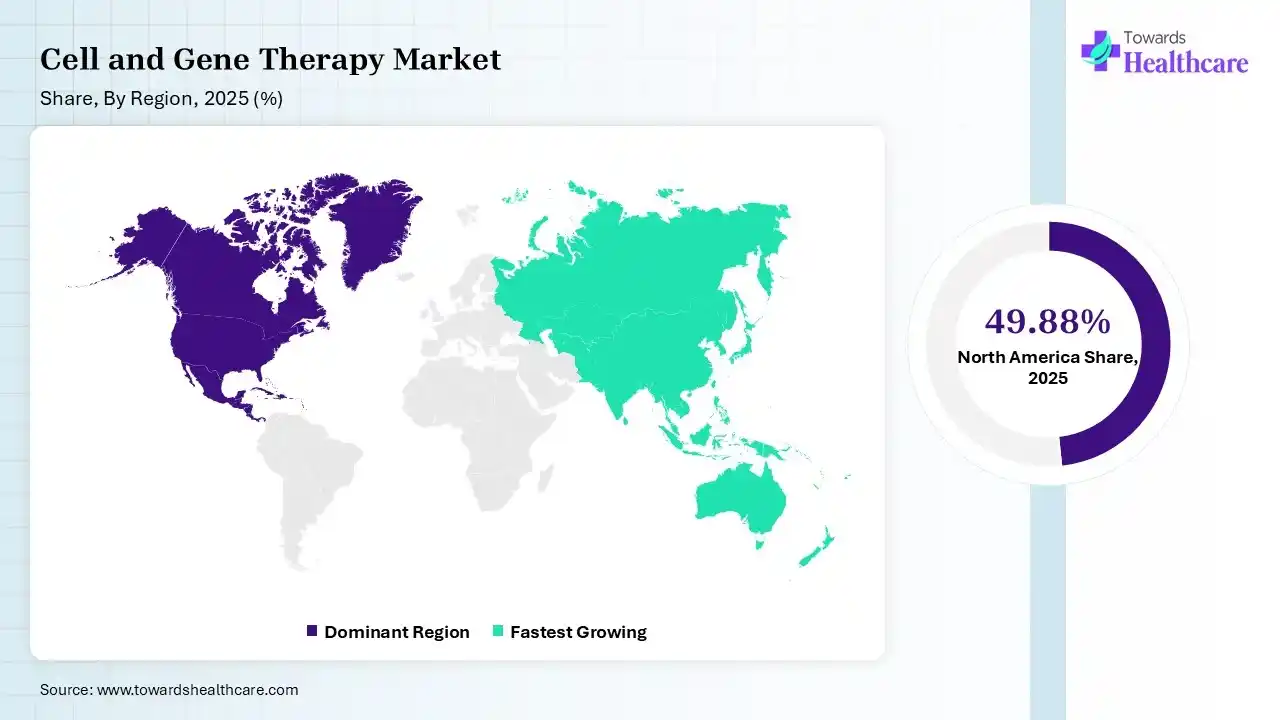
North America dominated the market with 49.88% share in 2025. The North American cell and gene therapy market was valued at US$ 4.46 billion in 2025, increased to US$ 5.17 billion in 2026, and is projected to reach approximately US$ 21.7 billion by 2035, growing at a CAGR of 17.1% from 2026 to 2035.
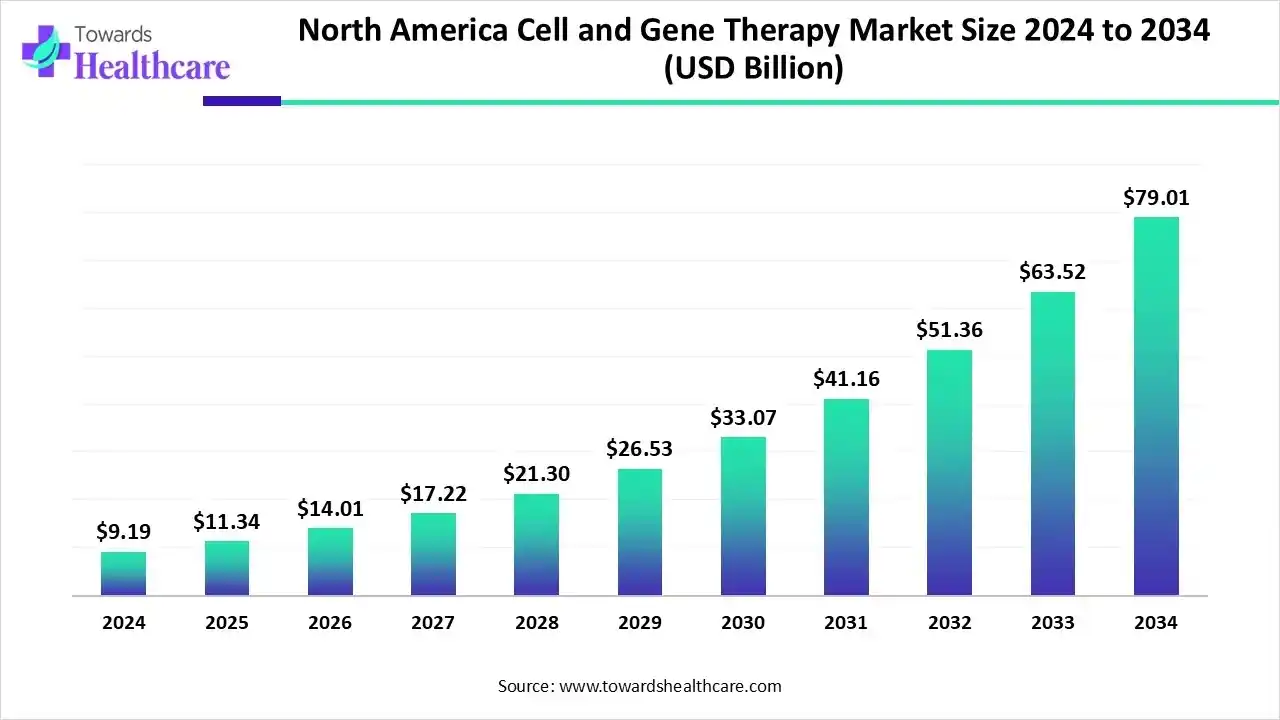
The North America region holds a prominent position in the global cell and gene therapy market. With its advanced healthcare infrastructure, strong regulatory framework, and significant investments in research and development, North America has become a hub for innovation and commercialization in this field.
The cell and gene therapy market in North America is experiencing significant growth due to the high prevalence of chronic diseases such as cancer, cardiovascular diseases, and genetic disorders. These conditions have created a demand for personalized and effective treatment options. Cell and gene therapies have emerged as promising solutions, addressing the root causes of these diseases and offering new possibilities for treatment and management.
The presence of leading pharmaceutical and biotechnology companies in North America further strengthens the market. These companies have been at the forefront of research and development in cell and gene therapies, investing heavily in clinical trials and infrastructure. Their expertise, combined with collaborations with academic institutions and research organizations, has accelerated the translation of scientific discoveries into commercial products. The regulatory landscape in North America has also been supportive of the cell and gene therapy market. The U.S. Food and Drug Administration (FDA) has established expedited regulatory pathways, such as the Regenerative Medicine Advanced Therapy (RMAT) designation and the Fast Track designation, to facilitate the development and approval of promising therapies.
The United States, in particular, dominates the cell and gene therapy market in North America. The country has a well-established biotechnology sector, a favorable regulatory environment, and a large patient population, making it an attractive market for cell and gene therapy companies. Several key players in the field are headquartered in the United States, driving innovation and market expansion. In January 2024, the Biden-Harris Administration announced that sickle cell disease would be the primary focus of the Cell and Gene Therapy (CGT) Access Model, owing to more than 100,000 cases in America. The main goal is to increase access to high-quality, affordable healthcare and lower healthcare costs.
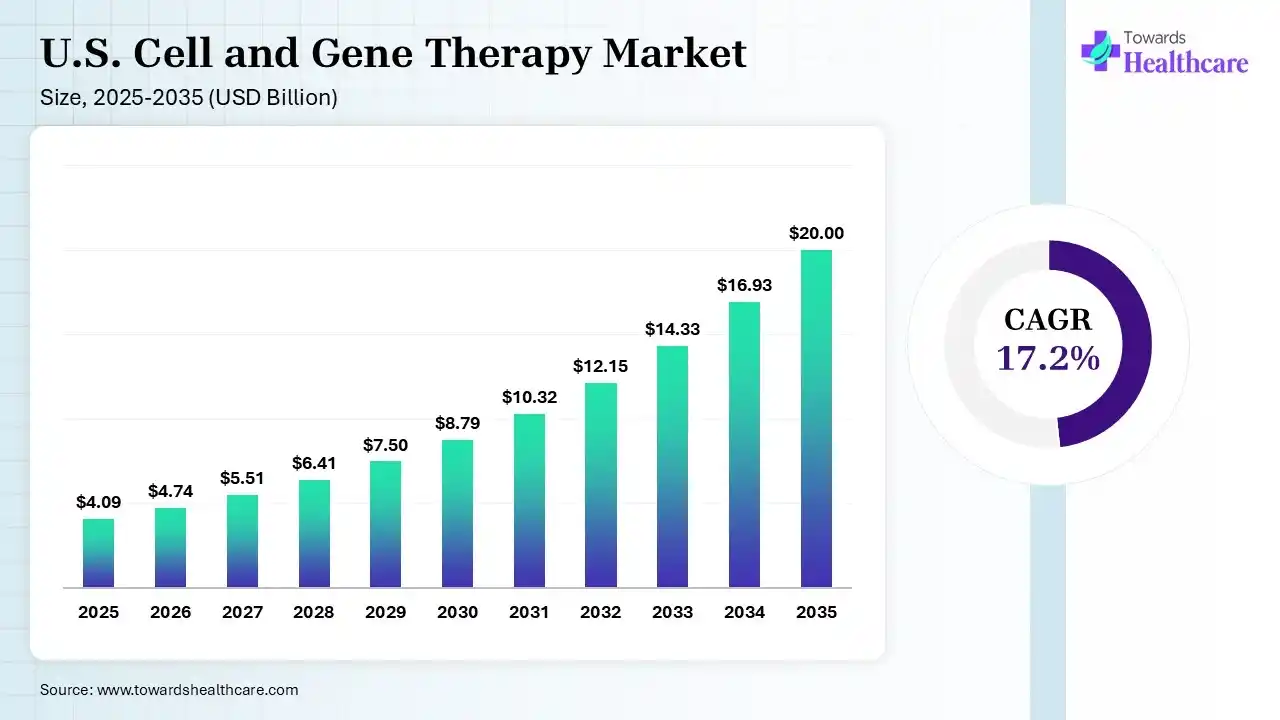
The U.S. cell and gene therapy market size was estimated at USD 4.09 billion in 2025 and is predicted to increase from USD 4.74 billion in 2026 to approximately USD 20 billion by 2035, expanding at a CAGR of 17.2% from 2026 to 2035.
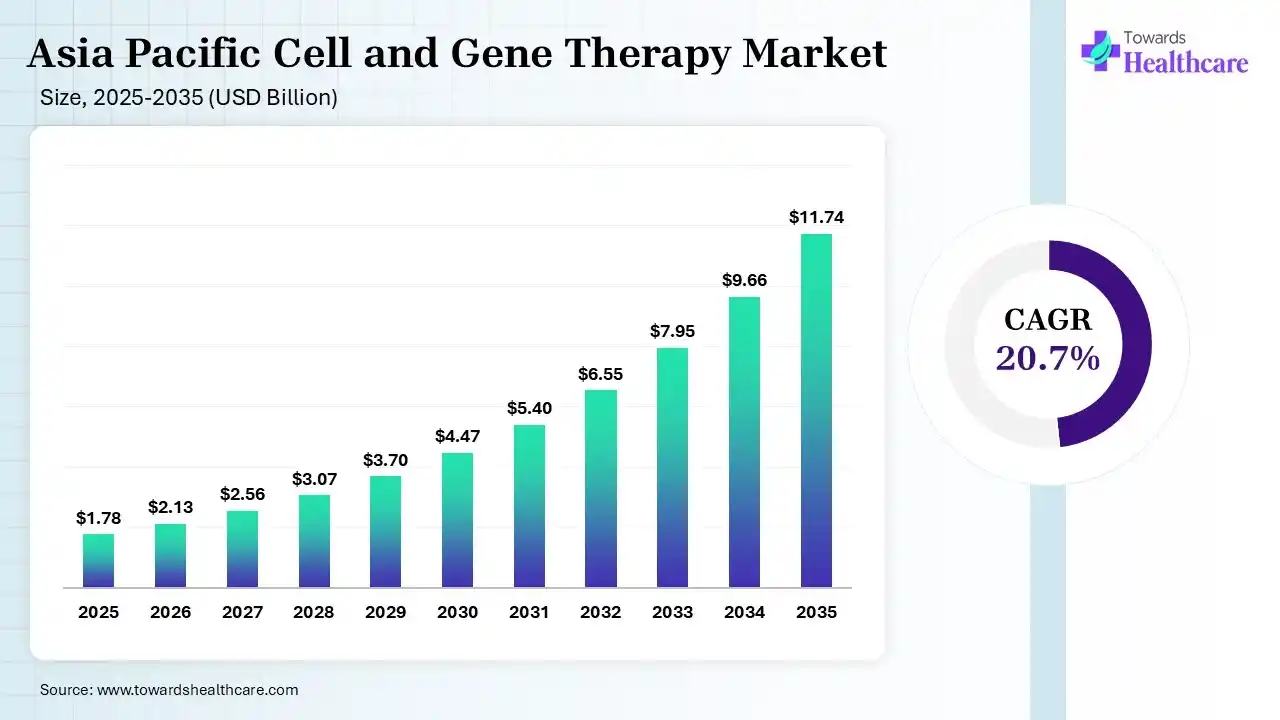
Asia Pacific held 19.94% of the total market share and is expected to grow at the fastest CAGR during the forecast period. The APAC cell and gene therapy market is valued at US$ 1.78 billion in 2025, increasing to US$ 2.13 billion in 2026, and is expected to reach approximately US$ 11.74 billion by 2035. This growth reflects a CAGR of 20.7% during the forecast period from 2026 to 2035.
The Asia Pacific region is emerging as a promising market for cell and gene therapy. With its large and diverse population, increasing prevalence of chronic diseases, and growing investments in healthcare infrastructure, the region offers significant opportunities for the development and commercialization of cell and gene therapies.
Several countries in the Asia Pacific region have shown strong commitment to advancing cell and gene therapies through supportive regulatory frameworks and government initiatives. For example, Japan has implemented the Act on the Safety of Regenerative Medicine, which aims to accelerate the approval process for regenerative medicine products. China has also made significant investments in the field, launching the Made in China 2025 initiative to promote the development of innovative therapies.
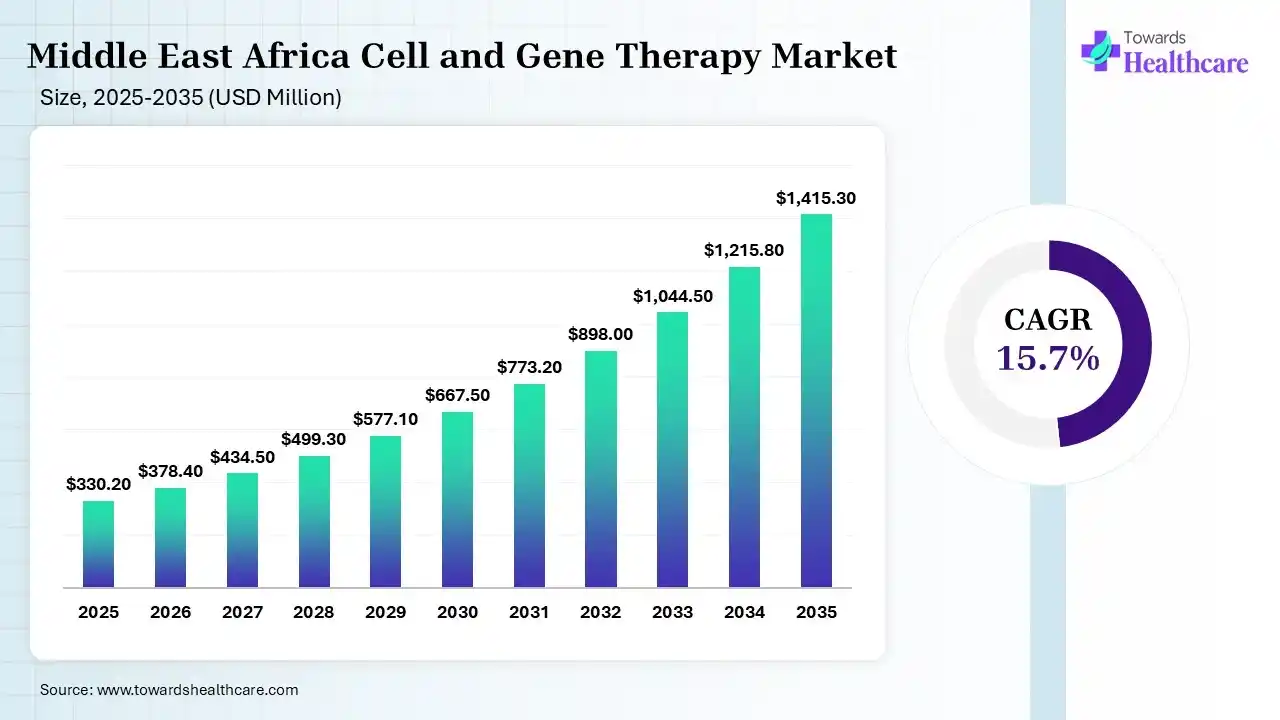
MEA captured 3.69% revenue of the market in 2025. The Middle East and Africa cell and gene therapy market is estimated at USD 330.2 million in 2025 and grew to USD 378.4 billion in 2026. It is expected to reach approximately USD 1415.3 million by 2035, expanding at a CAGR of 15.7% from 2026 to 2035.
The UAE is home to numerous foreign companies, such as Charles River Laboratories and SGS SA, that manufacture CGT products to meet local demand. The UAE government also makes constant efforts to support CGT development and delivery to patients. In August 2024, the Department of Health Abu Dhabi provided gene transfer therapy for Duchenne muscular dystrophy for the first time in the emirate.
Latin America captured 1.59% revenue of the market in 2025. The Latin American cell and gene therapy market was valued at US$ 142.4 million in 2025 and is expected to reach US$ 160.7 million in 2026, with projections indicating it could approach nearly US$ 495.4 million by 2035, growing at a CAGR of 13.3% over the forecast period.
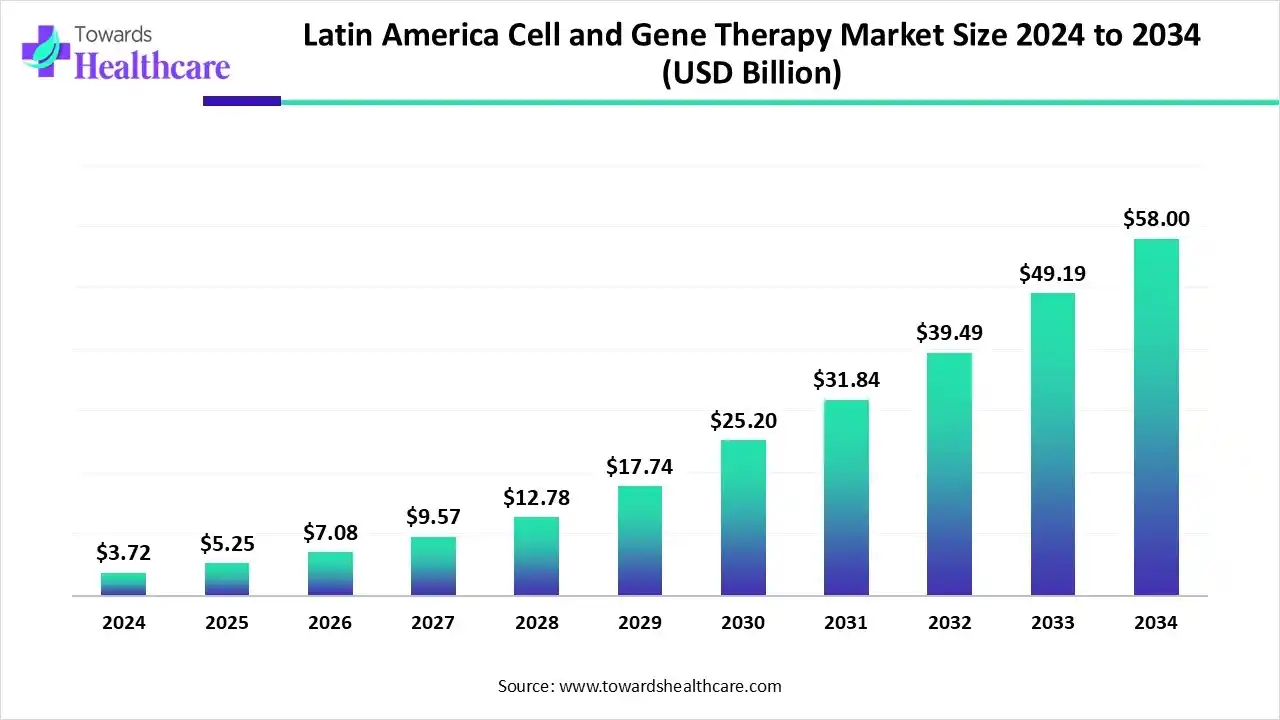
The Latin American cell and gene therapy market is experiencing strong growth, fueled by the rising prevalence of chronic and genetic diseases such as cancer, rare disorders, and immune conditions. Advances in biotechnology, gene-editing, and regenerative medicine are driving innovative therapies, while increased clinical trials in Brazil and Mexico, along with government support, investments, and strategic collaborations, are creating a favorable landscape. Growing awareness and adoption of personalized medicine are further boosting market potential across the region.
It is estimated that there are 186 biotech companies in Brazil. Brazilians are becoming more aware of CGT products and their benefits, necessitating their indigenous development. This demand is driven by the rising prevalence of chronic and genetic disorders. Approximately 13 million individuals are affected by a rare disease in Brazil. Also, the number of medical geneticists increased by 103% in a decade.
Asia Pacific held 19.94% of the total market share and is expected to grow at the fastest CAGR during the forecast period. The Europe cell and gene therapy market size is estimated at US$ 2.23 billion in 2025, is projected to grow to US$ 2.6 billion in 2026, and is expected to reach around US$ 11.83 billion by 2035. The market is projected to expand at a CAGR of 18.2% between 2026 and 2035.
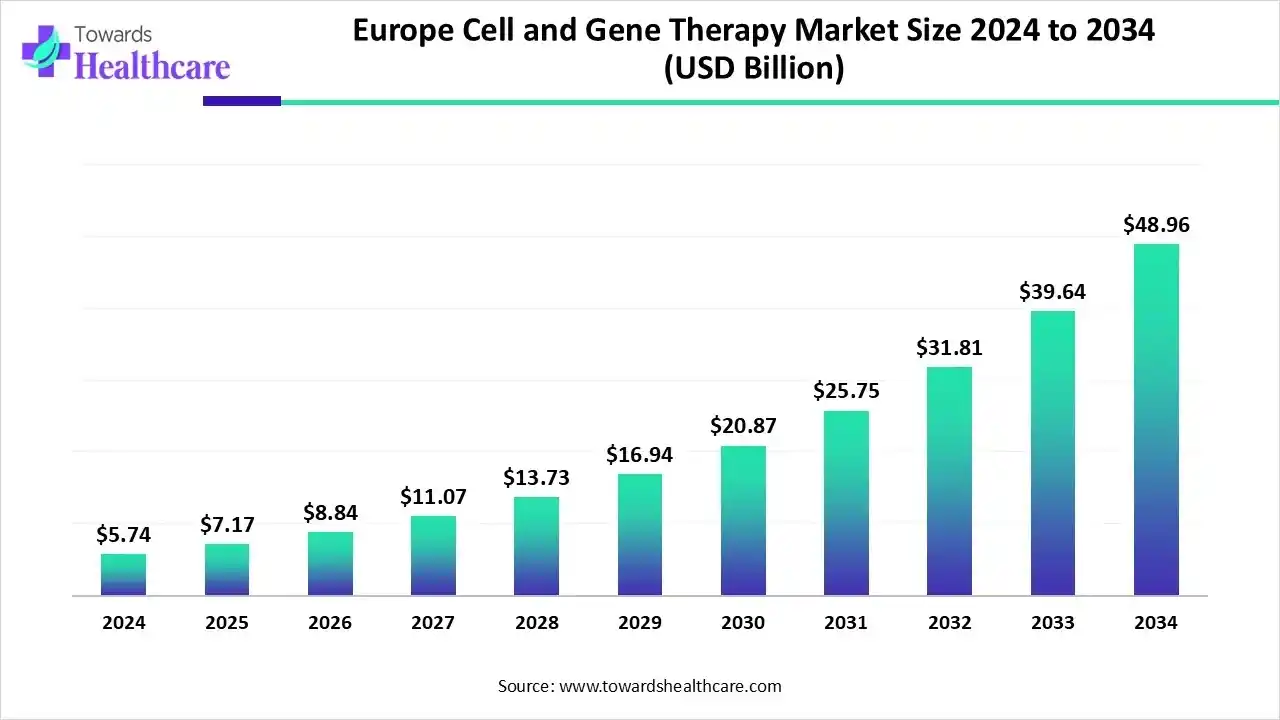
The Europe cell and gene therapy market is expanding due to increased government spending, supportive regulations, and rising demand for advanced therapies, with key players including biotech firms, pharmaceutical companies, and academic institutions. The growth is further fueled by the popularity of CAR-T cell therapies, backed by EMA approvals and funding initiatives like Horizon Europe.
The rapidly expanding research and clinical trial infrastructure propels market growth in the UK. As of 9th December 2025, around 56 clinical trials were registered in the UK related to clinicaltrials.gov website. The UK government has made several investments in the CGT field, including $10 million grant to the NHS Blood and Transplant and £17.9 million to the Advanced Therapy Treatment Center Network (ATCC Network).
Leonard Schleifer, CEO of Regeneron, in a recent interview, suggested that the big thing for the biotech sector is gene therapy or genetics. He said that associating genes with disease is going to drive the pharmaceutical industry by emphasizing innovative treatments to repair certain genes and silence others.

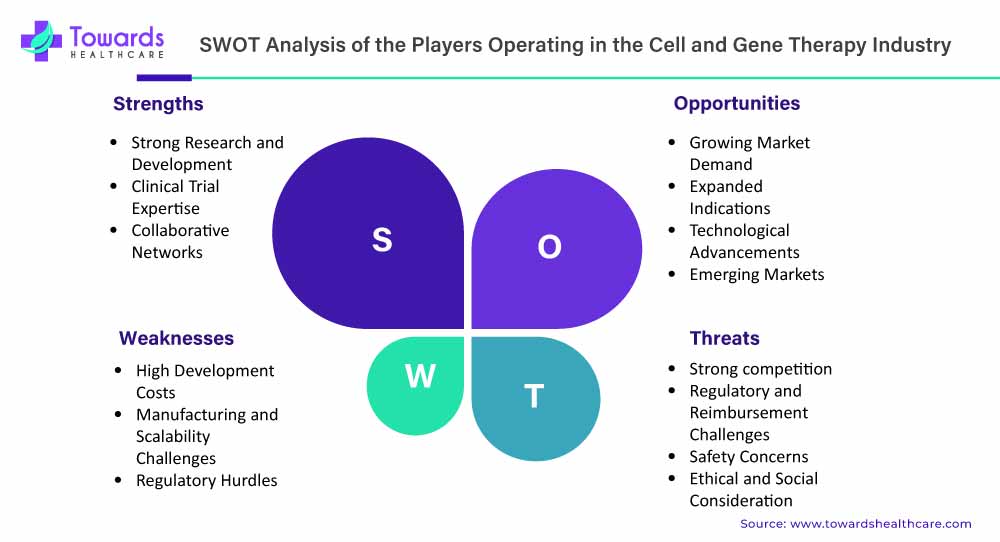
The cell and gene therapy market has experienced remarkable growth and progress in recent years, holding immense potential for transforming healthcare with personalized and targeted therapies. Several key players dominate the competitive landscape in this field. To gain a larger share of the market, players are using strategies like investments, alliances, acquisitions, and mergers.
By Therapy Type
By Indication / Therapeutic Area
By Vector Type (Gene Delivery Method)
By Manufacturing Type
By End Use
By Region
April 2026
April 2026
April 2026
April 2026