March 2026

The global contract pharmaceutical manufacturing market size was estimated at USD 232.32 billion in 2025 and is predicted to increase from USD 255.41 billion in 2026 to approximately USD 599.3 billion by 2035, expanding at a CAGR of 9.94% from 2026 to 2035.
The growing demand for biologics and specialty drugs is increasing the use of contract pharmaceutical manufacturing services. Additionally, growing innovations, generic product demand, technological advancement, new platform launches, and collaborations are also driving their use, enhancing the market growth.
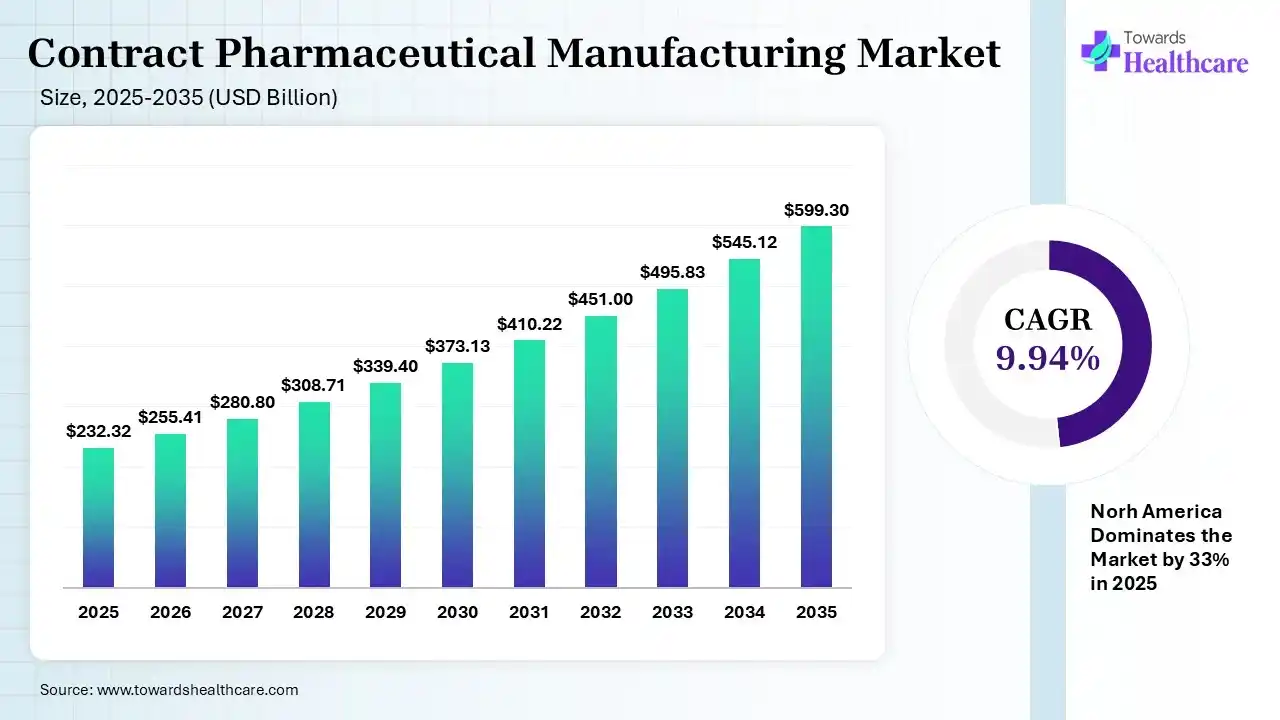
The contract pharmaceutical manufacturing market is driven by the need for affordable services, shorter drug development timelines, and specialized expertise. Contract pharmaceutical manufacturing refers to the business agreement between the pharmaceutical companies and third-party organizations that help in the manufacturing of various pharmaceutical products. The companies outsource the drug products where these organization offers production, formulation, quality control, and packaging services.
The use of AI in contract pharmaceutical manufacturing is increasing, as it helps in the optimization of drug formulation by offering quality control and faster defect detection. It also offers process automation, which reduces manual errors and enhances batch consistency, documentation, and efficiency, which promotes regulatory compliance. Its predictive analysis reduces the chances of equipment failure and accelerates R&D activities, as well as supports clinical trials.
Blooming Advanced Therapies
The growing advancement in the biologics, personalized medicines, and biosimilars is increasing the demand for contract pharmaceutical manufacturing services, where they are also being utilized for the development of vaccines and targeted therapies.
Expanding Outsourcing Trends
The growth in the demand for advanced therapies, biologics, and other sterile products is driving the demand for contract pharmaceutical manufacturing services, leading to a rise in the outsourcing trends to accelerate their R&D, innovations, and commercialization.
Technological Advancements
The growing technological innovations are driving the adoption of new technologies to offer continuous manufacturing, as well as to enhance the manufacturing efficiency, speed, quality, and affordability, which is grabbing the attention of the pharmaceutical companies.
| Table | Scope |
| Market Size in 2026 | USD 255.41 Billion |
| Projected Market Size in 2035 | USD 599.3 Billion |
| CAGR (2026 - 2035) | 9.94% |
| Leading Region | North America by 40% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Service Type, By Product/Service, By Drug Type, By Dosage Form, By Workflow, By End User, By Region |
| Top Key Players | Lonza Group, Thermo Fisher Scientific (Patheon), Catalent, Inc., Samsung Biologics, WuXi AppTec, Recipharm AB, , Piramal Pharma Solutions, Akums Drugs & Pharmaceuticals, Siegfried Holding AG |
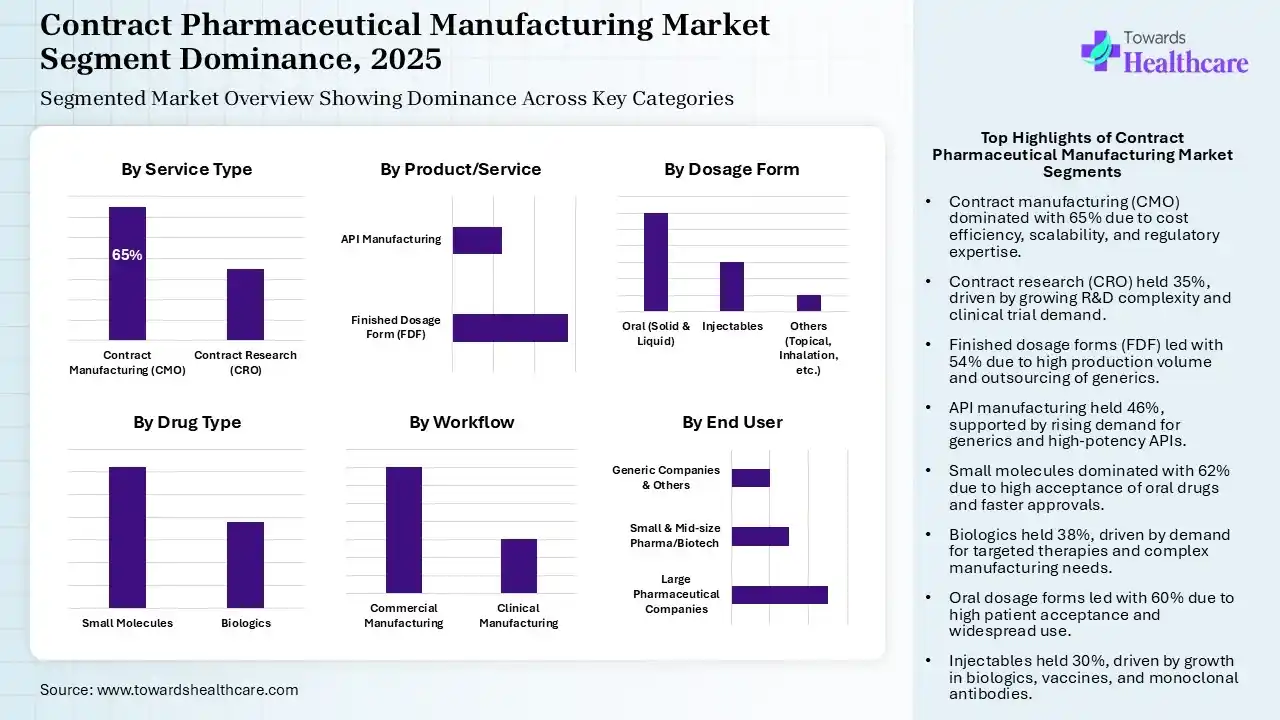
The Contract Manufacturing (CMO) Segment Dominated the Market in 2025
| Segment | Share 2025 (%) |
| Contract Manufacturing (CMO) | 65% |
| Contract Research (CRO) | 35% |
Explanation
The contract manufacturing (CMO) segment held the largest share of approximately 65% of the contract pharmaceutical manufacturing market in 2025, due to its affordable services and enhanced scalability. They focused on the R&D and innovation, which also attracted the consumers. The presence of specialized expertise and regulatory support also increased their use.
The contract research (CRO) segment held the second-largest share of approximately 35% of the market in 2025, driven by growing R&D complexities and rising cost pressure. Additionally, expanding clinical pipelines and clinical trials are also leading to their collaborations. Their expertise and advanced technologies are also attracting companies.
The Finished Dosage Form (FDF) Segment Dominated the Market in 2025
| Segment | Share 2025 (%) |
| Finished Dosage Form (FDF) | 54% |
| API Manufacturing | 46% |
Explanation
The finished dosage form (FDF) segment held the dominating share of approximately 54% of the contract pharmaceutical manufacturing market in 2025, due to growth in its production volume and formulation complexities. The rise in the demand for regulatory experts and faster product launches also increased the use of contract pharmaceutical manufacturing facilities. Additionally, growth in the outsourcing of generic products also increased their use.
The API manufacturing segment held the second-largest share of approximately 46% of the market in 2025, due to a surge in the generic pharmaceuticals. The growing interest in the development of new high-potency APIs is also increasing the use of contract pharmaceutical manufacturing services. The growing government support and outsourcing trends are also increasing their demand.
The Small Molecules Segment Dominated the Market in 2025
| Segment | Share 2025 (%) |
| Small Molecules | 62% |
| Biologics | 38% |
Explanation
The small molecules segment held the largest revenue share of approximately 62% of the contract pharmaceutical manufacturing market in 2025, driven by the presence of oral formulations, which increase their acceptance rates, leading to a rise in their production volume. The growth in the demand for their generic version also propelled the reliance on the contract pharmaceutical manufacturing services. Their faster approval also increased the use of these services, which contributed to their innovations.
The biologics segment held the second-largest share of approximately 38% of the market in 2025, due to growing demand for targeted therapies, increasing chronic disease burden, and complex manufacturing process. The rise in the biologics pipeline and their faster approvals are also driving their innovations. Thus, all these factors are increasing the use of contract pharmaceutical manufacturing facilities.
The Oral Segment Dominated the Market in 2025
| Segment | Share 2025 (%) |
| Oral (Solid & Liquid) | 60% |
| Injectables | 30% |
| Others (Topical, Inhalation, etc.) | 10% |
Explanation
The oral segment held the major revenue share of approximately 60% of the contract pharmaceutical manufacturing market in 2025, driven by high patient acceptance rates and growth in its product volume. Their widespread applications and high demand for generic products also increased the use of contract pharmaceutical manufacturing for its affordable services. Additionally, their advanced technologies and standard processes also led to new collaborations.
The injectables segment held the second-largest share of approximately 30% of the market in 2025, due to growth in the biologics. Their enhanced efficacy, bioavailability, and rapid action also increase their acceptance rates. The growing advancements in vaccines and monoclonal antibodies are also increasing their use, which in turn is promoting the use of contract pharmaceutical manufacturing facilities.
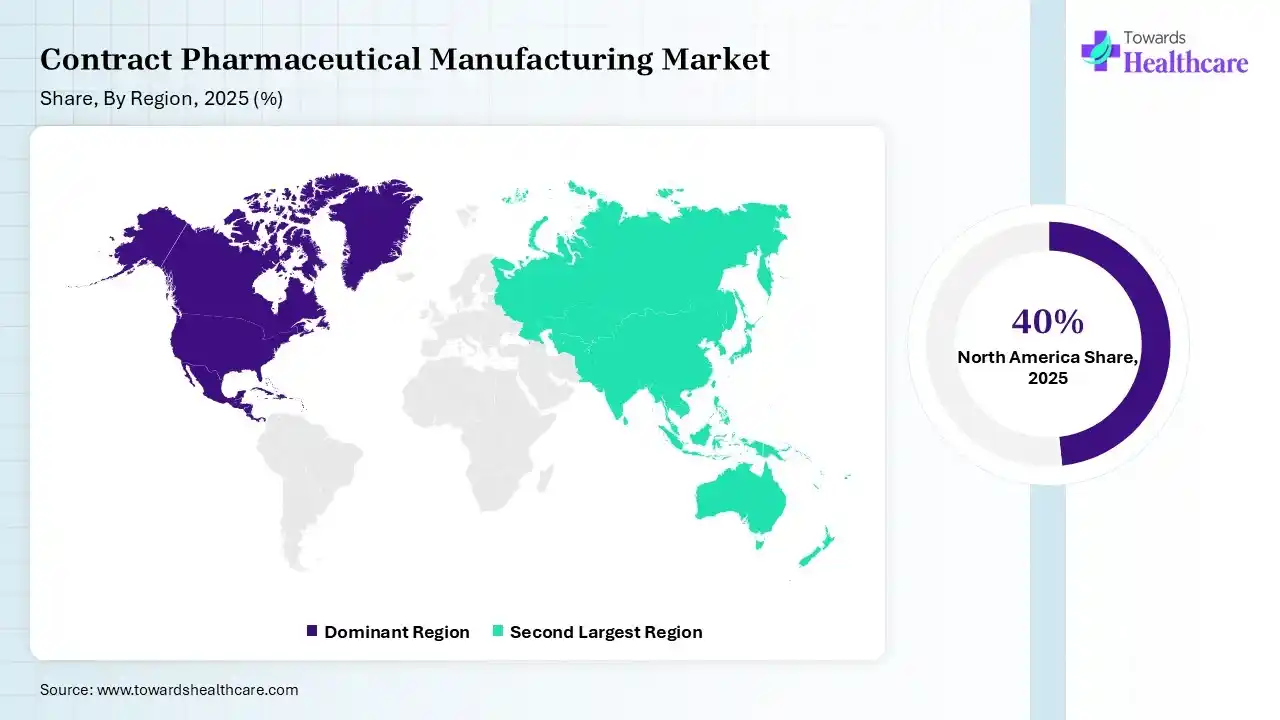
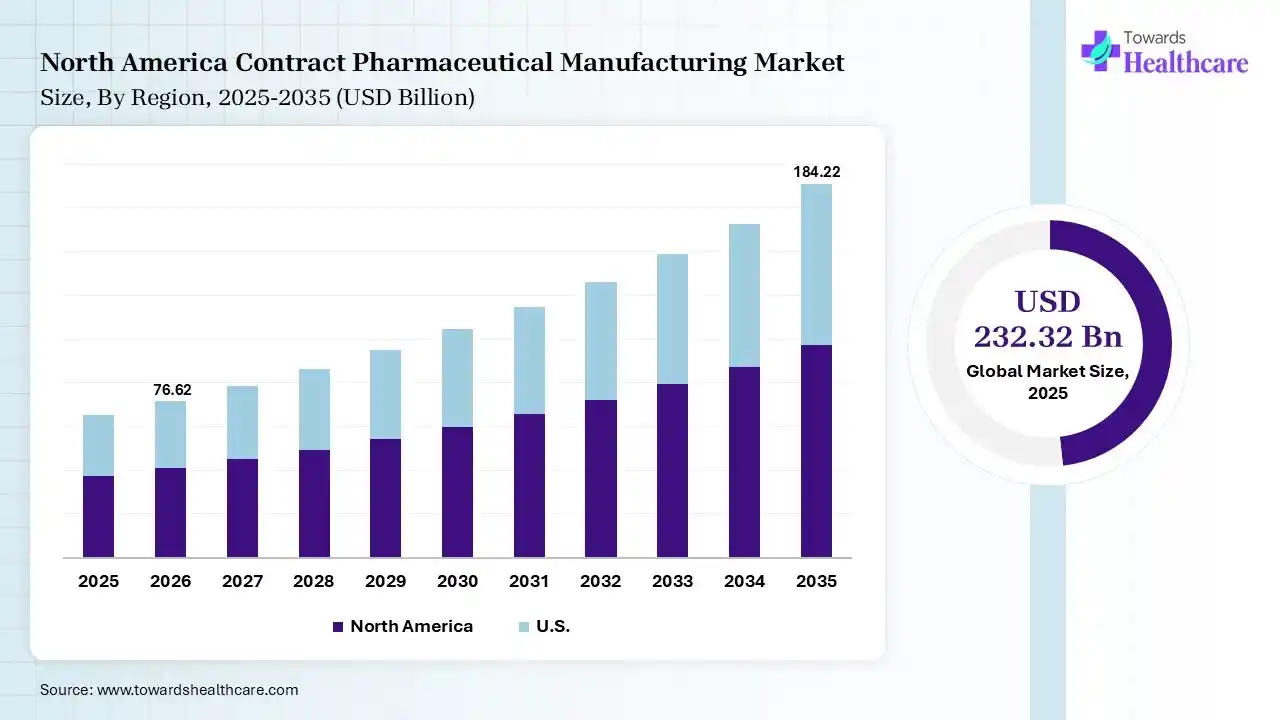
North America dominated the contract pharmaceutical manufacturing market with approximate 40% in 2025, due to the rise of robust pharmaceutical industries, which increased the outsourcing trends driving the adoption of contract pharmaceutical manufacturing services. The growth in the R&D activities and investments also increased their use, where the technological advancement also contributed to their increased use and market growth.
U.S. Market Trends
The presence of top pharma and biotech companies in the U.S. is increasing the use of CMO and CDMO services due to growing outsourcing trends. The growing R&D investments and increasing advancement in APIs, sterile products, and biologics are also increasing their demand. Additionally, advanced technologies and regulatory expertise are also attracting the companies.
Asia Pacific held the second-largest share of approximately 30% of the contract pharmaceutical manufacturing market in 2025 and is expected to grow with a significant CAGR of 9.8% during 2026-2035 due to the presence of a large manufacturing base, where the growing demand for generics and biologics is also increasing the collaboration with CMOs & CDMOs. The growing diseases and government support are also increasing the use of contract pharmaceutical manufacturing services, enhancing the market growth.
India Market Trends
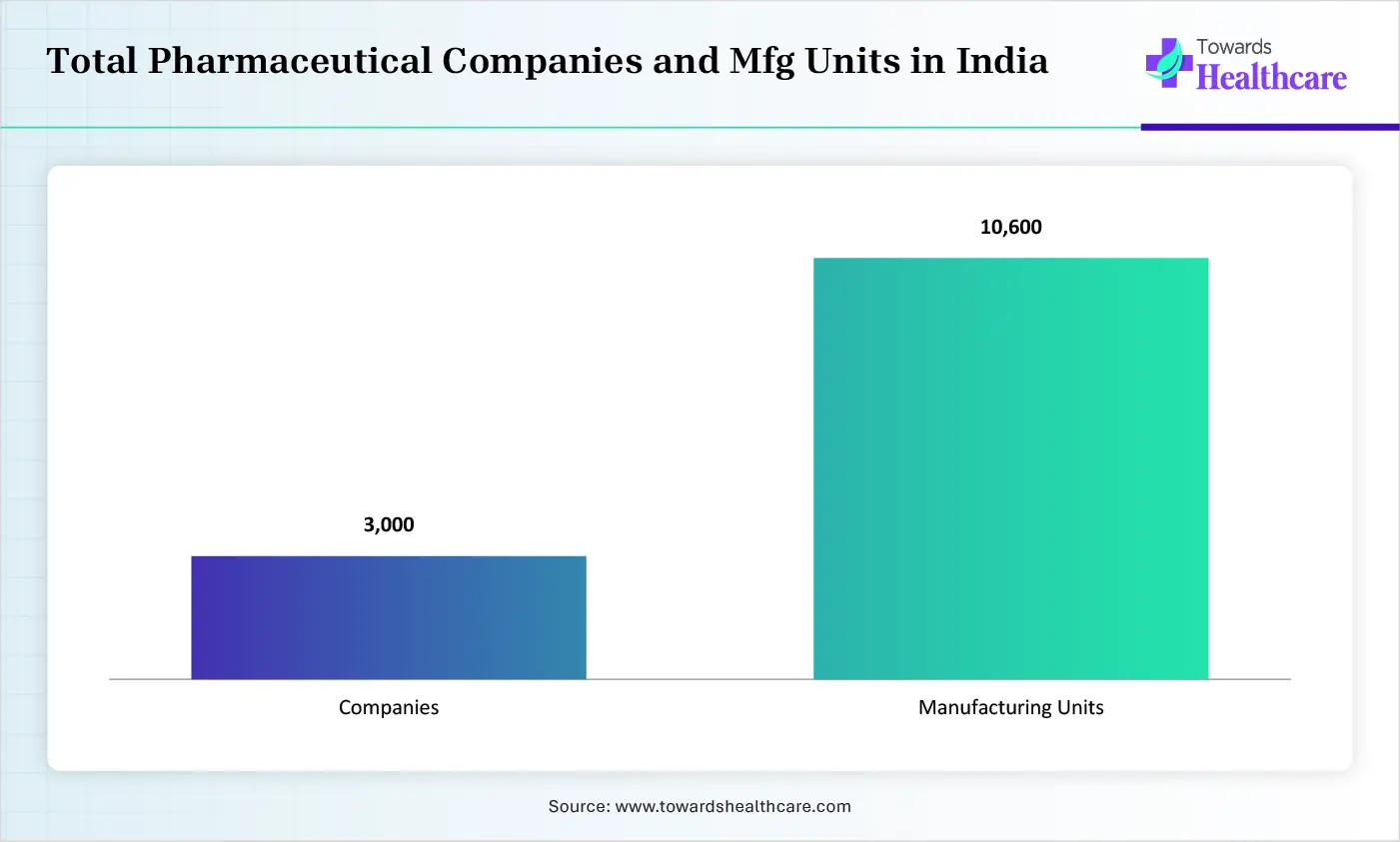
The growing adoption of generic products and biosimilars in India is increasing the collaboration with the contract pharmaceutical manufacturing facilities. The presence of a large manufacturing base and its affordable services are also increasing its use. The growing government initiatives and increasing outsourcing trends are also increasing their partnerships.
Europe is expected to grow significantly in the contract pharmaceutical manufacturing market due to a robust regulatory framework, which increases the demand for high-quality manufacturing, driving collaborations with the contract pharmaceutical manufacturing facilities. The presence of advanced pharmaceutical companies and growing demand for biologics are also increasing their use, promoting the market growth.
UK Market Trends
The UK consists of robust pharmaceutical and biotechnology industries, which are increasing the use of contract pharmaceutical manufacturing services due to a rise in the development of small molecules and biologics. The advanced manufacturing infrastructure, skilled personnel, and regulatory expertise are also attracting the companies.
R&D
Clinical Trials and Regulatory Approvals
Formulation and Final Dosage Preparation
Packaging and Serialization
Distribution to Hospitals, Pharmacies
Patient Support and Services

| Companies | Headquarters | Products Under Contract Pharmaceutical Manufacturing Services |
| Lonza Group | Basel, Switzerland | Biologics, small molecule API, antibody-drug conjugates, and cell and gene therapies |
| Thermo Fisher Scientific (Patheon) | Waltham, U.S. | Sterile injectable, biologics, and oral solid dosage forms |
| Catalent, Inc. | Somerset, U.S. | Advanced drug delivery, gene therapy, oral solids, and biologics |
| Samsung Biologics | Incheon, South Korea | Monoclonal antibodies, ADCs, mRNA vaccines |
| WuXi AppTec | Shanghai, China | Small molecules, peptides, cell and gene therapies, and oligonucleotides |
| Recipharm AB | Stockholm, Sweden | Sterile injectables, advanced therapeutic medicinal products, and oral solids |
| Boehringer Ingelheim (BioXcellence) | Ingelheim, Germany | Monoclonal antibodies, recombinant proteins, plasmid DNA, and fusion proteins. |
| Piramal Pharma Solutions | Mumbai, India | ADCs, potent APIs, sterile injectables, and complex oral solids |
| Akums Drugs & Pharmaceuticals | Haridwar, India | Tablets, capsules, injectables, and syrups |
| Siegfried Holding AG | Zofingen, Switzerland | Tablet, capsules, sterile vials, and ointments |
Strengths
Weaknesses
Opportunities
Threats
By Service Type
By Product/Service
By Drug Type
By Dosage Form
By Workflow
By End User
By Region
March 2026
March 2026
March 2026
March 2026
