Learn About the Diabetes Drugs Market Companies with SWOT Analysis
- AstraZeneca
- Bayers AG
- Boehringer Ingelheim International GmbH
- Dr. Reddy’s Laboratories
- Eli Lilly and Co.
- Johnson & Johnson
- Merck & Co.
- Novartis AG
- Novo Nordisk
- Sanofi
- Sun Pharmaceuticals
- Takeda Pharmaceuticals
Market Growth
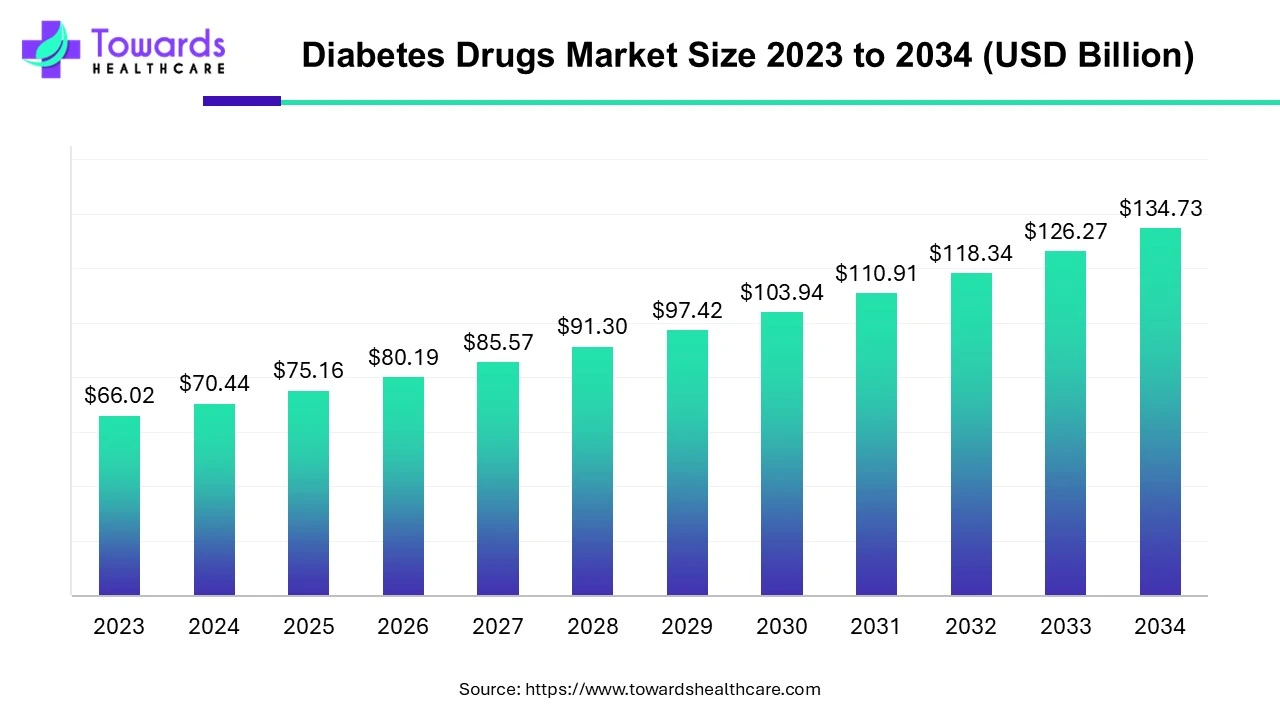
The global diabetes drugs market size is calculated at USD 75.16 billion in 2025 and is expected to be worth USD 134.73 billion by 2034, expanding at a CAGR of 6.7% from 2024 to 2034, as a result of the rising prevalence of diabetes, and rising patient support.
Diabetes Drugs Market Trends
- In July 2024, Novo Nordisk A/S was issued a Complete Response Letter (CRL) by the U.S. FDA regarding its Biologics License Application for icodec, a once-weekly basal insulin intended for diabetes treatment.
- In March 2024, Eli Lilly and Company teamed up with Amazon Pharmacy to distribute its GLP-1 drugs via Amazon and Truepill. This collaboration helped expand the global reach of its injectable GLP-1 medications, Mounjaro and Zepbound.
- In January 2024, Glenmark Pharmaceuticals Ltd. launched Lirafit, a biosimilar version of the anti-diabetic medication Liraglutide, in the Indian market. The cost for a typical 1.2 mg daily dose is approximately USD 1.21.
Competitive Landscape
The diabetes drugs market is highly competitive, with several key players competing for market share. These companies focus on developing innovative drugs and treatments to meet the evolving needs of patients with diabetes. Several key players dominate the competitive landscape in this field. To gain a larger share of the market, players are using strategies like investments, alliances, acquisitions, and mergers.
SWOT Analysis of Diabetes Drugs Market
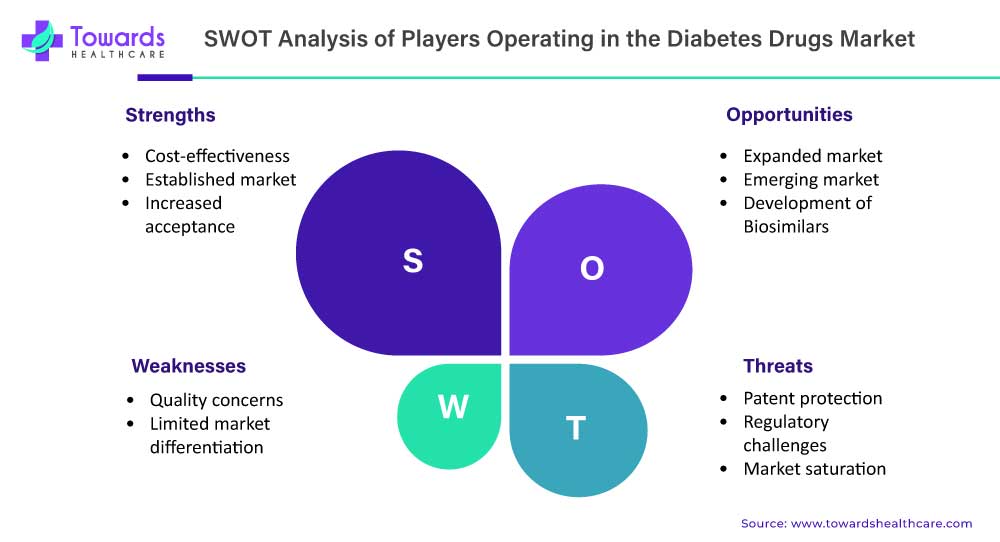
Strengths:
- Generic drugs are generally more affordable compared to their branded counterparts, making them accessible to a larger population.
- The generic drugs market has a well-established presence and is recognized as an essential component of healthcare systems worldwide.
- There is a growing acceptance and trust in generic drugs among patients, healthcare professionals, and regulatory authorities.
Weaknesses:
- Some stakeholders may have concerns about the quality and efficacy of generic drugs, although they undergo rigorous regulatory scrutiny and quality control.
- Generic drugs often have similar formulations and therapeutic effects, making it challenging for manufacturers to differentiate their products.
Opportunities:
- The demand for generic drugs is expected to increase due to factors such as the rising prevalence of chronic diseases, the aging population, and cost-containment efforts in healthcare.
- There is a significant opportunity for growth in emerging markets, where access to affordable healthcare is a major concern.
- The development and commercialization of biosimilars, which are generic versions of biologic drugs, present new opportunities for market expansion.
Threats:
- The expiration of patents on branded drugs may lead to increased competition from generic manufacturers, posing a threat to market share and profitability.
- Compliance with regulatory requirements and the need for extensive clinical trials can be time-consuming and costly for generic drug manufacturers.
- The generic drugs market may become saturated in some therapeutic areas, limiting growth potential.
Latest Announcements by Industry Leaders
Erez Israeli, CEO of Dr. Reddy’s Laboratories Ltd., announced that the company is planning to have 15 GLP-1 agonists used to treat obesity and diabetes in all the markets when they open, including the 2026 markets. The 15 products will be introduced in different timelines. Through this, the company is set to introduce semaglutide, a GLP-1 agonist, in India once the Novo Nordisk patent expires.
Recent Developments in the Diabetes Drugs Market
- In April 2024, Sanofi India Limited launched its new diabetes drug, Soliqua, for treatment in adults with obesity and type 2 diabetes mellitus after the CDSCO approval.
- In July 2024, Zydus Lifesciences announced the US FDA marketing approval for its new diabetes drug, Zituvimet XR (sitagliptin and metformin hydrochloride extended-release). The drug was approved for the treatment of type 2 diabetes mellitus in adults to improve glycemic control.
Contact our experts to explore the Diabetes Drugs Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
