Latest Updates of the Leadless Pacemakers Market Companies with Market Growth

Company Latest Updates
| Company | Headquarters | Latest Update |
| Abbott | United States | In October 2025, Abbott, the worldwide healthcare company, announced the launch of AVEIR™ dual chamber (DR) leadless pacemaker system, the first of its kind in the world. |
| Medtronic | Ireland | Micra AV2 and Micra VR2 offer an unmatched leadless pacing experience for bradycardia patients. With enhanced battery longevity and smarter algorithms. |
| EBR Systems, Inc. | United States | EBR Systems enrolled the first patients in the WiSE-UP research assessing its WiSE cardiac resynchronization therapy (CRT) technology. |
| Boston Scientific Corporation | United States | It focuses on a modular, inclusive strategy that combines leadless pacing with its established subcutaneous implantable cardioverter-defibrillator (S-ICD) expertise |
| MicroPort Scientific Corporation | China | In February 2025, MicroPort CRM, Andhra Pradesh MedTech Zone (AMTZ), and Purple MicroPort signed a Memorandum of Understanding (MoU) to drive the expansion of MicroPort® CRM’s pacemaker business in India and its neighboring countries |
Supply Chain Analysis
R&D:
- The Research and Development (R&D) processes for leadless pacemakers include miniaturization and electronics design, battery technology and longevity, fixation mechanisms, catheter-based delivery systems, retrieval and end-of-service (EOS) management, clinical trials, safety validation, and MRI compatibility.
- Key Players: EBR Systems and CAIRDAC
Manufacturing Processes:
- Manufacturing processes for leadless pacemakers (LCPs) include advanced micro-engineering to create a catheter-deliverable, self-contained, and biocompatible tool.
- Key Players: MicroPort Scientific
Patient Services:
- Leadless pacemaker patient services mainly focus on minimally invasive procedures, featuring rapid recovery times and remote monitoring abilities, often eliminating the requirement for surgical pocket care.
- Key Players: Boston Scientific and Biotronik
Market Growth
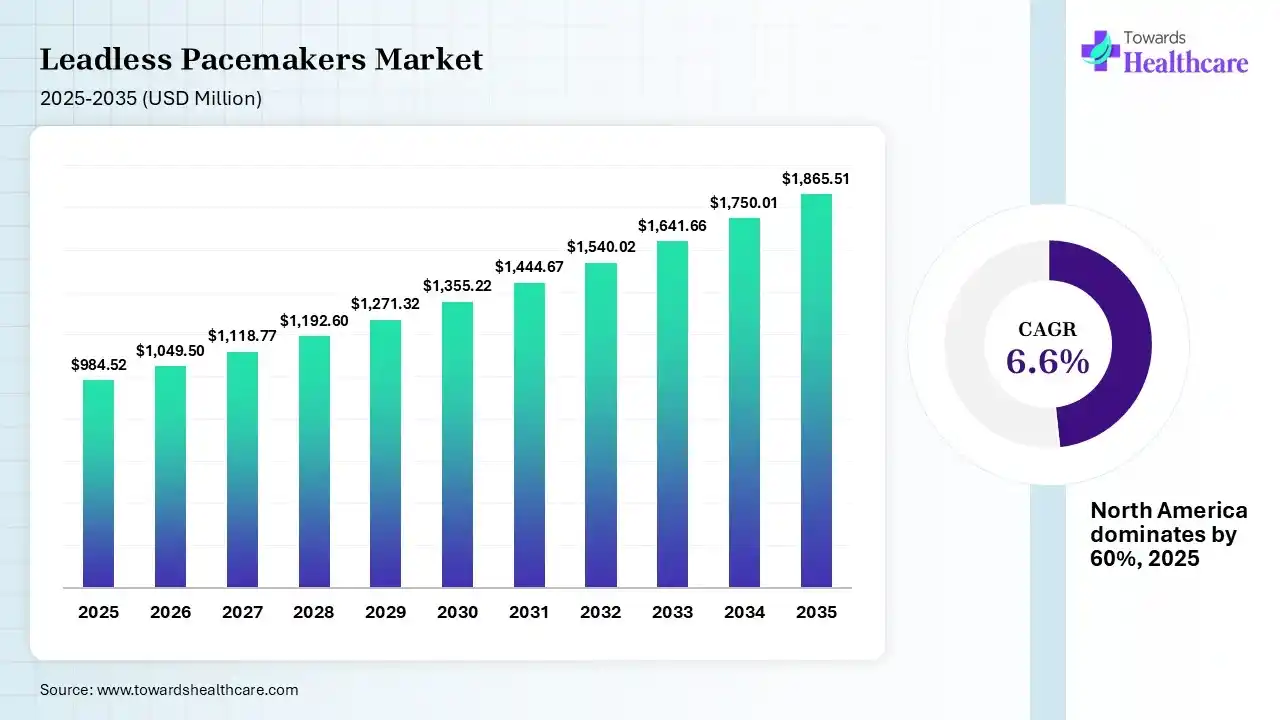
The global leadless pacemakers market size was estimated at USD 984.52 million in 2025 and is predicted to increase from USD 1049.5 million in 2026 to approximately USD 1865.51 million by 2035, expanding at a CAGR of 6.6% from 2026 to 2035.
Recent Developments in the Leadless Pacemakers Market
- In October 2025, Max Hospital, Saket, and Abbott announced the launch of India’s first dual-chamber leadless Aveir pacemaker, intending to deploy next-generation technologies in healthcare delivery. This new technology minimizes the risks associated with lead-related complications and optimizes comfort and long-term safety for the patient.
- In July 2025, Orchestra BioMed Holdings, Inc., a biomedical organization accelerating high-impact technologies to patients through risk-reward sharing partnerships, announced that the Organization has secured $70 million in novel capital from Ligand Pharmaceuticals Incorporated and Medtronic plc to advance its late-stage combined cardiology programs.
- In April 2025, EBR Systems, Inc. received FDA approval of the WiSE System, which marks a significant leap forward in the management of heart failure. Unlike conventional CRT, the WiSE Technology is the first and only to deliver leadless left ventricular endocardial pacing (LVEP), which thoroughly aligns with the heart’s natural transmission pathway.
Access a free Leadless Pacemakers Market sample with clear stats, segment analysis, and competitor data: Download Sample
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
