Top Companies in the U.S. Cell and Gene Therapy Market & Their Offerings

| Companies | Headquarters | Offerings |
| Alnylam Pharmaceuticals Inc. | Massachusetts, U.S. | Focuses on RNAi-based gene-silencing therapies for rare genetic and cardiometabolic diseases. |
| Amgen Inc. | California, U.S. | Offers biologics and oncology therapies with growing investments in cell and gene therapy pipelines. |
| Biogen Inc | Cambridge, Massachusetts, U.S. | Develops gene therapies and advanced treatments for neurological and rare diseases. |
| Dendreon Pharmaceuticals LLC. | California, U.S. | focuses on cell-based immunotherapies for cancer treatment. |
| Helixmith Co. Ltd. | Seoul, South Korea | Develops DNA-based gene therapies targeting chronic and rare diseases. |
What are the Recent Developments in the U.S. Cell and Gene Therapy Market?
- In April 2026, the U.S. Food and Drug Administration approved Otarmeni (lunsotogene parvec-cwha), marking the first-ever dual adeno-associated virus (AAV) vector-based gene therapy. This treatment is indicated for pediatric and adult patients with severe-to-profound sensorineural hearing loss associated with biallelic OTOF gene variants.
- In March 2026, Children’s Hospital Los Angeles is committed to providing top-tier access to cutting-edge cell and gene treatments for children facing serious or life-threatening illnesses. The facility recently grew its portfolio to 12 FDA-cleared therapies, making it the leading pediatric center on the West Coast for these advanced medical options.
Supply Chain Analysis
Clinical Trials
- The U.S. cell and gene therapy landscape is progressing quickly, with strong clinical focus on CAR-T treatments for cancer and viral vector-based therapies for rare genetic conditions. More than 2,600 investigational therapies are currently being regulated by the FDA’s Office of Therapeutic Products, with many targeting blood disorders and inherited diseases.
- Key players: Novartis AG, Gilead Sciences, Bristol Myers Squibb, and Bluebird Bio.
Regulatory Approvals
- In the U.S., cell and gene therapy approvals are regulated by the Center for Biologics Evaluation and Research under the U.S. Food and Drug Administration. Developers must submit a Biologics License Application to demonstrate safety, quality, and effectiveness before commercialization under the Public Health Service Act.
- Key players: Novartis AG, Gilead Sciences, Bristol Myers Squibb, and Pfizer Inc.
Patient Support and Services
- In the U.S., cell and gene therapy approvals are overseen by the Center for Biologics Evaluation and Research under the U.S. Food and Drug Administration, where companies must submit a Biologics License Application to prove safety, quality, and efficacy.
- Key players: Novartis AG, Gilead Sciences, Bristol Myers Squibb, and Pfizer Inc.
Market Growth
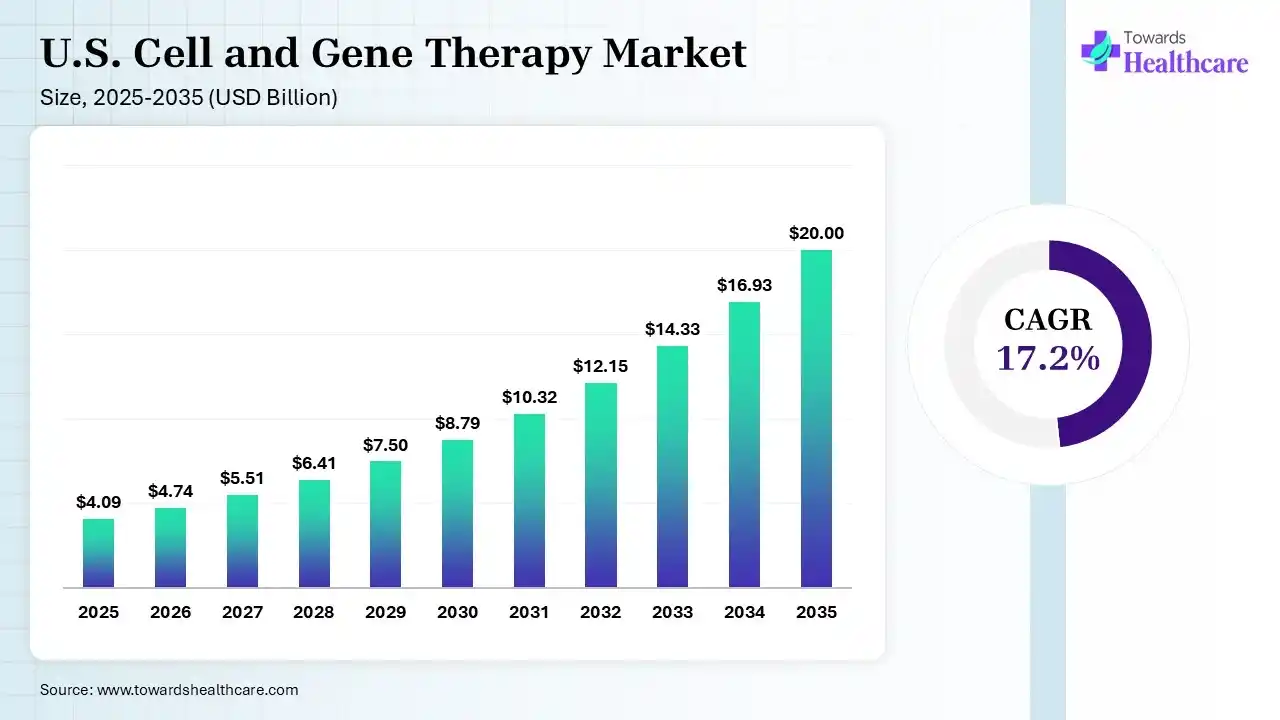
The U.S. cell and gene therapy market size was estimated at USD 4.09 billion in 2025 and is predicted to increase from USD 4.74 billion in 2026 to approximately USD 20 billion by 2035, expanding at a CAGR of 17.2% from 2026 to 2035. The market is rapidly expanding due to strong clinical innovation, increasing approvals of advanced therapies, and growing demand for curative treatments in oncology and rare genetic disorders. It is driven by heavy biotech investment, breakthroughs in gene editing and personalized medicine, and is positioned as a global leader in next-generation therapeutic development.
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
