
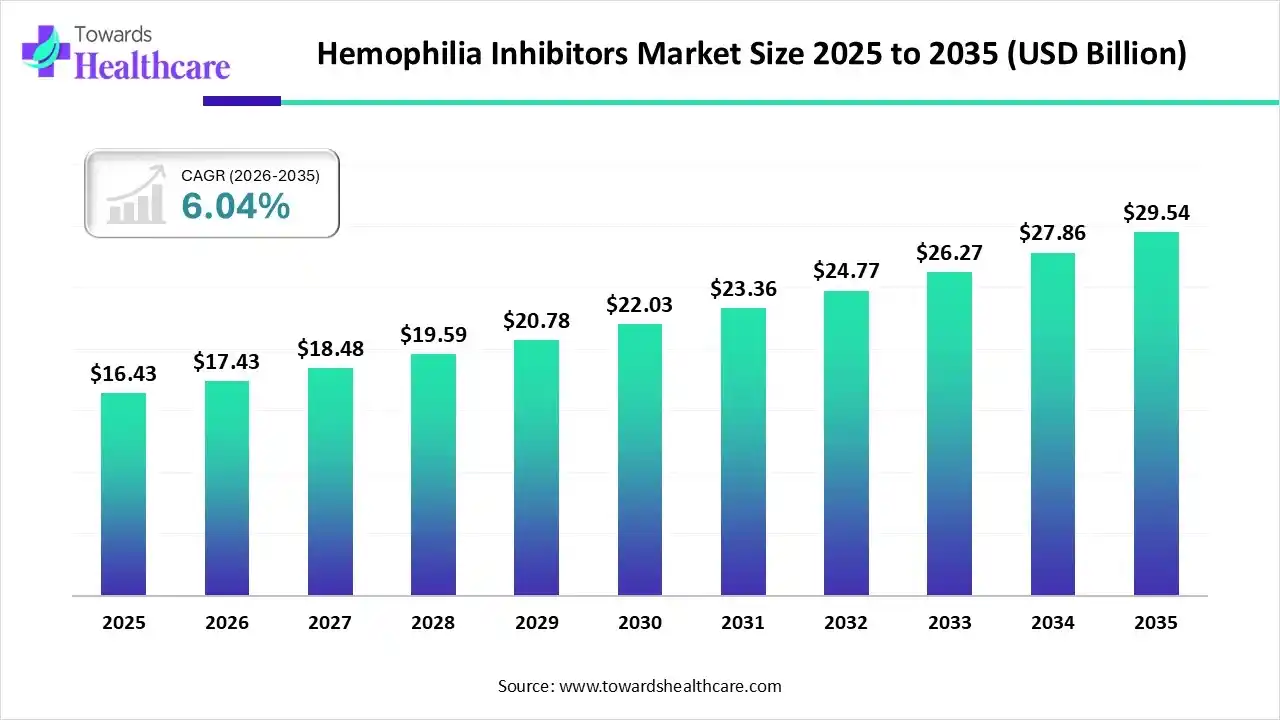
The hemophilia inhibitors market size stood at US$ 16.43 billion in 2025, grew to US$ 17.43 billion in 2026, and is forecast to reach US$ 29.54 billion by 2035, expanding at a CAGR of 6.04% from 2026 to 2035.
| Key Elements | Scope |
| Market Size in 2026 | USD 17.43 Billion |
| Projected Market Size in 2035 | USD 29.54 Billion |
| CAGR (2026 - 2035) | 6.04% |
| Leading Region | North America |
| Top Key Players | Pfizer Inc. (U.S.), Novo Nordisk A/S (Denmark), F. Hoffmann-La Roche Ltd (Switzerland), Bayer AG (Germany), Shire (Ireland), CSL Behring (Australia), Grifols, S.A. (Spain), Biogen Inc. (U.S.), Sanofi S.A. (France), BioMarin Pharmaceutical Inc. (U.S.), Takeda Pharmaceutical Company Limited (Japan), Octapharma AG (Switzerland), Sobi (Sweden), Pfizer Inc. (U.S.) |
In addition to 5% of patients with mild and moderate hemophilia A and 3% of patients with hemophilia B, over 30% of individuals with severe hemophilia A will develop inhibitors. By making it more difficult to manage bleeding episodes, inhibitors have a detrimental impact on disease morbidity and mortality, raise the cost of care dramatically, and increase the financial and psychological strain on patients and their families. The main therapy options now offered for people with inhibitors are bypassing agents (BPAs), even though several cutting-edge therapeutic solutions have been developed to treat bleeding and lessen the disease burden in patients without inhibitors. As a result, there is ongoing research in the field into new strategies to deal with bleeding while antifactor inhibitors are being used.
The term "inhibitors" refers to IgG alloantibodies to exogenous clotting factor VIII (FVIII) or factor IX (FIX), which neutralize the effects of infused clotting factor concentrates (CFCs) in hemophilia. Inhibitors are more common in individuals with severe hemophilia than in those with moderate or mild hemophilia, and they are more common in patients with hemophilia A than in those with hemophilia B. Hemophilia B patients have a cumulative rate of inhibitor development as low as 5%.
Recently, machine learning (ML) and artificial intelligence (AI) approaches have been used to predict the clinical severity of hemophilia. Machine learning (ML) in hemophilia has demonstrated encouraging outcomes in the following domains, which pertain to the virtual aspects of artificial intelligence: predicting the severity of the disease, identifying factor V as a critical modifier of thrombin generation in mild to moderate hemophilia A, creating a hemophilia-specific user-centered application, gene therapy, calculating the risk of myocardial infarction, and determining CRISPR/Cas9 nuclease off-target for hemophilia treatment. Artificial intelligence has the potential to revolutionize hemophilia. Clinicians can diagnose and treat hemophilia more accurately with the use of a variety of artificial intelligence (AI) techniques.
Blooming Therapies
The companies are focused on developing novel non-factor therapies to offer simple and proactive clotting controls, where they are also utilizing bispecific antibodies, which will help in reducing the diagnosis frequencies.
Shift towards factor rebalancing
There is a growth in the demand for factor rebalancing therapies, which enhance clotting by stimulating the proteins, which is driving their development and creating new opportunities as well.
Growing Advancements
The companies are developing new drugs with reduced dosing frequency, like once a week or once a month, which can be administered subcutaneously, in order to enhance patient adherence to the treatments.
Personalized medicine is the practice of tailoring a patient's treatment based on their medical and genetic traits. This approach can help with early intervention and a more effective treatment plan by identifying those who are more prone to acquire inhibitors. Recent years have seen a paradigm shift toward personalized medicine, which uses advances in genetic testing, pharmacogenomics, and pharmacokinetics to tailor treatment to each patient's needs. Due to the rapid advancement of medical technology, the field of hemophilia therapy has seen a significant expansion in the therapeutic options accessible to patients.
The average cost of medication for hemophiliacs is around $393,000 annually. Most of the total annual health care expenses per patient, which ranged from $213,874 to $869,940, are related to the cost and severity of preventive therapy with FVIII replacement concentrates, bypassing agents, and, most recently, emicizumab. The annual costs of FVIII therapy for a group of adult patients without inhibitors increased with time, while the costs for those who had inhibitors were significantly higher. Only two trials revealed indirect costs, which were $13,220 per year for individuals without inhibitors and $27,978 for those with inhibitors. Parents of children with HA spent $8,252 on non-mental health treatments and $258 on mental health care per year.
Gene therapy offers a highly attractive possibility for treatment by making the body create FVIII or FIX on its own after transplanting a functional copy of the gene. Hemophiliacs have long been thought to benefit from gene therapy. Gene therapy is anticipated to command a higher price, at least initially, in order to recoup the cost of development. Effective gene therapy, on the other hand, would improve quality of life, reduce comorbidities, and eliminate the need for frequent medical interventions by preventing breakthrough bleeding and microhemorrhages by ongoing endogenous synthesis of the clotting factor. As a result, gene therapy has a lot to offer society and the healthcare industry.
North America dominated the hemophilia inhibitors market in 2025, fueled by a highly developed healthcare system, significant R&D investments, and the presence of large pharmaceutical corporations. Patients may receive cutting-edge therapies like gene therapy because of the region's widespread insurance coverage. The United States leads the globe in hemophilia research and treatment innovation, which is led by Pfizer, BioMarin, and Takeda. Further solidifying North America's leadership in hemophilia treatment is the region's extensive use of digital health technologies, including telemedicine platforms and AI-powered monitoring tools.
The U.S. Hemophilia Inhibitors Market Trends
The US Centers for Disease Control and Prevention (CDC) estimate that hemophilia affects around 1 in 5,617 live male births. Approximately 30,000 to 33,000 males in the US have hemophilia. The continuous investment in research and development has led to the creation of several new hemophiliac developments in the United States, each of which provides unique benefits to sufferers. Benefits include less treatment burden, improved adherence, and improved quality of life, including improved ability to exercise and achieve significant life goals.
The Canada Hemophilia Inhibitors Market Trends
Members of the Canadian Hemophilia Society (CHS) get access to first-rate activities and services. Several goals have been set by the CHS for 2025. The biennial CHS Rendez-vous, which includes a medical and scientific symposium, will bring together the entire Canadian bleeding disorder community, including patients and their families, physicians, nurses, physiotherapists, and social workers, to discuss the latest developments in bleeding disorder knowledge, research, and therapies. Keeping up the distribution of clinical and research funding to leading Canadian researchers in bleeding disorders.
Asia Pacific is estimated to host the fastest-growing hemophilia inhibitors market during the forecast period brought on by growing healthcare costs and hemophilia rates in heavily populated countries like China and India. Greater access to healthcare and improved understanding of hemophilia management are helping to facilitate the introduction of innovative medications. Governments and charitable organizations are playing a crucial role, particularly in poor countries, by financing diagnostic programs and offering financial support for treatments. Additional factors propelling the area market's expansion include clinical trials and local manufacturing activities aimed at reducing costs and facilitating access to innovative therapies.
The China Hemophilia Inhibitors Market Trends
The Hemophilia Treatment Centers Cooperative Network of China organized three national seminars in collaboration with the WFH. Pediatricians, physiotherapists, rehabilitation specialists, and nurses went from all across the country to offer comprehensive care for those with hemophilia. At a course for nurses, the process of training and certification for hemophiliacs was also described.
The India Hemophilia Inhibitors Market Trends
With 1.36,000 cases, India has the second-highest hemophilia case burden worldwide despite the condition being rare. Severe hemophilia A is treated with hemostatic drugs that stop or prevent bleeding or with repeated factor VIII replacement. Gene therapy for hemophilia A (FVIII deficiency) was first clinically studied in humans in February 2024 at Christian Medical College (CMC) in Vellore, India.
Europe is expected to grow at a considerable CAGR in the upcoming period. The rising prevalence of hemophilia, the increasing number of clinical trials, and favorable regulatory support augment market growth. The European Medicines Agency (EMA) regulates the approval of hemophilia inhibitors in Europe. The increasing R&D investments and collaboration among key players also contribute to market growth.
The UK Hemophilia Inhibitors Market Trends
In October 2025, the MHRA approved concizumab (Alhemo) to prevent or reduce the frequency of bleeding disorders in people aged 12 years and older with hemophilia A or B with inhibitors. A total of 52 trials were registered on the clinicaltrials.gov website related to hemophilia inhibitors from the UK, highlighting the need for novel inhibitors.
The Middle East & Africa are expected to grow at a notable CAGR in the foreseeable future. The growing research and development activities, the rising adoption of advanced technologies, and the increasing awareness of hemophilia treatment boost the market. Government organizations launch initiatives to encourage the general public about the screening and early diagnosis of hemophilia.
The UAE Hemophilia Inhibitors Market Trends
Abu Dhabi marked a truly historic moment in medical care with the first administration of a gene therapy injection, CASGEVY, for inherited blood disorders, including hemophilia, in the UAE. In addition, Pfizer launched a new and innovative therapy for people living with hemophilia in the UAE.
South America is expected to grow significantly in the hemophilia inhibitors market during the forecast period, due to growing early diagnosis, driven by increasing health awareness through the government health programs. The growing healthcare investments are also increasing the accessibility of these inhibitors, which is promoting the market growth.
Brazil Hemophilia Inhibitors Market Trends
The growing incidence of hemophilia in Brazil is increasing the demand for its inhibitors. Additionally, due to high unmet medical needs, they are also being preferred for effective treatment of hemophilia, which is increasing their production rates. Additionally, increasing government support is also increasing their affordability and accessibility, encouraging their use.
| Companies | Headquarters | Hemophilia Inhibitors |
| Roche | Basel, Switzerland | Hemlibra |
| Takeda | Tokyo, Japan | FEIBA |
| Novo Nordisk | Bagsvaerd, Denmark | NovoSeven RT and Alhemo |
| Sanofi | Paris, France | Qfitlia |
| Pfizer | New York, U.S. | Hympavzi |
In April 2024, according to Aamir Malik, Chief U.S. Commercial Officer and Executive Vice President, Pfizer, this milestone demonstrates the company's ongoing efforts to improve the quality of care for individuals with hemophilia by delivering a medication that may provide both long-term bleed protection and value to the healthcare system due to its one-time administration.