
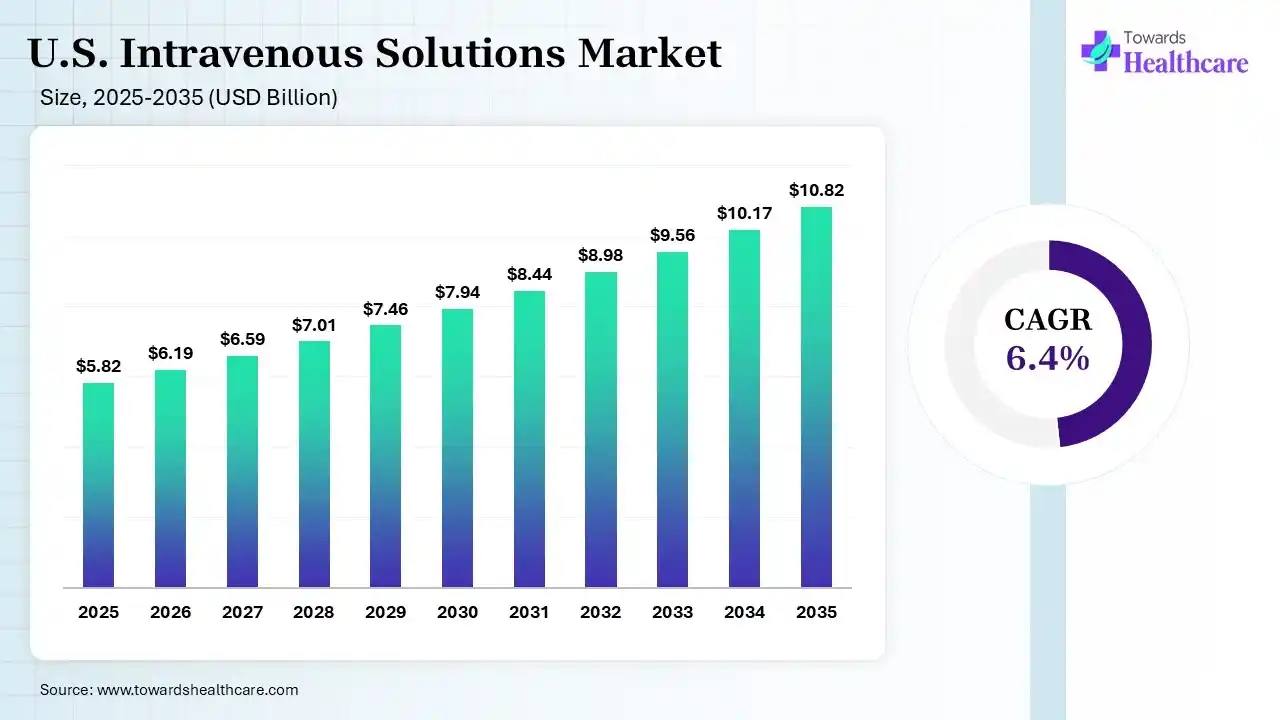
The U.S. intravenous solutions market size was estimated at USD 5.82 billion in 2025 and is predicted to increase from USD 6.19 billion in 2026 to approximately USD 10.82 billion by 2035, expanding at a CAGR of 6.4% from 2026 to 2035. The U.S. intravenous solutions market is growing because of the rising prevalence of chronic and acute health diseases that need fluid therapy, boosted by technological development and increasing healthcare infrastructure.
The U.S. intravenous solutions market is growing, as IV fluids are particularly formulated liquids that are injected into a vein to avoid or manage dehydration. They are applicable to patients of various ages who are injured, sick, dehydrated from a workout or heat, or suffering from surgery. IV fluids are liquids injected into an individual’s veins via an IV tube. IV fluids are also applied specifically to treat an emerging electrolyte or acid-base disorder. IV fluids are significant in the management of certain subtypes of hypernatremia, hyponatremia, metabolic alkalosis, and metabolic acidosis. IV fluid restores fluid to the intravascular space, and IV fluids are applied to enable the movement of fluid among compartments due to osmosis.
AI-driven technology is increasingly playing an important role in different aspects of medical care, involving infusion therapy. Intravenous therapy includes the administration of medications, fluids, or nutrients via an intravenous (IV) line, and AI-based technology improves safety, effectiveness, and patient results. An AI-based algorithm is responsible for processing the sensor data in real-time. It is trained to recognize patterns and irregularities related to blood backflow actions. This training includes a dataset of known backflow events and non-events. An AI-driven auto-shutoff mechanism would engage with different functions of IV services.
| Table | Scope |
| Market Size in 2026 | USD 6.19 Billion |
| Projected Market Size in 2035 | USD 10.82 Billion |
| CAGR (2026 - 2035) | 6.4% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product Type, By Nutritional Content, By Volume, By End User, By Packaging Type, By Distribution Channel |
| Top Key Players | Baxter International Inc., ICU Medical, Inc., B. Braun Medical Inc., Fresenius Kabi USA, Pfizer Inc. |
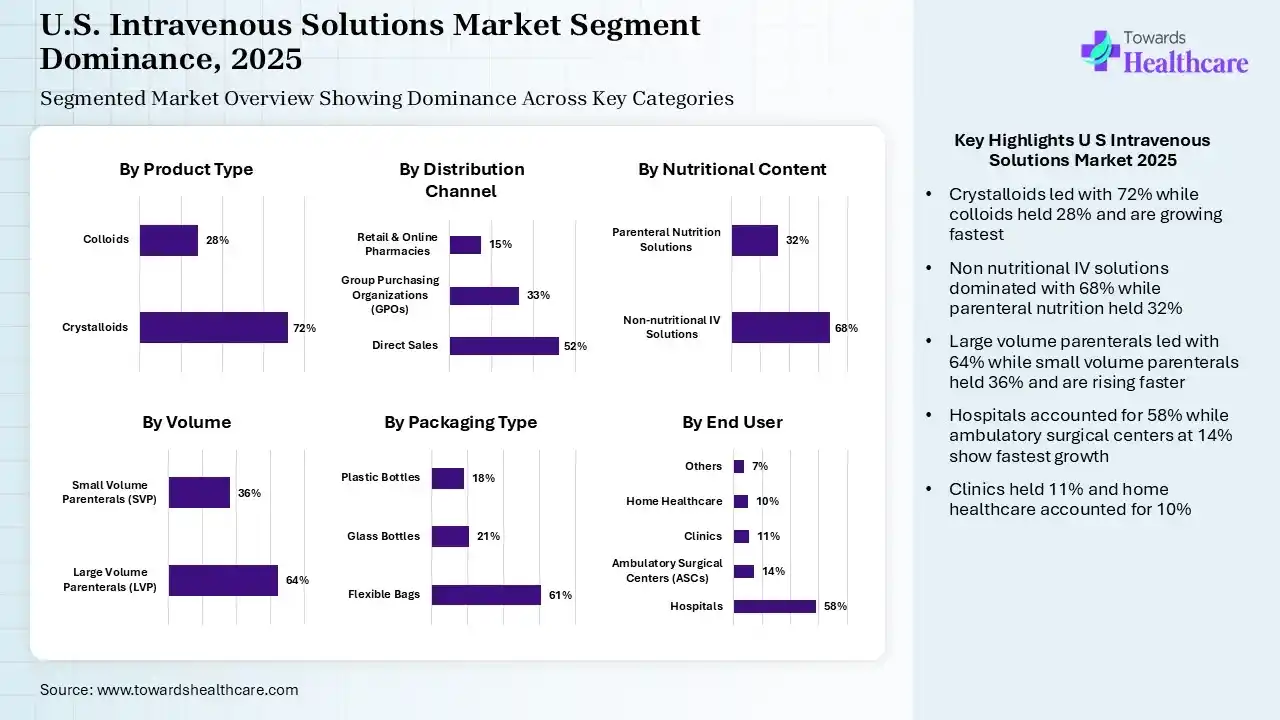
| Segment | Share 2025 (%) |
| Crystalloids | 72% |
| Colloids | 28% |
Crystalloids Segment Led the U.S. Intravenous Solutions Market in 2025
The crystalloids segment contributed the largest market share of 72% in 2025, as crystalloids are most often preferred because of their ease of administration and reduced challenges of allergic reactions. These fluids are further classified on the basis of their electrolyte composition, osmolarity, and metabolic properties. They are applied for growing the intravascular volume in conditions such as dehydration, hypotension, and sepsis.
The colloids segment held the second-largest share of 28%, and is expected to grow at the fastest CAGR of 7.0% in the market, as colloids have larger molecules, are affordable, and also provide swifter volume increase in the intravascular space. Colloids are better than crystalloids at escalating the circulatory volume, because their larger molecules are retained more simply.
| Segment | Share 2025 (%) |
| Non-nutritional IV Solutions | 68% |
| Parenteral Nutrition Solutions | 32% |
Non-nutritional IV Solutions Segment Led the U.S. Intravenous Solutions Market in 2025
The non-nutritional IV solutions segment contributed the largest market share of 68%, as it offers subjective developments in energy levels, mental clarity, and overall well-being following administration. It provides rapid recovery from conditions like dehydration, hangovers, jet lag, and physical exhaustion when managed with IV fluids improved with electrolytes, B vitamins, and vitamin C.
The parenteral nutrition solutions segment held a significant share of 32% of the market in 2025. Parenteral nutrition is the ability to help patients who can’t eat or absorb nutrients via conventional means. It allows nutritional services to be infused for 8 to 18 hours daily. It provides satisfactory nutrients in the face of gastrointestinal dysfunction.
| Segment | Share 2025 (%) |
| Large Volume Parenterals (LVP) | 64% |
| Small Volume Parenterals (SVP) | 36% |
The Large Volume Parenterals (LVP) Segment held the Largest Share in the Market in 2025
The large volume parenterals (LVP) segment contributed the largest U.S. intravenous solutions market share of 64% in 2025, as it is a single-dose injection that is designed for intravenous applications and is packaged in containers labelled as containing more than 100 mL. It is applied to offer fluids, electrolytes, and nutrition intravenously. These solutions in either the plastic bag or the glass bottle flow from the containers to the patient via an administration set.
The small volume parenterals (SVP) segment held a notable share of 36% of the market in 2025, expected to grow at the fastest CAGR of 6.8% of the market, as small volume parenterals have volumes less than 100mL and involve solutions, emulsions, suspensions, and dry powders. It is packaged in various ways based on the intended use.
| Segment | Share 2025 (%) |
| Hospitals | 58% |
| Ambulatory Surgical Centers (ASCs) | 14% |
| Clinics | 11% |
| Home Healthcare | 10% |
| Others | 7% |
The Hospitals Segment held the Largest Share in the Market in 2025
The hospitals segment contributed the largest U.S. intravenous solutions market share of 58% in 2025, as in hospital applicable to manage pain and nausea by rapidly achieving therapeutic levels, and they are more reliably and completely absorbed compared with medications given by various routes of injection. IV infusion therapy delivers blood, fluids, nutrients, and medication directly to the bloodstream to supports to feel better rapidly.
The ambulatory surgical centers (ASCs) segment held the second largest share of 14%, and is expected to grow at the fastest CAGR of 7.1% of the market, as ambulatory infusion centers offer optimum services for field physicians committed to bringing timely, efficient treatments. It is making treatments more effective and patient-friendly. It provides comfortable treatment environments with private or semi-private rooms.
The clinics segment held a notable share of 11% the market, as IV therapy use in clinics delivers fluids straight to the bloodstream, offering rapid and efficient hydration that helps significant organ function and supports to feel better almost rapidly.
The home healthcare segment held a notable share of 10% of the U.S. intravenous solutions market. Home infusion therapy reduces travel expenses, lowers hospital admissions, and limits the likelihood of hospital-acquired infections, eventually creating more positive patient results. Home infusion therapy enhances end-of-life care by increasing the time patients spend with their family at home.
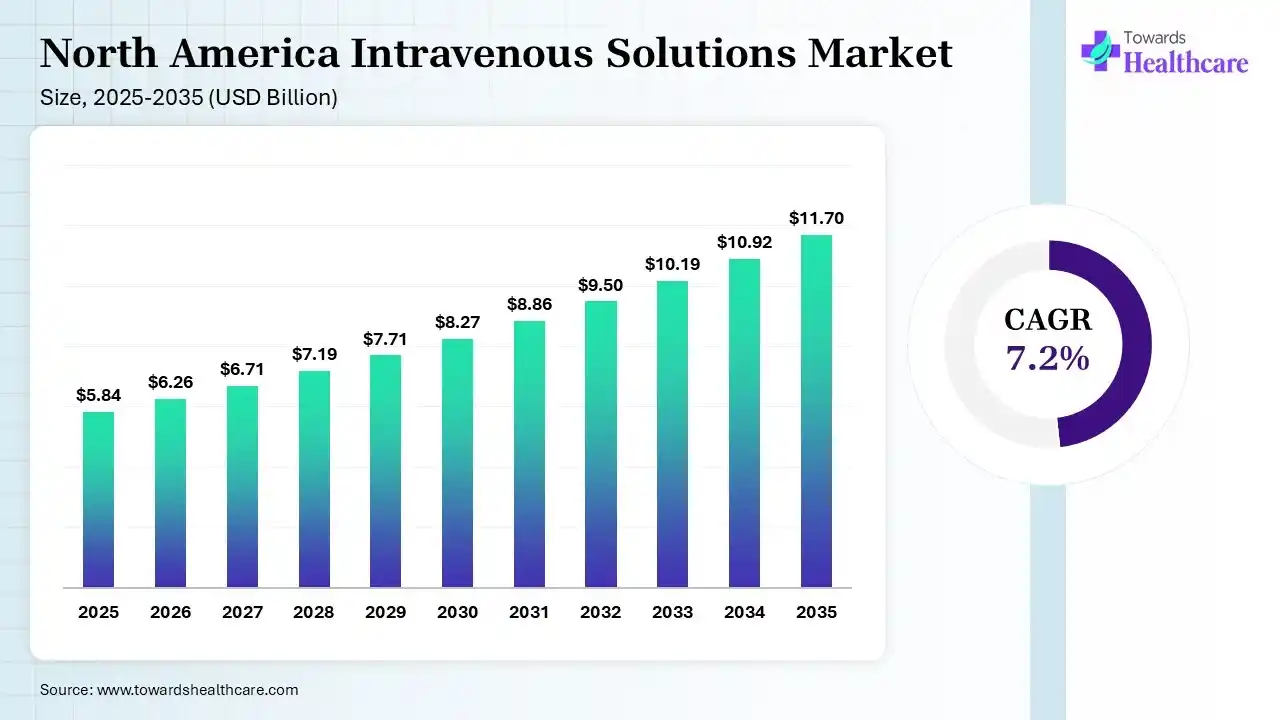
The North America intravenous solutions market size was estimated at USD 5.84 billion in 2025 and is predicted to increase from USD 6.26 billion in 2026 to approximately USD 11.7 billion by 2035, expanding at a CAGR of 7.2% from 2026 to 2035.
R&D:
Manufacturing Processes:
Patient Services:

| Company | Headquarters | Latest Update |
| Baxter International Inc. | United States | In March 2026, Baxter International Inc., a novel leader in infusion therapies and platforms, debuted the IV Verify Line Labeling System, an automated service, at the 2026 American Organization for Nursing Leadership Conference. |
| ICU Medical, Inc. | United States | In May 2025, ICU Medical Inc., in partnership with VisionHealth GmbH, announced the launch of MyAcapella, a mobile application intended to support Oscillating Positive Expiratory Pressure (OPEP) therapy for patients with chronic respiratory conditions. |
| B. Braun Medical Inc. | United States | In December 2025, B. Braun Medical Inc., a leader in smart infusion therapy and pain management, launched Piperacillin and Tazobactam, one of the most utilized injectable antibiotics in the U.S. |
| Fresenius Kabi USA | United States | Fresenius Kabi provides a broad range of generic intravenously administered medications. Significantly severely ill patients in hospitals are treated with these drugs. |
| Pfizer Inc. | United States | In September 2025, Pfizer and BioNTech remain deeply committed to making safe and efficient vaccines that supports to protect lives in the U.S. and around the world. |
Strengths
Weakness
Opportunities
Threat
By Product Type
By Nutritional Content
By Volume
By End User
By Packaging Type
By Distribution Channel