The growth in regulatory complexities, expanding clinical trials, and a shift towards AI-driven cloud platforms are promoting the use of regulatory information management systems. Furthermore, rapid healthcare digitalization and frequent regulatory updates are also increasing their use.
The advancements in the R&D activities across the pharmaceutical, medical devices, and biotechnology industries are increasing the development of new products, where avoiding the complexities in the clinical trials and regulatory approval is driving the use of regulatory information management systems (RIMS). Expanding healthcare IT infrastructure is also increasing the development of new RIMS platforms, which is promoting their launches and creating new opportunities for next-generation RIMS platforms.
The Regulatory Backbone: Why RIMS is the New Standard in Life Sciences
The regulatory information management system encompasses the software platforms responsible for the management, organization, and streamlining of regulatory data and compliance processes across life science organizations, which is promoting their adoption rates. The system combines centralization, automation, compliance, and lifecycle management in one platform, where each platform plays a vital role.
- Centralization of Regulatory Data: All regulatory information, such as product registration, submission, labelling, and approvals are consolidated in a single repository by the RIMS, which enhances the accuracy and minimizes the risk of duplication.
- Global Regulatory Compliance: The regulatory information management system helps in the navigation of complex regulations across numerous countries, promoting the extraction of information for submission formats and deadlines for regulatory authorities, as well as offering regulatory updates.
- Lifecycle Management: The RIMS platform offers help in the management of the products throughout their lifecycle, that is, from their initial development till their post-market maintenance, supporting their submission management, tracking variations, renewals, approvals, and recalls.
- Automation: The regulatory information management systems offer automation, enhancing workflow efficiency and promoting automated documentation, submission, and approvals tracking, where it also provides notification to avoid deadlines and regulatory commitments.
- Auditability: The RIMS contains the ability to maintain auditable trails for the regulatory submissions and approvals, where it also offers real-time dashboards, analytics, and compliance reporting, enhancing the regulatory performance of the companies.
- Enhanced Integration: The easy integration of RIMS with other healthcare systems, such as quality management systems, clinical trial management systems, and laboratory information management systems, offers seamless data flow and faster access to the information, promoting their use.
- Scalability: The regulatory information management systems show improved compliance due to management of numerous products across various regions and languages, which is increasing their use across multiple organizations.
- Regulatory Intelligence: The RIMS also offers insights on the latest regulatory trends across multiple countries, promoting new strategies and changes as per regulations for faster regulatory approvals.
Next-Gen Compliance: Key Catalysts Behind RIMS Adoption
Turning Regulatory Complexities into Opportunities
The frequent updates in the regulations, with strict regulatory submission, compliance, and reporting requirements, where failure to comply leads to the rejection of products and their approvals. This, in turn, is increasing the challenges in the product approval for the companies developing multiple products, which is increasing the adoption of regulatory information management systems. The systems also help in the development of detailed data with minimal errors
Innovations Fueling RIMS Demand
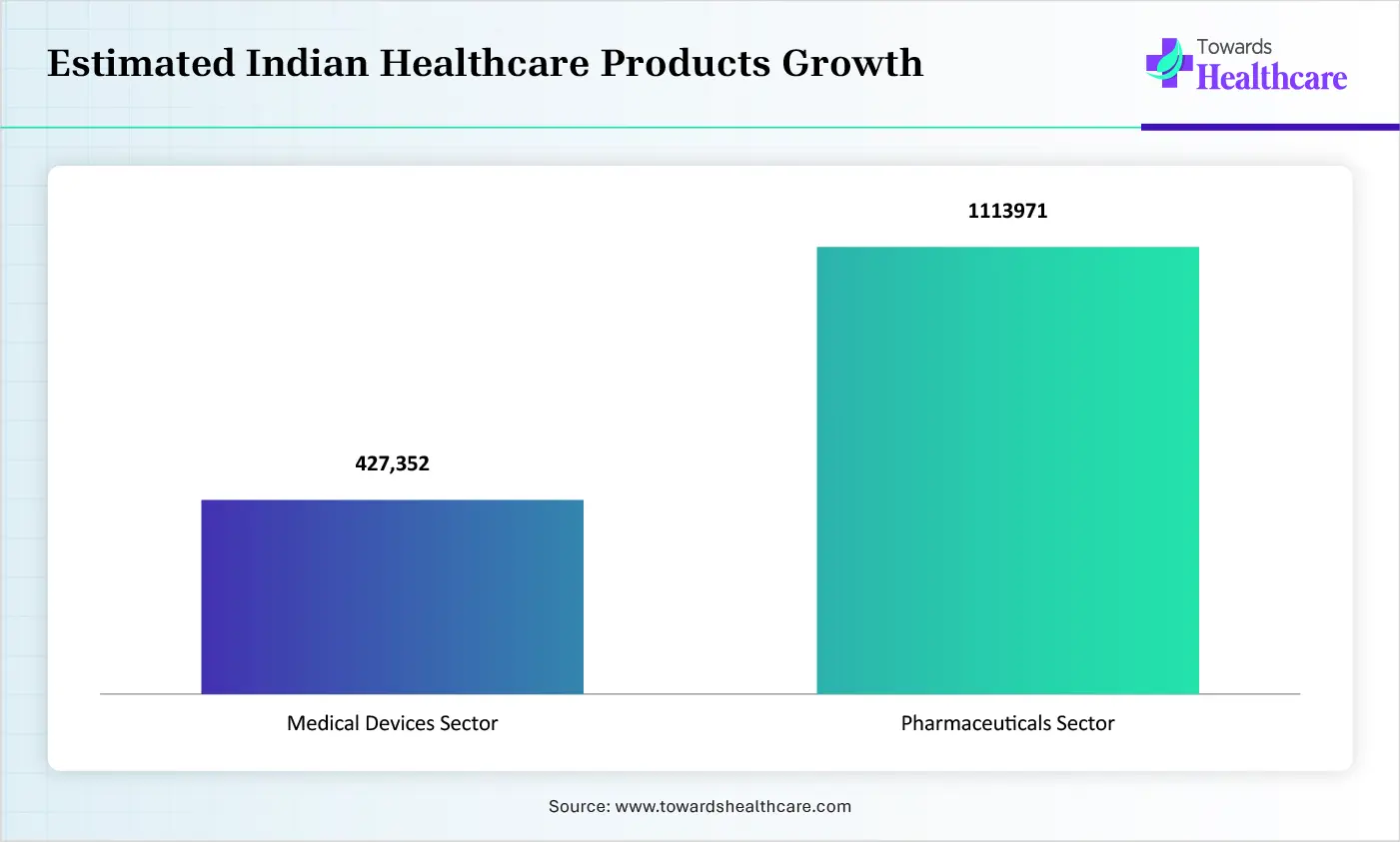
The growing innovations across pharmaceutical, biotechnology, and medical device companies are increasing the demand for regulatory information management systems to enhance the regulatory submission, approvals, and post-marketing activities of the products. Additionally, the growing development of next-generation medical devices, novel technologies, and complex pipelines is also increasing their use for lifecycle management and structured documentation.
Growth of Outsourced Regulatory Services
To deal with the growing regulatory and clinical trials complexities, various companies are outsourcing their products to third-party vendors. These vendor offers regulatory information management systems promoting improved clinical trials and regulatory approvals, while maintaining transparency with their external partners.
Regulatory Intelligence Powering Smart Compliance
The rapidly evolving regulatory standards are increasing the adoption of regulatory information management systems in order to meet the regulatory requirements, where the systems offer insights into the changing guidelines, regulations, and safety standards. It also helps in anticipating the regulatory changes and thus accelerates the product development and approval with minimal product rejection risk.
The Rise of Digital Health Ecosystem
The rapid digital transformation across the healthcare sector is increasing the use of various digital tools, eliminating the need for paper-based workflows. This is encouraging the adoption of the regulatory information management systems for efficient management of various regulatory processes, as well as promoting the development of new automated and cloud-based RIMS platforms.
Smart Systems, Fast Approvals: The AI Revolution in Regulatory Affairs
AI offered various applications, promoting automation of complex tasks, enhanced decision-making, and accelerating regulatory compliance, making it a game-changer in the regulatory information management systems. Some important features of AI enhancing RIMS efficiency are as follows,
- Automated Documentation: AI helps in automatic classification and submission of regulatory documents, reducing the need for submission preparation and manual tagging. It also helps in minimizing human errors and the time required for the regulatory filing.
- Regulatory Intelligence: The global regulatory updates, changes, and guidelines are efficiently detected by AI algorithms, which help companies to remain updated with the growing regulatory changes and reduce the risk of their non-compliance.
- Predictive Analytics: The predictive analytics help in predicting the potential bottlenecks, approval timelines, and promote planning for the regulatory submission and product launch timelines.
- Enhanced Accuracy: The AI helps in detecting the inconsistencies, non-compliance, and missing information in the regulatory documents, where it provides notifications, which help in enhancing the data integrity and regulatory compliance.
Stringent Regulations and Rising Drug Development Fuel North America�s RIMS Growth
North America held the major revenue share of the regulatory information management system market in 2025, due to the presence of major pharmaceutical and biotechnology industries, and stringent regulations, which promoted the adoption of the regulatory information management systems. Furthermore, growth in the R&D investments, advancements in the healthcare IT sector, and a rise in the development of new drug products and medical devices, also increased their use, where technological advancements also promoted their innovation.
Asia Pacific Emerges as a Fast-Growing Hub for Regulatory Information Management Systems
Asia Pacific is expected to show rapid expansion in the regulatory information management system market during the forecast period, due to rapid growth in the pharmaceutical and medical device sectors. This, in turn, is increasing the development of new products, while the growing clinical trial and regulatory complexities are also increasing the adoption of regulatory information management systems. Additionally, healthcare digitalization, the growing healthcare investments, and government initiatives are also encouraging their use.
Government-Led Regulatory Modernization
In July 2025, a whitepaper titled Streamlining Compliance in the Indian Healthcare Sector was submitted to NITI Aayog by a collaboration between the Healthcare Federation of India and EY India, where the paper highlights the roadmap for regulatory reform, which also supports the adoption of smart, technology-driven solutions and proposes digitisation, simplification, and centralisation to build a modern, efficient regulatory framework.
Europe Solidifying its RIMS Ecosystem
Europe is expected to show significant growth in the regulatory information management system market during the forecast period, due to a well-established regulatory framework and healthcare systems, which are increasing the use of regulatory information management systems. At the same time, growth in the pharma and biotech startups and technological advancements is also promoting their use and innovations.
Strategic Offerings of RIMS Innovators
| Companies | Headquarters | Regulatory Information Management System | Role Of the Systems |
| Veeva Systems | Pleasanton, California | Veeva Vault RIM Suite | Unified registration tracking, submissions, and archival� |
| ArisGlobal | Miami, Florida | LifeSpere RIM Platform | Automates regulatory workflows and data extraction |
| IQVIA | Durham, North Carolina | RIM Smart | Integrates global intelligence for real-time compilation |
| LRENZ Life Sciences Group | Frankfurt, Germany | docuBridge | eCTD submission management and compilation |
| EXTEDO | Munich, Germany | EXTEDOscope suite | End-to-end regulatory journeys and product registration |
| MasterControl | Salt Lake City, Utah� | Registrations software | Integrates quality management with regulatory submission tracking |
| Rimsys | Pittsburgh, Pennsylvania | Specialized RIMS | Supports medical device development, centralized product registration, and UDI data globally |
| Calyx | Nottingham UK | Calyx RIM | Simplified global submission management and health authority interaction� |
| Ennov | Paris, France | Unified RIM | Manage entrie regulatory lifecycle |
| Navitas Life Sciences | Princeton, New Jersey | pharmaREADY RIM Suite | eCTD submissions and document management |
The Future of RIMS: Cloud Adoption and Intelligent Technologies Lead the Way
Cloud-Based Platforms Accelerating the RIMS Growth
The growing shift from traditional on-premise systems to cloud-based RIMS platforms is increasing their innovation, where these platforms offer improved scalability, real-time updates, remote access, and lower infrastructure costs, driving their adoption rates across various organizations. These platforms offer easy access to the regulatory data from different countries, as well as provide alerts about changes in the regulatory requirements.
Seamless Integration Propel RIMS Efficiency
The growing application of the RIMS platforms is increasing their use across various industries, where their seamless integration with electronic health records, quality management systems, clinical trials management systems, and enterprise resource planning systems is also driving their adoption rates. Furthermore, their seamless data flow, regulatory traceability, data accuracy, and faster submission preparation are also propelling their use.
Technology Bloom Promotes RIMS Evolution
The growing use of advanced technologies is transforming the regulatory management systems, driving the development of more proactive, intelligent, and automated RIM systems. The companies are also focusing on enhancing their ability to detect compliance risk, regulation performance, and submission timelines, which, in turn, is creating new opportunities for RIMS.
Conclusion
The use of regulatory information systems is increasing globally due to frequent regulatory changes and the increasing development of pharmaceutical products and medical devices. The growing healthcare IT infrastructure, AI integration, and expanding industries are also increasing their demand. Furthermore, digital transformation, government initiatives, and healthcare investments are also promoting their innovations, creating future opportunities.
Expert Insights
According to a recent report on the regulatory information management system market, published on Towards Healthcare, experts highlight that increasing regulatory complexities and the rapid growth of the pharmaceutical and medical device industries are significantly driving the demand for advanced regulatory information management systems. Specialists emphasize that organizations are increasingly adopting cloud-based platforms to streamline compliance processes, manage global regulatory submissions, and improve data transparency. Additionally, the integration of artificial intelligence and automation is enhancing regulatory tracking, risk management, and document handling efficiency. Experts also note that rising clinical trials, expanding product pipelines, and stronger regulatory oversight will continue to accelerate the adoption of RIMS solutions worldwide.
About the Experts

Aditi Shivarkar
Aditi leads as Vice President at Towards Healthcare and brings over 15 years of experience in healthcare research, innovation, and strategy. She works closely with data from across the healthcare sector and turns it into clear direction that companies can actually use. Her work covers pharmaceuticals, medical devices, and digital health. She helps businesses understand where the market is going and how to respond with confidence. Aditi focuses on practical thinking, strong decision-making, and delivering real results that make a difference.

Aman Singh
Aman Singh brings over 13 years of experience in healthcare research and consulting. He studies global healthcare trends and keeps a close eye on areas like biotech, AI in healthcare, and new treatment approaches. At Towards Healthcare, he leads the research team and makes sure the work stays accurate, useful, and easy to understand. Aman breaks down complex changes in the industry and helps businesses make smart, informed decisions.

Piyush Pawar
Piyush Pawar works as Senior Manager for Sales and Business Growth at Towards Healthcare, with more than 10 years of experience in the healthcare space. He works directly with clients and helps them find the right research for their needs. He makes sure clients understand the insights and know how to use them in their business. Piyush builds strong relationships and focuses on helping companies grow by turning research into clear, practical action.




 Request Consultation
Request Consultation


