April 2026

The global regulatory information management system market size was estimated at USD 2.54 billion in 2025 and is predicted to increase from USD 2.77 billion in 2026 to approximately USD 6.09 billion by 2035, expanding at a CAGR of 9.14% from 2026 to 2035.
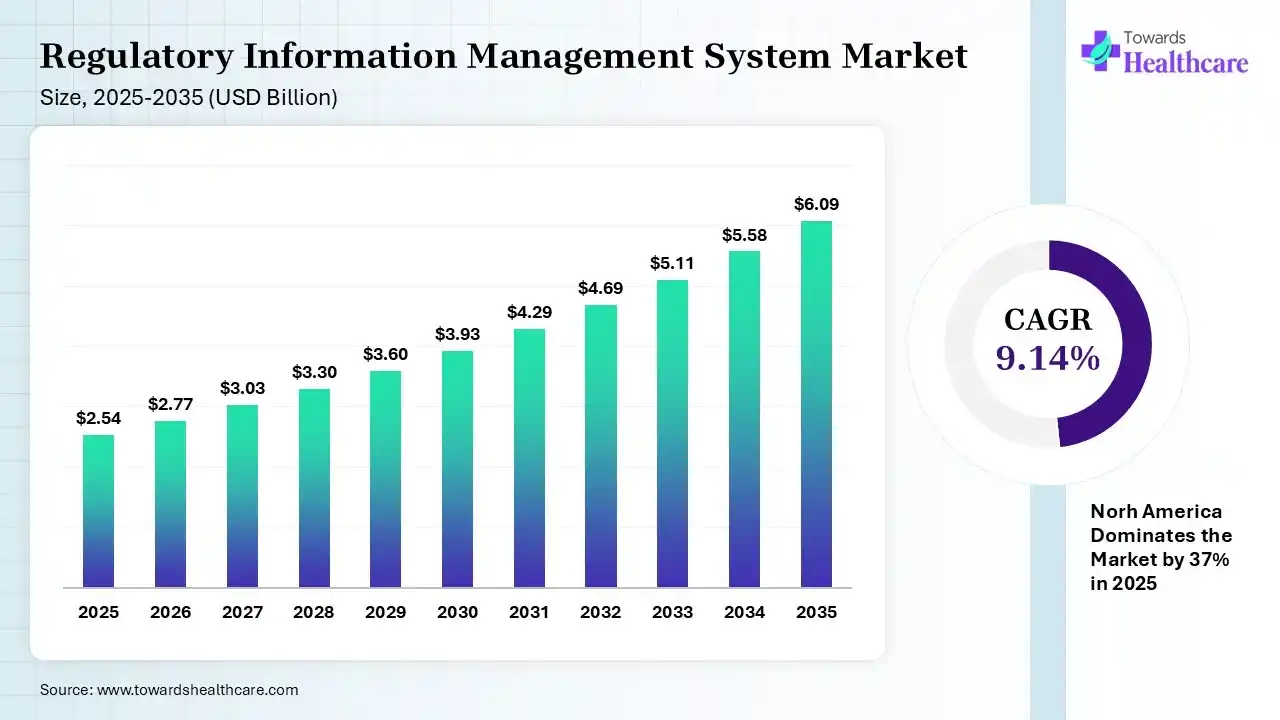
The growing regulatory updates, clinical trials, and healthcare digitalization are increasing the use of regulatory information management systems (RIMS). The growing R&D activities, technological advancements, and new platform launches are also enhancing the market growth.
The regulatory information management system market is driven by growing regulatory complexities, clinical trials, and a shift towards AI-driven cloud platforms. The regulatory information management system refers to the software platform developed for the management, organization, and streamlining of regulatory data and compliance processes across life science organizations. These systems help in centralized regulatory data management, submission management, lifecycle management, analytics, global regulatory tracking, and workflow automation.
AI offers a wide range of applications in the regulatory information management system market, promoting automated documentation and submission, where it also helps in data extraction and validation with enhanced accuracy. It also analyzes global regulations, predicts regulatory approval timelines, and offers faster decisions. It also enhances the integration of RIMS with other healthcare systems, increasing their use and promoting the accuracy, efficiency, and compliance with regulatory operations.
Growth in R&D Activities
The expanding pharmaceutical, biotechnology, and medical devices industries are increasing the development of new products, where a rise in investments is also propelling their advancements. This is increasing the adoption of the regulatory information management systems to reduce the regulatory burden, clinical trials complexities, and accelerate the development of new products. They also enhance regulatory compliance, reduce manufacturing errors, streamline workflows, and propel the product approval rates.
High Cost and Complex Integration
The regulatory information management systems are often expensive due to their manufacturing, deployment, and customization costs, and their challenges in integration with other healthcare systems increase the demand for skilled personnel. This reduces their use, limiting the regulatory information management system market growth.
Healthcare Digitalization
The growing digital transformation in the healthcare sector is increasing the use of advanced regulatory information management systems. It is also increasing the shift towards the use of cloud-based RIMS platforms to enhance accessibility, affordability, and scalability, where their integrations with other health care management systems are also increasing their adoption rates. New features offering electronic submission, real-time dashboards, analytics, digital recordkeeping, and regulatory integration are also being developed, which is increasing their use.
The frequent changes in the regulations, depending on the global regulatory standards and harmonization initiatives, increase the demand for detailed and safe data, where their failure to comply leads to the rejection of products and their approvals, which increases the demand for regulatory information management systems.
The cloud-based regulatory information management system (RIMS) offers real-time data access, faster system deployment, regulatory updates, enhanced scalability, and lower implementation costs, which is increasing the shift from traditional on-premise systems to cloud-based RIMS platforms.
The growth in technological advancements is driving the development of new regulatory information management systems with advanced analytics, improved traceability, automation, and compliance verification features, and enhanced integration with clinical trials systems and quality management systems.
| Table | Scope |
| Market Size in 2026 | USD 2.77 Billion |
| Projected Market Size in 2035 | USD 6.09 Billion |
| CAGR (2026 - 2035) | 9.14% |
| Leading Region | North America by 37% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By End Use, By Region |
| Top Key Players | Veeva Systems, ArisGlobal, IQVIA, LRENZ Life Sciences Group, EXTEDO, MasterControl, Rimsys, Calyx, Ennov, Navitas Life Sciences |
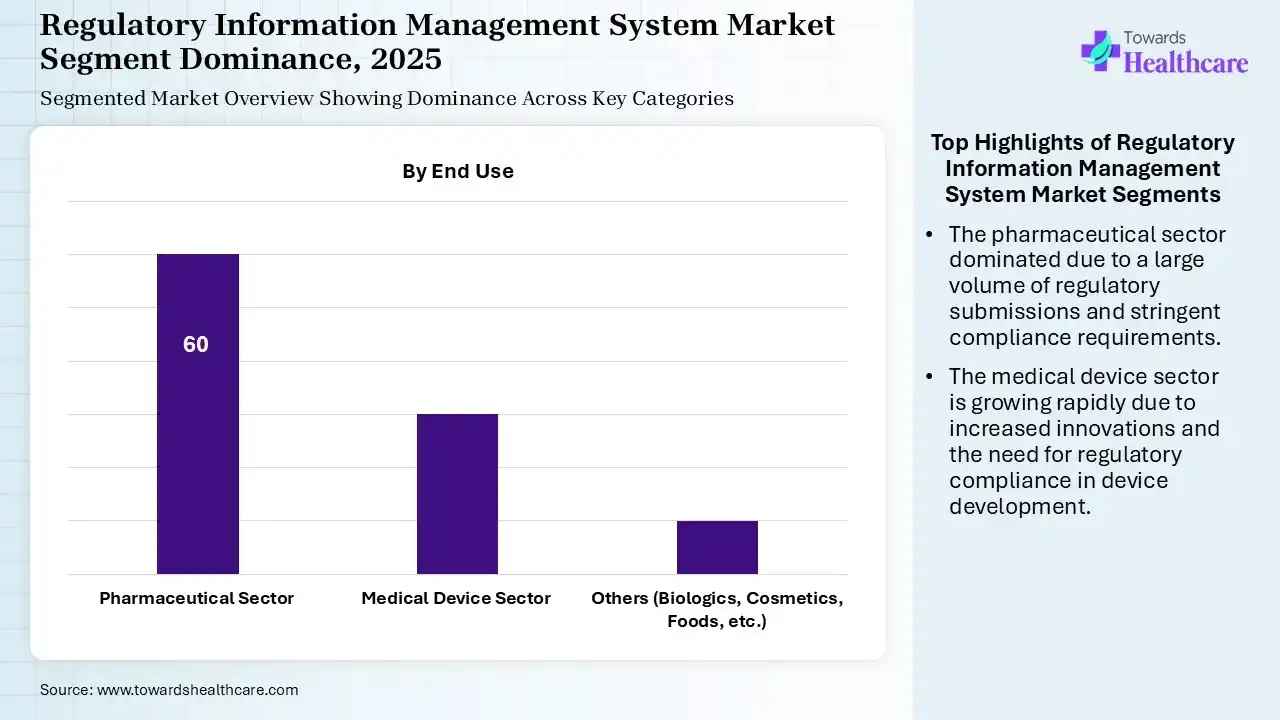
Why Did the Pharmaceutical Sector Segment Dominate in the Market in 2025?
The pharmaceutical sector segment held the largest revenue share of the regulatory information management system market by 60% in 2025, due to a large volume of regulatory submissions of various products. The stringent regulations, complex drug development process, and frequent drug regulation updates also increased the adoption of regulatory information management systems to meet the stringent regulatory requirements.
Medical Device Sector
The medical device sector segment is expected to grow with the highest CAGR in the market during the predicted time, due to growing medical device innovations. This, in turn, is increasing the use of regulatory information management systems for regulatory compliance and data management, where the expanding healthcare digitalization is also increasing the advancements of the devices as well as regulatory systems.
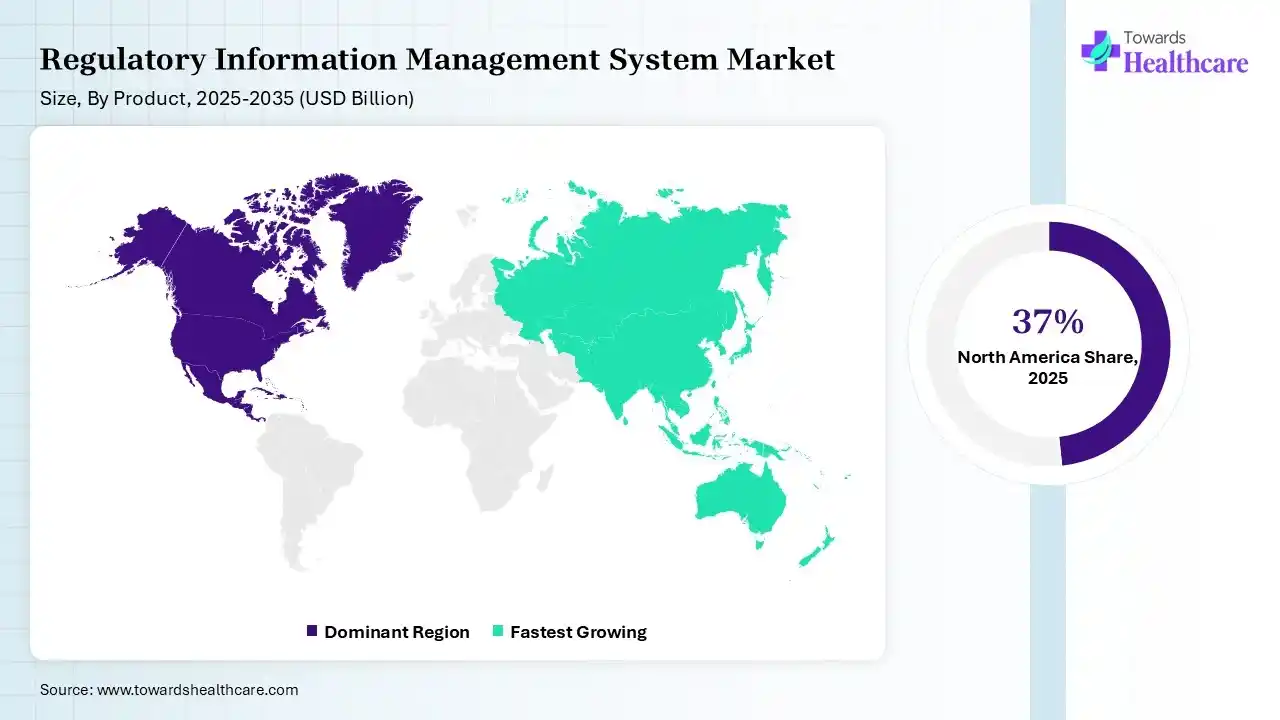
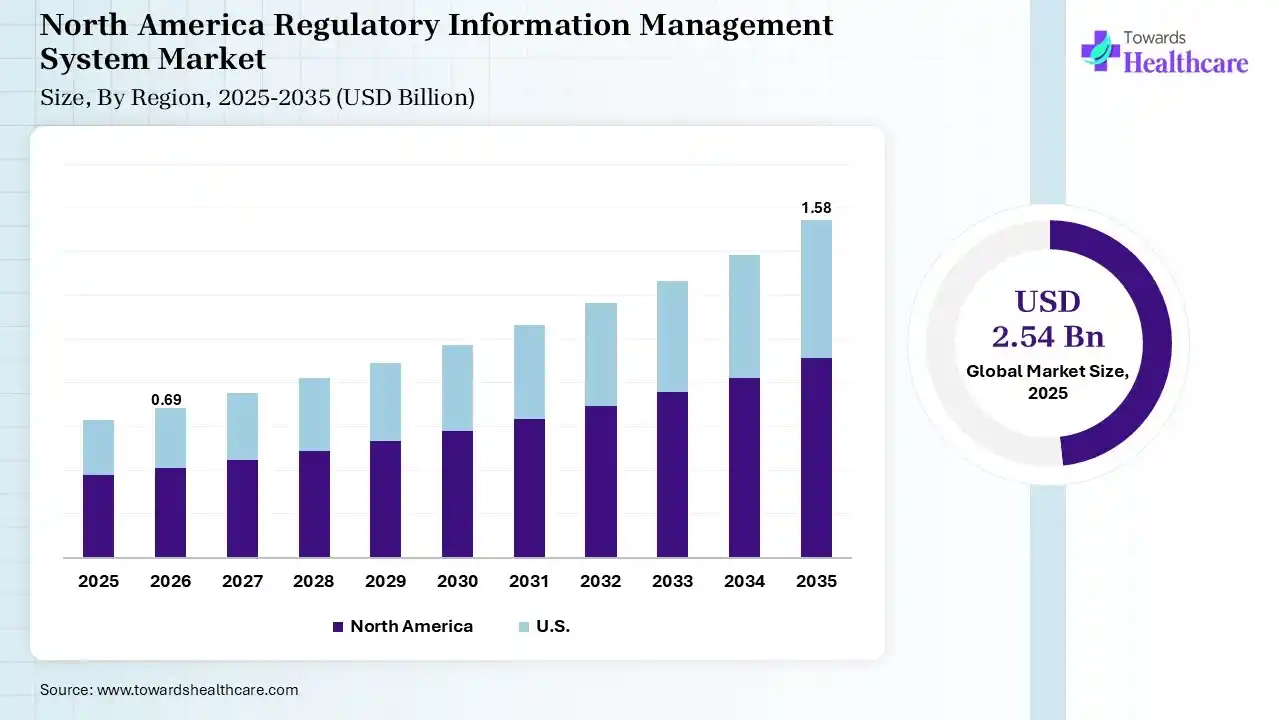
North America dominated the regulatory information management system market by 37% in 2025, due to the presence of major pharmaceutical and biotechnology industries, where the stringent regulations also increased the adoption of the regulatory information management systems. High R&D investments, growth in the development of new drug products and medical devices, and increased their use, where technological advancements promoted their innovation and market growth.
U.S. Market Trends
Strict regulatory requirements for the approval of the products are increasing the adoption of the regulatory information management systems across the U.S. At the same time, the growing advancements in the healthcare IT sector are also increasing their use for faster drug and medical device development, where the growing R&D infrastructure and technological advancements are also increasing their use and innovations.
Asia Pacific is expected to host the fastest-growing regulatory information management system market during the forecast period, due to an expansion in the pharmaceutical and medical device sectors. The growing clinical trials and regulatory complexities are also increasing the adoption of regulatory information management systems, where the growing healthcare investments and government initiatives are also enhancing the market growth.
India Market Trends
Rapid expansion of the pharmaceutical and biotechnology companies in India is increasing the adoption of regulatory information management systems, where the growing drug approvals and clinical trials are also increasing their use to meet the set regulatory standards. Additionally, growing healthcare digitalization is also promoting their adoption rates.
Europe is expected to grow significantly in the regulatory information management system market during the forecast period, due to a well-established regulatory framework, which is increasing the use of regulatory information management systems to promote compliance with regulatory requirements across the healthcare sector. Growing clinical trials and technological advancements are also increasing their use and promoting market growth.
UK Market Trends
The growing pharmaceutical and biotechnology ecosys tems in the UK are increasing the use of regulatory information management systems for faster product development and approval. Additionally, stringent regulatory requirements and their frequent updates are also increasing their use in clinical trials and are driving the development of new systems.
Latin America is expected to show lucrative growth in the regulatory information management system market during the forecast period, due to growing regulatory complexities, which are driving the demand for regulatory information management systems. The strengthening regulatory frameworks are also promoting the demand for electronic submission, increasing the use of RIMS, where the expanding healthcare sector is also increasing its use and innovation, contributing to the market growth.
Brazil Market Trends
Brazil is experiencing a growth in the pharmaceutical and medical device sector, which is increasing the demand for regulatory information management systems for their regulatory compliance and lifecycle management. Growing shift towards the electronic submission and post-market surveillance is also increasing the adoption of these systems, as well as new cloud-based RIMS.
MEA is expected to show notable growth in the regulatory information management system market during the forecast period, due to growing healthcare investments, which are increasing the adoption of advanced solutions like regulatory information management systems, where the digitization is also encouraging their use. Additionally, modernizing regulatory frameworks and technological innovations are also propelling the market growth.
The growing digital regulatory transformation in Saudi Arabia is increasing the adoption of regulatory information management systems to enhance the safety standards, documentation management, compliance monitoring, and submission tracking. Growing R&D activities and healthcare investments are also increasing their adoption rates.

| Companies | Headquarters | Regulatory Information Management System |
| Veeva Systems | Pleasanton, California | Veeva Vault RIM Suite |
| ArisGlobal | Miami, Florida | LifeSpere RIM Platform |
| IQVIA | Durham, North Carolina | RIM Smart |
| LRENZ Life Sciences Group | Frankfurt, Germany | docuBridge |
| EXTEDO | Munich, Germany | EXTEDOscope suite |
| MasterControl | Salt Lake City, Utah | Registrations software |
| Rimsys | Pittsburgh, Pennsylvania | Specialized RIMS |
| Calyx | Nottingham UK | Calyx RIM |
| Ennov | Paris, France | Unified RIM |
| Navitas Life Sciences | Princeton, New Jersey | pharmaREADY RIM Suite |
By End Use
By Region
April 2026
April 2026
April 2026
April 2026