U.S. respiratory durable medical equipment refers to long-lasting, reusable medical devices designed to support patients with breathing difficulties. These devices, such as oxygen concentrators, ventilators, and CPAP/BIPAP machines, are used in home or clinical settings to manage chronic respiratory conditions and improve airflow and oxygen delivery.
With a growing aging population and increasing prevalence of chronic respiratory conditions, there is a strong shift toward home-based care. Technologically, there is a strong shift toward home-based care. Technological advancements, including portable and connected devices, are making respiratory support more convenient, efficient, and patient-friendly, improving overall quality of life and long-lasting disease management.
Graying America: Tracking the Rise of the 65+ Population
| Year | Total (65+) | Male | Female |
| 2023 | 59.6 million | 26.9 million | 32.7 million |
| 2024 | 61.2 million | 27,952,606 | 33.2 million |
Lifelines for Lungs: Why Respiratory DME is Essential
Respiratory durable medical equipment (DME) is crucial in the U.S. for managing chronic breathing conditions such as COPD, asthma, and sleep apnea. These devices ensure a continuous oxygen supply, improve lung function, and reduce the risk of complications. They also enable patients to receive effective care at home, minimizing hospital visits and healthcare costs. With rising respiratory disease cases, an aging population plays a key role in enhancing patient comfort, independence, and overall quality of life.
Data Points Highlighting U.S. Respiratory Care Demand
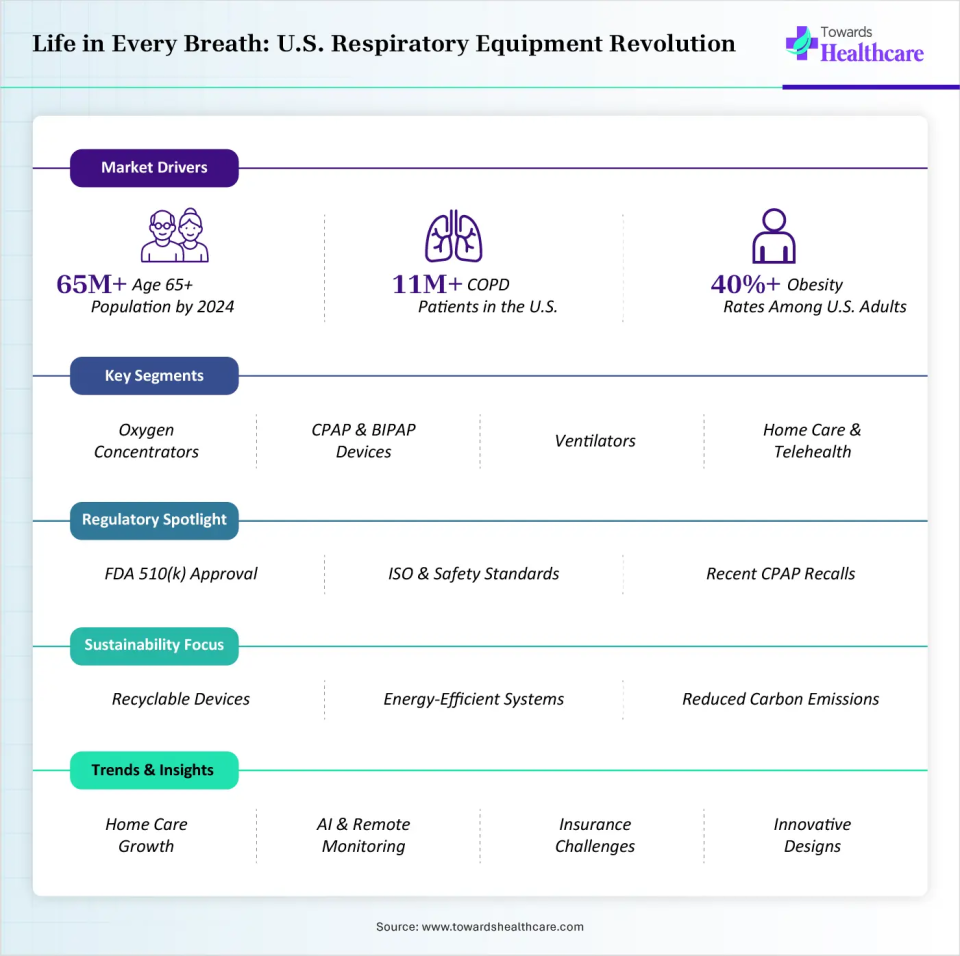
In the U.S., more than 11 million adults are diagnosed with COPD, while asthma affects nearly 25-27 million people, reflecting a significant patient base requiring respiratory support. Chronic lower respiratory diseases account for over 145000 deaths annually, making them a leading cause of mortality. Additionally, the 65+ population exceeds 60 million, increasing demand for long-term breathing assistance devices, especially in home-based care settings.
Breath of Growth: Powering the Rise of Respiratory DME
- Lifestyle-Linked Disorder: Rising obesity affects over 40% of U.S. adults, increasing the risk of sleep apnea and other breathing disorders.
- Post-Pandemic Awareness: Respiratory health monitoring has increased significantly after COVID-19, leading to higher adoption of oxygen therapy and monitoring devices.
- Insurance & Reimbursement Support: Medicare covers a large share of DME users, with millions of beneficiaries relying on respiratory equipment annually.
- Hospital-to-Home Transition: Nearly 80% of older adults prefer aging at home, accelerating demand for home-based respiratory solutions.
- Product Design Improvements: Modern devices are becoming 30-50% more compact and quieter, improving comfort and long-term adherence.
Breathing Barriers: The Roadblocks Slowing Respiratory DME Adoption
The high cost of advanced devices often restricts affordability for many patients, especially those with limited insurance coverage. Complex reimbursement procedures can delay access to essential equipment. In addition, patient non-compliance with therapies such as CPAP reduces treatment effectiveness. Regular maintenance and servicing requirements add to the burden for users. Furthermore, strict regulatory standards can slow down product approvals, creating challenges for timely innovation and wider availability of advanced respiratory solutions.
Regulatory Pulse: Navigating Compliance in U.S. Respiratory DME
Respiratory DME in the U.S. is regulated under Food and Drug Administration pathways, with most devices classified as Class II requiring 510(k) clearance. Manufacturers must comply with ISO standards and strict safety regulations. Recent recalls, including CPAP devices, have intensified scrutiny, pushing companies toward higher quality benchmarks, improved risk management, and stronger post-market surveillance systems.
Green Breaths: Sustainability Shaping Respiratory DME
Sustainability is becoming central to respiratory DME innovation, with manufacturers focusing on recyclable components and reduced medical waste. Energy-efficient oxygen systems lower electricity consumption and operating costs. The shift toward home-based care significantly reduces hospital-related carbon emissions, supporting greener healthcare delivery. These ESG-driven initiatives align with investor priorities while enhancing long-term environmental and economic value across respiratory care ecosystems.
Air of Innovation Shaping U.S. Respiratory Care
Connected Health Platforms
Respiratory devices are increasingly integrated with mobile apps and cloud platforms, allowing healthcare providers to track patient progress, monitor adherence, and intervene early if issues arise.
Telehealth Integration
Respiratory equipment is increasingly linked with telemedicine platforms, enabling remote monitoring, virtual consultations, and real-time data sharing between patients and providers.
Patient-Centric Design
Devices are now designed for greater comfort, usability, and portability, helping patients manage respiratory conditions more easily at home or on the go.
Advanced Non-Invasive Ventilation
New non-invasive ventilators and high-flow oxygen systems offer improved breathing support without hospitalisation, reducing patient discomfort and complications.
Government Breath of Life: Funding the Future of Respiratory Care
The U.S. government actively supports respiratory healthcare through funding, reimbursement programmes, and policy initiatives that improve access to durable medical equipment. Programmes like Medicare and Medicaid cover essential devices such as oxygen concentrators and CPAP machines, helping elderly and low-income patients afford treatment. Federal agencies also invest in research and technology integration, including National Heart, Lung, and Blood Institute (NHLBI) grants for home ventilation devices, FDA programmes for connected respiratory equipment, and the 2024 Department of Health and Human Services (HHS) initiative supporting telehealth-enabled respiratory care, advancing innovation, and improving patient outcomes.
Key Stats on U.S. Respiratory Care
| Parameter | Data/Value |
| Oxygen therapy users | 1,500,000 patients |
| Sleep apnea device users | 8,000,000 adults |
| Ventilator use | 300,000 patients |
| Portable oxygen concentration adoption growth | 15% increase |
| Hospital vs home device usage | 40% hospital/60% home |
| Medicare Coverage for respiratory DME | Cover 70-80% of eligible patients |
| Average daily usage (CPAP devices) | 6-7 hours/night |
| Patients with multiple respiratory conditions | 35% of COPD or asthma patients |
Air in Action: Major Application of U.S. Respiratory DME
- COPD management with oxygen concentration, ventilators, and nebulisers to maintain oxygen levels and improve lung function.
- Sleep apnea treatment using CPAP and BIPAP machines to ensure uninterrupted airflow, improve sleep quality, and reduce cardiovascular risks.
- Asthma control through inhalers, nebulisers, and medication delivery devices for effective symptom management and attack prevention.
- Home-based respiratory therapy with portable oxygen systems and compact ventilators to support independent living and reduce hospital visits.
- Post-surgical recovery assistance using respiratory equipment to maintain lung function and accelerate rehabilitation.
- Telehealth and remote monitoring via connected devices to track therapy adherence and adjust treatment plans in real time.
Major Investment Driving Respiratory DME Innovation
United States - Private & Public Funding for Connected Respiratory Tech
Large healthcare companies and startups are receiving venture capital and strategic funding to build AI‑enabled, connected respiratory devices and remote monitoring platforms. Investments also support scalable home‑healthcare solutions that integrate with telehealth networks.
- Major funding rounds for smart ventilator and digital oxygen therapy startups aimed at enhancing real‑time patient tracking and care.
Recent Breakthroughs: Innovation in U.S. respiratory Care
- In January 2025 � Smart CPAP Devices Launched: U.S. manufacturers introduced AI-enabled CPAP machines that adjust airflow in real time, enhancing therapy adherence and patient comfort.
- In August 2024 � Portable Oxygen Concentrator Upgrades: Lightweight, battery-powered concentrators with extended mobility support home and travel use, improving patient independence.
- In March 2025, Telehealth-Integrated Ventilators: Hospitals and homecare providers adopted ventilators capable of remote monitoring, enabling clinicians to adjust therapy without in-person visits.
- In June 2024 � Eco-Friendly Device Designs: New energy-efficient and quieter respiratory equipment reduces operational costs and environmental impact.
- In September 2025 � FDA Approvals for Advanced Devices: Clearance of next-generation ventilators and connected oxygen therapy devices accelerates adoption in home and clinical settings.
Leading U.S. Respiratory DME Companies: Key Offerings and Market Contributions
| Company | Key Offerings | U.S. Contributions | Strategic Focus |
| ResMed (U.S.) | CPAP, BiPAP, cloud-connected devices | Expands the digital sleep apnea ecosystem and remote monitoring adoption� | Homecare, SaaS platforms |
| Philips Respironics (U.S.) | CPAP, ventilators, oxygen systems | Major recall response and compliance restructuring in the U.S. market | Safety, regulatory alignment |
| Medtronic (U.S. operations) | Ventilators, Puritan Bennett systems | Strengthened critical care ventilation and portable solutions | Integrated respiratory care |
| GE HealthCare (U.S.) | ICU ventilators, monitoring systems | AI-enabled respiratory support in hospitals nationwide | Data-driven healthcare |
| Vyaire Medical (U.S.) | Ventilators, diagnostics | Supports U.S. hospitals with respiratory diagnostics and therapy tools | Clinical efficiency� |
| Invacare Corporation (U.S.) | Oxygen concentrators, homecare devices | Expands access to affordable home oxygen therapy nationwide | Accessibility, homecare |
| Teleflex Incorporated (U.S.) | Airway management, ventilation tools | Supplies respiratory critical care products across U.S. hospitals | Acute care focus |
| ICU Medical (U.S.) | Infusion, respiratory systems | Enhances respiratory support infrastructure in U.S. care settings | Integrated delivery |
| Masimo (U.S.) | Monitoring devices, sensors | Advances in non-invasive respiratory monitoring technologies | Patient monitoring |
| Lincare Holdings (U.S.) | Oxygen therapy, DME services | Serves over 1.8 million U.S. patients with home respiratory care | Homecare distribution |
Breathing Innovation: Unlocking Tomorrow�s Respiratory DME Potential
The U.S. respiratory durable medical equipment sector offers significant growth opportunities driven by rising chronic respiratory diseases, an aging population, and increased home-based care adoption. Emerging trends in smart, connected, and portable devices allow for remote patient monitoring and improved therapy adherence. Additionally, advancements in AI, telehealth integration, and personalized respiratory solutions create avenues for innovation, investment, and enhanced patient outcomes in both clinical and home settings.
Expert Insights
According to a recent report on the U.S. respiratory durable medical equipment (DME) market, published on Towards Healthcare. The market is rapidly evolving with a strong shift toward home-based care, digital integration, and patient-centric solutions. Experts highlight that rising chronic respiratory diseases and an aging population are accelerating long-term demand. At the same time, advancements in connected devices and AI-driven monitoring are improving adherence and clinical outcomes. However, reimbursement complexities and cost pressures remain key challenges. Industry leaders emphasize that companies investing in innovation, interoperability, and sustainable design will gain a competitive advantage in this expanding market landscape globally.
About the Experts

Aditi Shivarkar
Aditi leads as Vice President at Towards Healthcare and brings over 15 years of experience in healthcare research, innovation, and strategy. She works closely with data from across the healthcare sector and turns it into clear direction that companies can actually use. Her work covers pharmaceuticals, medical devices, and digital health. She helps businesses understand where the market is going and how to respond with confidence. Aditi focuses on practical thinking, strong decision-making, and delivering real results that make a difference.

Aman Singh
Aman Singh brings over 13 years of experience in healthcare research and consulting. He studies global healthcare trends and keeps a close eye on areas like biotech, AI in healthcare, and new treatment approaches. At Towards Healthcare, he leads the research team and makes sure the work stays accurate, useful, and easy to understand. Aman breaks down complex changes in the industry and helps businesses make smart, informed decisions.

Piyush Pawar
Piyush Pawar works as Senior Manager for Sales and Business Growth at Towards Healthcare, with more than 10 years of experience in the healthcare space. He works directly with clients and helps them find the right research for their needs. He makes sure clients understand the insights and know how to use them in their business. Piyush builds strong relationships and focuses on helping companies grow by turning research into clear, practical action.




 Request Consultation
Request Consultation


