The global breast biopsy devices market size accounted for USD 6.52 billion in 2025 and is predicted to increase from USD 7.03 billion in 2026 to approximately USD 13.82 billion by 2035, expanding at a CAGR of 7.8% from 2026 to 2035. Globally rising breast cancer cases, massive initiatives for cancer screening, and demand for minimally invasive methods are driving the overall market expansion.
Market Overview
The globe breast biopsy devices market covers advanced, specialized, minimally invasive medical devices, like fine-needles, core needles, & vacuum-assisted probes, that are employed to remove small tissue or fluid samples from the breast. These kinds of devices offer increased accuracy, conclusive & less invasive diagnoses, with minimal complications, like less scarring & rapid recovery time. They have a pivotal role in offering sufficient samples for pathologic, hormonal & genetic analysis.
Market Projection
In the coming era, large, well-developed companies will focus on fostering AI integration with imaging-guided platforms to facilitate real-time feedback & enhance diagnostic accuracy, with lower false-negative results. Including the UChicago Medicine Cancer Pavilion, a vast 575,000-square-foot facility, is scheduled to be inaugurated in Spring 2027, to execute AI for same-day diagnostic exams & biopsies. A little effort in the breast biopsy devices market is also impelling robotic-assisted biopsy systems to raise procedural accuracy.
Number of Breast Cancer New Cases & Deaths by 2050 (Approx. in Million)
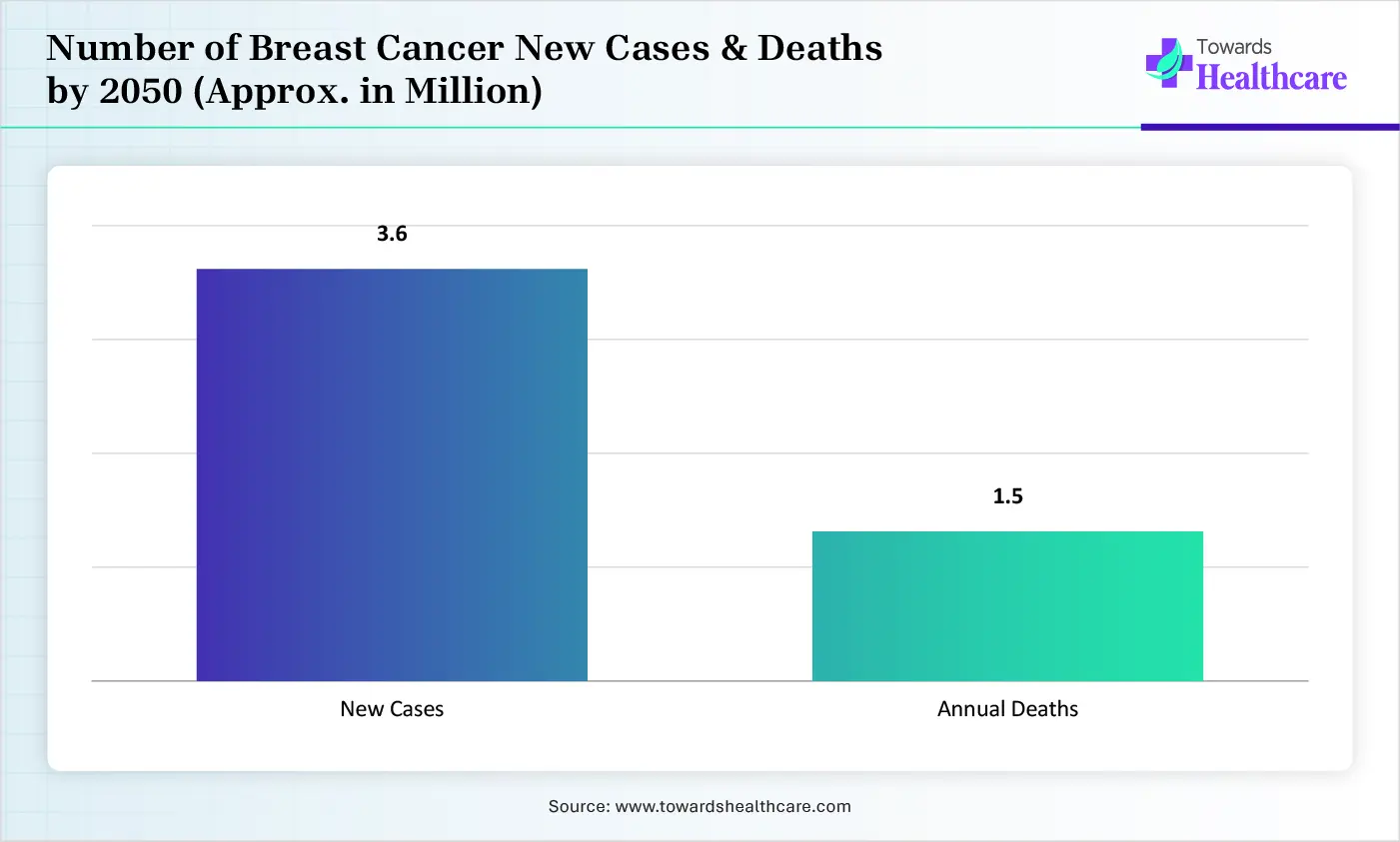
These forecast cases show the significance of biopsy devices, with the enforcement of screening programs, mainly in limited resource areas, to enable earlier & treatable cancer detection to mitigate life-threatening effects. Also, the globe will incorporate VABB & robotic systems to boost accuracy, comfort, & faster recovery period.
Market Segmentation Overview
rn- By product type, the needle-based biopsy instruments segment led with 58% share of the breast biopsy devices market in 2025. Major drivers are surging preference for core needle biopsy (CNB) & vacuum-assisted biopsy (VAB) due to affordability, lowered scarring, & expedited recovery. Sophisticated needles can often combine with ultrasound, MRI, or stereotactic imaging to bolster precision.
rn- By technique, the minimally invasive biopsy segment held 72% share in 2025 & is expected to grow at 8.6% CAGR. Dominance is driven by a rise in breast cancer cases, escalating demand for early detection through image-guided screenings & expansion of outpatient care. The market is increasingly leveraging high-frequency ultrasound, digital breast tomosynthesis (DBT) for 3D localization, & MRI to allow accurate targeting of non-palpable lesions.
rn- By imaging modality, the stereotactic X-ray segment dominated with 40% share of the breast biopsy devices market in 2025. They play a key role in the identification of small cancers & microcalcifications, often in pre-malignant stages. Also, they offer a broader range of histological accuracy, i.e., over 90%, as an option to open surgical biopsy.
rn- By end user, the hospitals segment captured a major share of 55% in 2025, due to the surging need for outpatient settings. Along with rigorous reimbursement insurance, hospitals facilitate comprehensive, immediate care, especially imaging, pathology, & surgery, often with trained specialists. Also, they widely invest in advanced diagnostic technology, like AI-enhanced systems, & innovative imaging modalities.
rn- By application, the breast cancer diagnosis segment led with 88% share of the breast biopsy devices market in 2025. Accelerating breast cancer instances are fueling demand for early diagnosis, minimally invasive procedures, & adoption of advanced technologies. Government-led & private screening programmes are demanding the need for pathological confirmation of suspected malignancies found through imaging or physical examination.
rn
Regional Analysis
North America held a dominant share of 38% of the market in 2025, due to the possession of highly advanced healthcare infrastructure & ongoing demand for less invasive diagnosis approaches. Besides this, incentives, such as the SCREENS for Cancer Act of 2025 in the U.S., are facilitating diagnostic services to underserved areas, while in Canada, government-funded screening programs are propelling persistent demand for these devices.
Asia Pacific captured 22% share of the breast biopsy devices market in 2025 & is predicted to witness rapid growth at 9.20% CAGR during 2026-2035. Prominent drivers are the escalating breast cancer incidences, with impressive government-funded screening programmes, especially in China & India. In recent days, the Fudan University Shanghai Cancer Center explored a portable AI-assisted ultrasound, trained on 300,000 instances, which allows efficient screening, specifically for high-risk lesions, i.e., BI-RADS 4A+.
Top Companies in the Breast Biopsy Devices Market
Hologic, Inc., Becton, Dickinson and Company (BD), Devicor Medical Products, Inc., Argon Medical Devices, Inc. are 4 key players of the market. From these, Hologic, Inc., perceives market leadership, with an emphasis on consistent offering of comprehensive breast lesion localization products, fueling both organic growth & strategic acquisitions to expand its technology portfolio.
Segments Covered in the Report
By Product Type
rn- Needle-Based Biopsy Instrumentsrn
rn- Core Needle Biopsy (CNB) Devices
rn- Fine Needle Aspiration (FNA) Devices
rn- Vacuum-Assisted Biopsy (VAB) Devices
rn
rn rn- Localization Wires
rn- Biopsy Tables
rn- Guidance Systemsrn
rn- Stereotactic Guidance Systems
rn- Ultrasound-Guided Systems
rn- MRI-Guided Systems
rn
rn rn
By Technique
rn- Minimally Invasive Biopsyrn
rn- Core Needle Biopsy
rn- Vacuum-Assisted Biopsy
rn- Fine Needle Aspiration
rn
rn rn- Surgical Biopsyrn
rn- Incisional Biopsy
rn- Excisional Biopsy
rn
rn rn
By Imaging Modality
rn- Stereotactic X-ray
rn- Ultrasound
rn- MRI
rn
By End User
rn- Hospitals
rn- Diagnostic Imaging Centers
rn- Ambulatory Surgical Centers
rn
By Application
rn- Breast Cancer Diagnosis
rn- Research & Clinical Trials
rn
By Region
rn- North Americarn
rn- U.S.
rn- Canada
rn- Mexico
rn- Rest of North America
rn
rn rn- Latin Americarn
rn- Brazil
rn- Argentina
rn- Rest of Latin America
rn
rn rn- Europe rn
rn- Western Europe rn
rn- Germany
rn- Italy
rn- France
rn- Netherlands
rn- Spain
rn- Portugal
rn- Belgium
rn- Ireland
rn- UK
rn- Iceland
rn- Switzerland
rn- Poland
rn- Rest of Western Europe
rn
rn rn- Eastern Europe rn
rn- Austria
rn- Russia & Belarus
rn- Türkiye
rn- Albania
rn- Rest of Eastern Europe
rn
rn rn
rn rn- Asia Pacificrn
rn- China
rn- Taiwan
rn- India
rn- Japan
rn- Australia and New Zealand
rn- ASEAN Countries (Singapore, Malaysia)
rn- South Korea
rn- Rest of APAC
rn
rn rn- MEA rn
rn- GCC Countriesrn
rn- Saudi Arabia
rn- United Arab Emirates (UAE)
rn- Qatar
rn- Kuwait
rn- Oman
rn- Bahrain
rn
rn rn- South Africa
rn- Egypt
rn- Rest of MEA
rn
rn rn

