Which Top Companies Play Vital Roles in the Hyperkalemia Drugs Market
- AstraZeneca plc
- CSL Vifor (Vifor Pharma AG)
- Sanofi
- Kowa Company, Ltd.
- KVK-Tech, Inc.
- Fresenius Kabi AG
- Novo Nordisk A/S
What are Hyperkalemia Drugs?
Hyperkalemia drugs are medications designed to reduce high potassium levels in the blood and prevent related cardiac complications. The hyperkalemia drugs market is growing due to the rising prevalence of chronic kidney disease, heart failure, and diabetes, all of which increase the risk of elevated potassium levels. Greater use of RAAS inhibitors in cardiovascular treatment is also driving demand for effective potassium-lowering therapies. Additionally, improved clinical awareness, advancements in novel potassium binders, and expanding access to outpatient management options are supporting wider adoption and market growth.
For Instance,
- In September 2025, AstraZeneca Pharma India gained approval from the Subject Expert Committee under CDSCO to begin a Phase IV trial of Sodium Zirconium Cyclosilicate (Lokelma) in India. The study will further evaluate the drug’s effectiveness and safety for treating hyperkalemia in real-world patients.
Market Growth
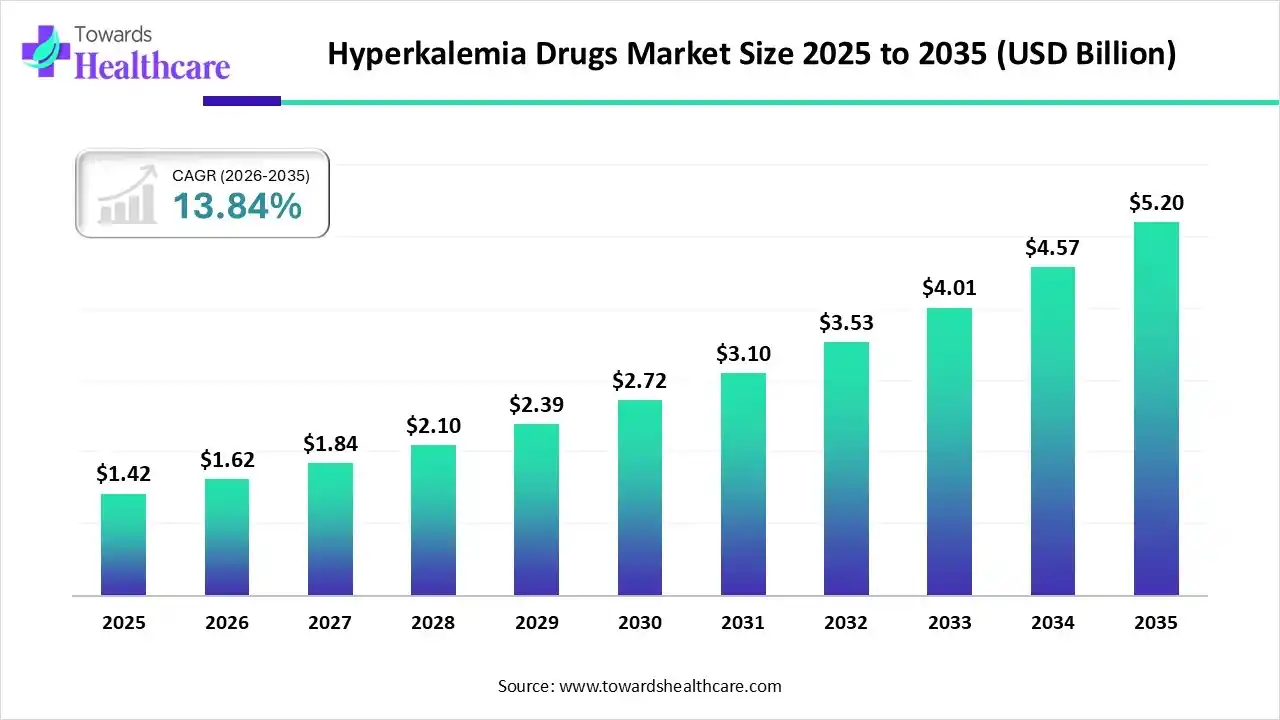
The hyperkalemia drugs market size was reported at US$ 1.42 billion in 2025 and is expected to rise to US$ 1.62 billion in 2026. According to forecasts, it will grow at a CAGR of 13.84% to reach US$ 5.20 billion by 2035.
Hyperkalemia Drugs Market Outlook
- Sustainability Trends: Sustainability trends in the market include a shift toward eco-efficient manufacturing, reduced waste in production, and increased use of recyclable packaging. Companies are also adopting greener supply chains and investing in energy-efficient facilities to lower environmental impact.
- Global Expansion: Global expansion of the hyperkalemia drugs market is driven by rising CKD and cardiovascular cases worldwide, broader access to advanced potassium binders, and increasing regulatory approvals across emerging regions. Growing investments by pharma companies further strengthen international market presence.
- Startup Ecosystems: The startup ecosystem in the hyperkalemia drugs market is supported by emerging biotech firms developing novel potassium binders, AI-driven diagnostic tools, and patient-monitoring platforms. Collaborations with research institutes and funding from venture capital accelerate innovation and early-stage clinical development.
Company and Its Offerings in Hyperkalemia
Sanofi
-
Produces Kayexalate (sodium polystyrene sulfonate).
-
A classic cation-exchange resin used for chronic hyperkalemia.
Kowa Company, Ltd.
-
No specific hyperkalemia potassium binder publicly reported.
-
Plays a minor or no role in this therapeutic area.
KVK-Tech, Inc.
-
Manufactures sodium polystyrene sulfonate (marketed as Kalexate / SPS).
-
Used for oral or rectal treatment of hyperkalemia.
Fresenius Kabi AG
-
Partners via VFMCRP to introduce Veltassa (patiromer) to China.
-
Supports renal patients in maintaining RAASi therapy.
Novo Nordisk A/S
-
No marketed hyperkalemia binder.
-
Previously linked to ocedurenone (KBP-5074), an MR antagonist in CKD.
-
Not a direct potassium binder.
Company Landscape
AstraZeneca plc (Key Drug: Lokelma)
Company Overview:
- AstraZeneca is a global, science-led biopharmaceutical company focused on the discovery, development, and commercialization of prescription medicines.
- It operates across three key therapy areas: Oncology, BioPharmaceuticals (Cardiovascular, Renal and Metabolism, and Respiratory & Immunology), and Rare Disease (via Alexion, AstraZeneca Rare Disease).
Corporate Information (Headquarters, Year Founded, Ownership Type):
- Headquarters: Cambridge, United Kingdom
- Year Founded: 1999 (formed by the merger of Astra AB of Sweden and Zeneca Group PLC of the UK)
- Ownership Type: Public (LSE: AZN, NASDAQ: AZN)
History and Background:
- Formed in 1999, the company has grown through significant R&D investment and strategic acquisitions.
- Its hyperkalemia drug, Lokelma (sodium zirconium cyclosilicate), was originally developed by ZS Pharma, which AstraZeneca acquired in 2015 for approximately $2.7 billion.
Key Milestones/Timeline:
- 2015: Acquired ZS Pharma for the novel potassium binder, sodium zirconium cyclosilicate (Lokelma).
- 2018: Lokelma approved in the US (FDA) and EU (EMA) for the treatment of hyperkalemia in adults.
- 2024: Announced a new co-marketing agreement in India for Lokelma with Sun Pharmaceutical Industries.
Business Overview:
- Focuses on specialty medicines in Oncology, Rare Diseases, and BioPharmaceuticals (CVRM and R&I).
- Hyperkalemia drug Lokelma is a key product within the Cardiovascular, Renal and Metabolism (CVRM) franchise.
Business Segments/Divisions:
- Oncology
- BioPharmaceuticals (Cardiovascular, Renal and Metabolism [CVRM], Respiratory & Immunology [R&I])
- Rare Disease (Alexion)
Geographic Presence:
- Global operations, with major markets in the US, Europe, China, and Emerging Markets.
- Lokelma is approved and commercialized in over 50 countries globally.
Key Offerings:
- Lokelma (sodium zirconium cyclosilicate): Oral suspension for the treatment of hyperkalemia.
- Major brands include Tagrisso, Imfinzi, Farxiga/Forxiga, and others.
End-Use Industries Served:
- Pharmaceutical/Healthcare
- Specific therapeutic areas: Cardiovascular, Renal, Metabolic, Oncology, Rare Disease, Respiratory, and Immunology.
Key Developments and Strategic Initiatives:
Mergers & Acquisitions:
No major M&A specific to hyperkalemia drugs in 2024/2025.
Partnerships & Collaborations:
2024: Entered a "second brand partnership" with Sun Pharmaceutical Industries in India to co-market Lokelma, aiming for broader patient access.
Product Launches/Innovations:
Focus on post-marketing studies to expand Lokelma's clinical data in real-world settings and specific patient populations.
Capacity Expansions/Investments:
Ongoing investment in global manufacturing and supply chain to meet increasing demand for CVRM products.
Regulatory Approvals:
No major new drug approvals for hyperkalemia in 2024/2025; focus is on geographic expansion and label updates for Lokelma.
Distribution channel strategy:
- Uses a mix of specialty pharmacy, hospital distribution, and retail pharmacy, tailored by region.
- Focus on driving adoption in chronic care settings for patients with Chronic Kidney Disease (CKD) and Heart Failure (HF).
Technological Capabilities/R&D Focus:
- Core Technologies/Patents: Lokelma (sodium zirconium cyclosilicate) is a highly selective inorganic, non-polymer potassium binder.
- Research & Development Infrastructure: Extensive R&D centers globally (e.g., Cambridge, Gaithersburg, Gothenburg).
- Innovation Focus Areas: Cardiorenal space, focusing on managing comorbidities like hyperkalemia to enable optimal use of life-saving RAAS inhibitor therapy.
Competitive Positioning:
- Strengths & Differentiators: Strong global commercial infrastructure; Lokelma's rapid onset of action (median $2.2$ hours); extensive clinical trial data supporting chronic use.
- Market presence & ecosystem role: A dominant leader in the novel oral potassium binder segment; plays a crucial role in enabling guideline-directed medical therapy for CKD and HF patients.
SWOT Analysis:
- Strengths: Patented, best-in-class novel potassium binder (Lokelma); Global commercial footprint; Strong financial backing; Focus on CVRM.
- Weaknesses: High price point compared to older resins; Competition from Veltassa (Patiromer).
- Opportunities: Penetration into developing markets (e.g., India co-marketing); Expanding use in managing RAASi therapy in HF/CKD patients.
- Threats: Patent expiry; Development of next-generation hyperkalemia treatments; Pressure on drug pricing/reimbursement.
Recent News and Updates:
Press Releases:
- November 2025: Announcement of a new co-marketing deal for Lokelma with Sun Pharmaceutical Industries in India.
- Industry Recognitions/Awards:
- Recognized as a global leader in CVRM/Specialty care in various industry reports (general corporate recognition).
CSL Vifor (Key Drug: Veltassa)
Company Overview:
- CSL Vifor is a business unit of CSL Limited, specializing in treatments for kidney disease, iron deficiency, and cardiorenal conditions.
- It was formed following CSL Limited's acquisition of Vifor Pharma in August 2022.
Corporate Information (Headquarters, Year Founded, Ownership Type):
- Headquarters: St. Gallen, Switzerland (CSL Vifor business unit)
- Year Founded: Vifor Pharma was founded in 1999; Acquired by CSL Limited in 2022.
- Ownership Type: Subsidiary of a Public Company (CSL Limited: ASX: CSL)
History and Background:
- Vifor Pharma was historically focused on nephrology and cardio-renal treatments.
- Veltassa (patiromer) was originally developed by Relypsa, which was acquired by Galenica/Vifor Pharma.
- The hyperkalemia business became part of the CSL Vifor division after CSL Limited completed its acquisition of Vifor Pharma in August 2022 for approximately $$11.7$ billion.
Key Milestones/Timeline:
- 2015: Veltassa (patiromer) approved by the US FDA for the treatment of hyperkalemia.
- 2022: CSL Limited completes the acquisition of Vifor Pharma, establishing CSL Vifor and integrating Veltassa into its portfolio.
- 2024: Continued focus on global expansion and leveraging the CSL's larger infrastructure for Veltassa commercialization.
Business Overview:
- CSL Vifor is dedicated to patient blood management, kidney disease, and iron deficiency treatment.
- Veltassa is a flagship product in its cardiorenal portfolio, focusing on the chronic management of hyperkalemia.
Business Segments/Divisions:
- CSL Vifor operates as a key business unit within CSL Limited, alongside CSL Behring and CSL Seqirus.
- Focuses on Nephrology and Cardiorenal Therapy (Veltassa is a key component).
Geographic Presence:
- Major presence in the US and Europe; expanding reach into Asia Pacific and other global markets under the CSL umbrella.
- Veltassa is approved and marketed in numerous countries, including the US, EU, and Australia.
Key Offerings:
- Veltassa (patiromer): Oral suspension powder for the treatment of hyperkalemia.
- Other key products include Ferinject/Injectafer (iron deficiency) and various nephrology products.
End-Use Industries Served:
- Pharmaceutical/Healthcare
- Specific therapeutic areas: Nephrology, Cardiology, and Primary Care settings treating CKD and HF patients.
Key Developments and Strategic Initiatives:
Mergers & Acquisitions:
The 2022 acquisition by CSL Limited is the most significant event, integrating Vifor's hyperkalemia expertise into the larger CSL framework.
Partnerships & Collaborations:
- Leveraging CSL's established global network for greater commercial reach for Veltassa.
- Ongoing collaborations with key opinion leaders and medical societies to reinforce Veltassa's role in chronic hyperkalemia management.
Product Launches/Innovations:
Focus on data generation from real-world evidence and post-marketing studies to support Veltassa's chronic use and RAASi enabling potential.
Capacity Expansions/Investments:
Investment in manufacturing and supply chain optimization under CSL ownership.
Regulatory Approvals:
Gaining new country-specific approvals for Veltassa's use in chronic hyperkalemia.
Distribution channel strategy:
- Strong focus on specialty pharmacies and patient support programs to manage access due to its chronic, take-at-home nature.
- Utilizes a dedicated sales force targeting nephrologists and cardiologists.
Technological Capabilities/R&D Focus:
- Core Technologies/Patents: Veltassa (patiromer) is a non-absorbed, non-polymer, inorganic potassium binder.
- Research & Development Infrastructure: Leveraging the combined R&D resources and clinical expertise of the CSL Group, particularly in cardiorenal space.
- Innovation Focus Areas: Optimization of dosing and administration; generating long-term outcome data in high-risk patient groups like those on RAAS inhibitors for HF.
Competitive Positioning:
- Strengths & Differentiators: Proven efficacy for chronic control; favorable safety profile; benefit of being taken with food (unlike older resins).
- Market presence & ecosystem role: A co-leader in the novel oral potassium binder market; essential provider in the nephrology and cardiorenal treatment ecosystem.
SWOT Analysis:
- Strengths: Global resources and stability from parent company CSL; Established efficacy for chronic hyperkalemia; Strong position in nephrology care.
- Weaknesses: Pricing pressure in some markets; Requires an interval before/after taking other oral medications.
- Opportunities: Integration into CSL's patient blood management and iron deficiency portfolio; Increased adoption in newly diagnosed CKD/HF patients.
- Threats: Aggressive competition from Lokelma; Potential for new, faster-acting therapies; Reimbursement challenges.
Recent News and Updates:
Press Releases:
CSL Limited's financial updates often highlight the strong performance of Veltassa as a growth driver in the CSL Vifor segment (2024/2025 financial reports).
Industry Recognitions/Awards:
None specifically highlighted in 2024/2025 public releases for Veltassa.
Hyperkalemia Drugs Market Value Chain Analysis
R&D
- R&D is increasingly focused on next-generation oral potassium binders designed to deliver higher efficacy, quicker potassium reduction, and improved safety compared to traditional resin therapies.
- The rise in CKD, heart failure, and RAASi-treated patients is creating strong demand for safer long-term hyperkalemia solutions, pushing companies to accelerate innovation.
- New formulations aim to reduce gastrointestinal issues, enhance tolerability, and support chronic use, improving patient adherence and overall outcomes.
Key Players: AstraZeneca, CSL Vifor, Ardelyx, ZS Pharma, Cipla
Clinical Trials
- Clinical trials studies are increasingly centered on advanced potassium binders such as patiromer and sodium zirconium cyclosilicate, assessing how well they perform against older options like sodium polystyrene sulfonate.
- Trials are evaluating their speed in reducing high potassium, ability to keep levels stable long-term, and overall safety, particularly in patients with CKD, heart failure, and diabetes.
- Research also compares tolerability, side-effect profiles, and suitability for chronic use to support better treatment decisions.
Key Players: AstraZeneca, CSL Vifor, Ardelyx, ZS Pharma, Cipla, Dr. Reddy’s Laboratories.
Distribution to Hospitals, Pharmacies
- Hospital pharmacies mainly manage urgent hyperkalemia cases, supplying fast-acting treatments needed in emergency and inpatient settings.
- Retail pharmacies support ongoing outpatient care, offering easy access to oral potassium binders for patients requiring routine management.
- Online pharmacies are becoming important for long-term therapy, providing convenient home delivery and refill options for chronic users.
Key Players: AstraZeneca, CSL Vifor, Ardelyx, ZS Pharma, Cipla, Dr. Reddy’s Laboratories.
Recent Developments in the Hyperkalemia Drugs Market
- In October 2025, AstraZeneca initiated a Phase IV clinical trial in India to assess the long-term safety and efficacy of sodium zirconium cyclosilicate in treating hyperkalemia in patients with chronic kidney disease and heart failure. This study aims to provide real-world data on the drug’s performance in these patient groups.
- In September 2025, AstraZeneca submitted the Phase IV clinical trial protocol for sodium zirconium cyclosilicate to the CDSCO's Subject Expert Committee, which closely evaluated the study’s design, safety plans, and statistical methods before approving the trial to move forward.
Partner with our experts to explore the Hyperkalemia Drugs Market at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
