April 2026

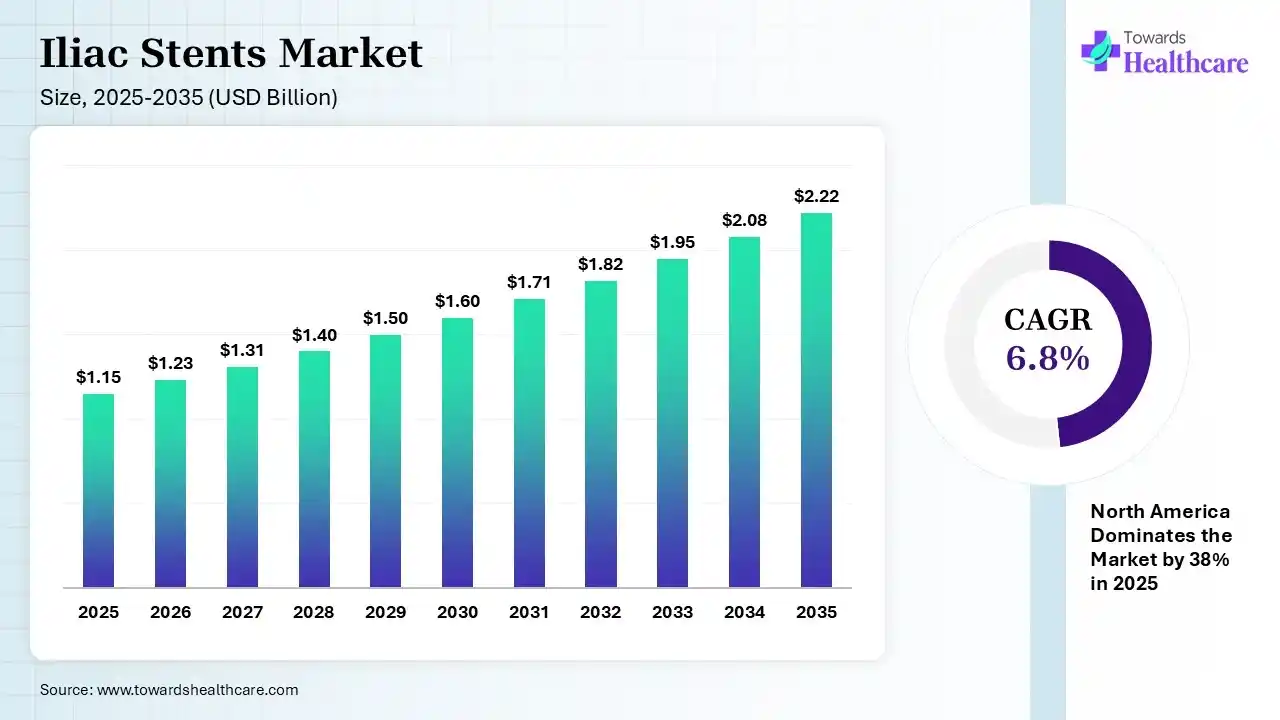
The global iliac stents market size was estimated at USD 1.15 billion in 2025 and is predicted to increase from USD 1.23 billion in 2026 to approximately USD 2.22 billion by 2035, expanding at a CAGR of 6.8% from 2026 to 2035. The iliac stents market is growing because of rising demand for stents to manage diseases such as those affecting the heart and blood vessels of ageing and middle-aged people.
The iliac stents market is growing, as an iliac stent is a small wire mesh tube that is used to hold open an iliac artery that has been narrowed by atherosclerotic disease. In an iliac stenting technology, a thin guide wire is inserted into a small incision in the upper thigh. This technology is generally visualized using X-rays and a special dye that reveals the arteries. It is a minimally invasive technology to place a small metal mesh tube in the affected vein to open it actively and restore normal blood flow. Stenting opens up the blocked vein, letting blood flow freely and lowering symptoms like pain and swelling. Iliac vein stenting is a safe and efficient treatment choice for a person with iliac vein compression syndrome.
AI-based algorithms in iliac stents can accurately identify vital anatomical features, forecast complications, and optimize stent placement by analyzing data from imaging techniques such as intravascular ultrasound and optical coherence tomography. Fully leverage AI to improve the effectiveness and safety of interventional procedures. AI's abilities enable precise identification of anatomical details, complication prediction, and optimal stent positioning, improving procedural results.
| Table | Scope |
| Market Size in 2026 | USD 1.23 Billion |
| Projected Market Size in 2035 | USD 2.22 Billion |
| CAGR (2026 - 2035) | 6.8% |
| Leading Region | North America by 38% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product Type, By Material, By Indication, By End User, By Region |
| Top Key Players | Medtronic, Abbott Laboratories, Boston Scientific Corporation, Becton Dickinson, W. L. Gore & Associates, Inc. |
| Segment | Share 2025 (%) |
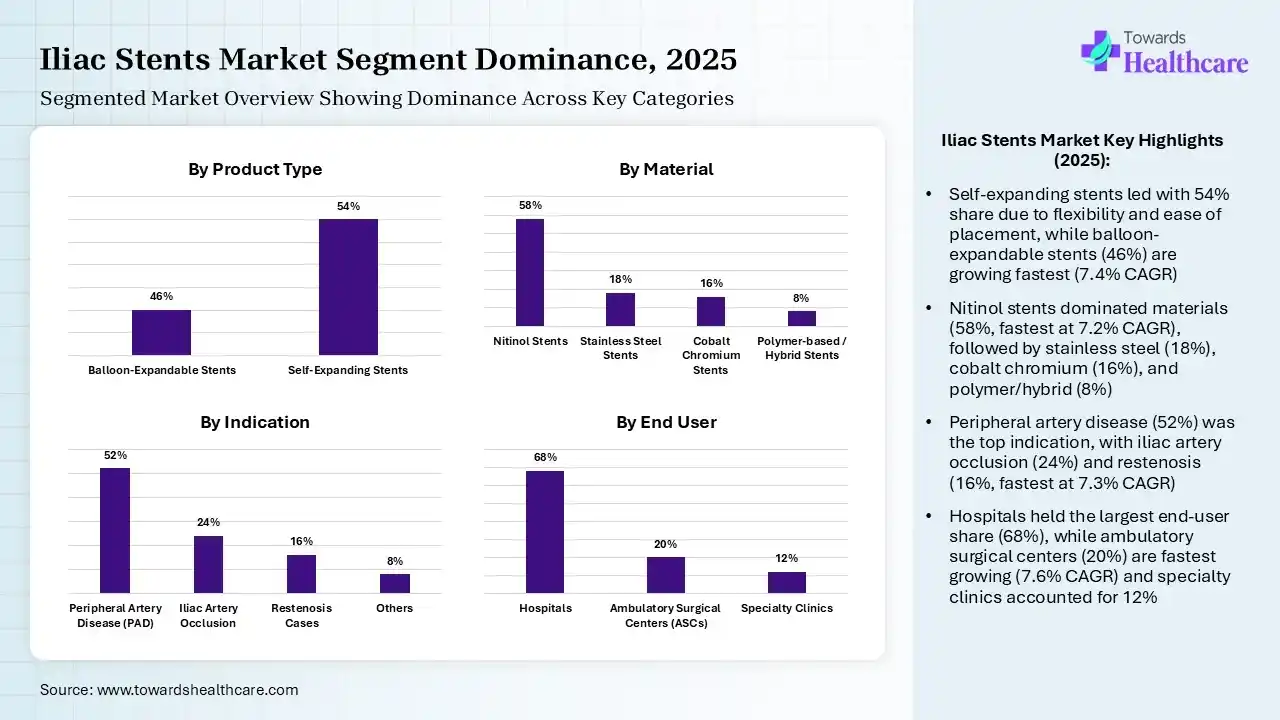
| Balloon-Expandable Stents | 46% |
| Self-Expanding Stents | 54% |
Self-Expanding Stents Segment Led the Iliac Stents Market in 2025
The self-expanding stents segment contributed the largest market share of 54% in 2025, as their design enables a high degree of elasticity, relative simplicity of placement, and smaller-diameter delivery technology for large-diameter stents. A self-expandable stent allows a larger-sized stent to be used with a small sheath. They are familiar with the natural curving of blood vessels, lowering the challenges of vessel wall injury.
The balloon-expandable stents segment held the second largest share of 46%, expected to grow at the fastest CAGR of 7.4% of the market, as balloon-expandable coronary stenting has transformed the field of interventional cardiology into a potential, negligibly invasive modality for managing coronary artery disease. It is less shocking to the patient and should carry a lower risk of dissection or vessel rupture.
| Segment | Share 2025 (%) |
| Nitinol Stents | 58% |
| Stainless Steel Stents | 18% |
| Cobalt Chromium Stents | 16% |
| Polymer-based / Hybrid Stents | 8% |
Nitinol Stents Segment Led the Iliac Stents Market in 2025
The nitinol stents segment contributed the largest market share of 58%, and it is the fastest growing in the forecast period with a 7.2% CAGR, as the stent avoids negative remodelling and provides distinct benefits over bare-stenting long SFA lesions. It reduces fracture and mechanical failure challenges. Nitinol is biocompatible; it is not toxic or harmful to living tissue. These characteristics make it a standard material for medical implants.
The stainless-steel stents segment held a significant share of 18% the market in 2025. Stainless steel is a common material because it combines processability, durability, corrosion resistance, and biocompatibility. These stents play a significant role in restoring blood flow, stabilizing vessels, and maintaining long-term vascular health.
The cobalt chromium stents segment held a significant share of the iliac stents market of 16% in 2025. Cobalt-chromium stents have condensed the restenosis rate. Cobalt–chromium stents are evolving as this material is stronger than 316 stainless steel, therefore enabling the construction of stents with thinner struts without compromising radial strength.
The polymer-based/hybrid stents segment held a significant share of 8% the market in 2025, as polymer stents offer advanced design, quality healing, and outstanding safety. They are made from polymers or metallic alloys and are designed to prevent restenosis and encourage normal vessel function throughout their absorption period.
| Segment | Share 2025 (%) |
| Peripheral Artery Disease (PAD) | 52% |
| Iliac Artery Occlusion | 24% |
| Restenosis Cases | 16% |
| Others | 8% |
The Conventional Dialysis Technology Segment held the Largest Share in the Market in 2025
The peripheral artery disease (PAD) segment contributed the largest iliac stents market share of 52% in 2025, as stenting is a significant procedure that can significantly improve blood flow and enhance the quality of life for patients with peripheral artery disease. Stenting evades the challenges of early elastic recoil, residual stenosis, and flow-limiting dissection after balloon angioplasty.
The iliac artery occlusion segment held a notable share of the market of 24% in 2025, as stent placement for iliac artery occlusion lowers major procedural complications compared with PTA. It's improved blood flow to the legs via the artery being treated.
The restenosis cases segment held a notable share of 16% the iliac stents market in 2025, expected to grow at the fastest CAGR of 7.3% of the market, as repeat stenting with enviroximes-eluting stents presented a statistical advantage over other methods in the primary results, binary restenosis rates, and target lesion revascularization. Drug-eluting stents have transformed the practice of interventional cardiology. They are safe and efficient in dramatically enhancing patient results.
| Segment | Share 2025 (%) |
| Hospitals | 68% |
| Ambulatory Surgical Centers (ASCs) | 20% |
| Specialty Clinics | 12% |
The Hospitals Segment held the Largest Share in the Market in 2025
The hospitals segment contributed the largest iliac stents market share of 68% in 2025, as iliac venous stenting provides long-term relief from CPP even with residual OVR and post-stent pregnancy. With 73% of women having full pain resolution. Stenting supports alleviating painful symptoms caused by iliac vein compression, like heaviness, swelling, achiness, discoloration, and restless legs.
The ambulatory surgical centers (ASCs) segment held the second largest share of 20%, expected to grow at the fastest CAGR of 7.6% of the market, as stenting for iliac vein outflow obstruction is related to excellent long-term stent patency and symptom resolution. Endovascular stenting enables same-day discharge, removing the requirement for long hospital stays related to open surgical reconstruction.
The specialty clinics segment held a notable share of 12% the market in 2025, as this is a minimally invasive, outpatient alternative to traditional open surgery. Patients frequently report a noteworthy advancement in QoL, involving better sleep, lower pain, and augmented mobility.
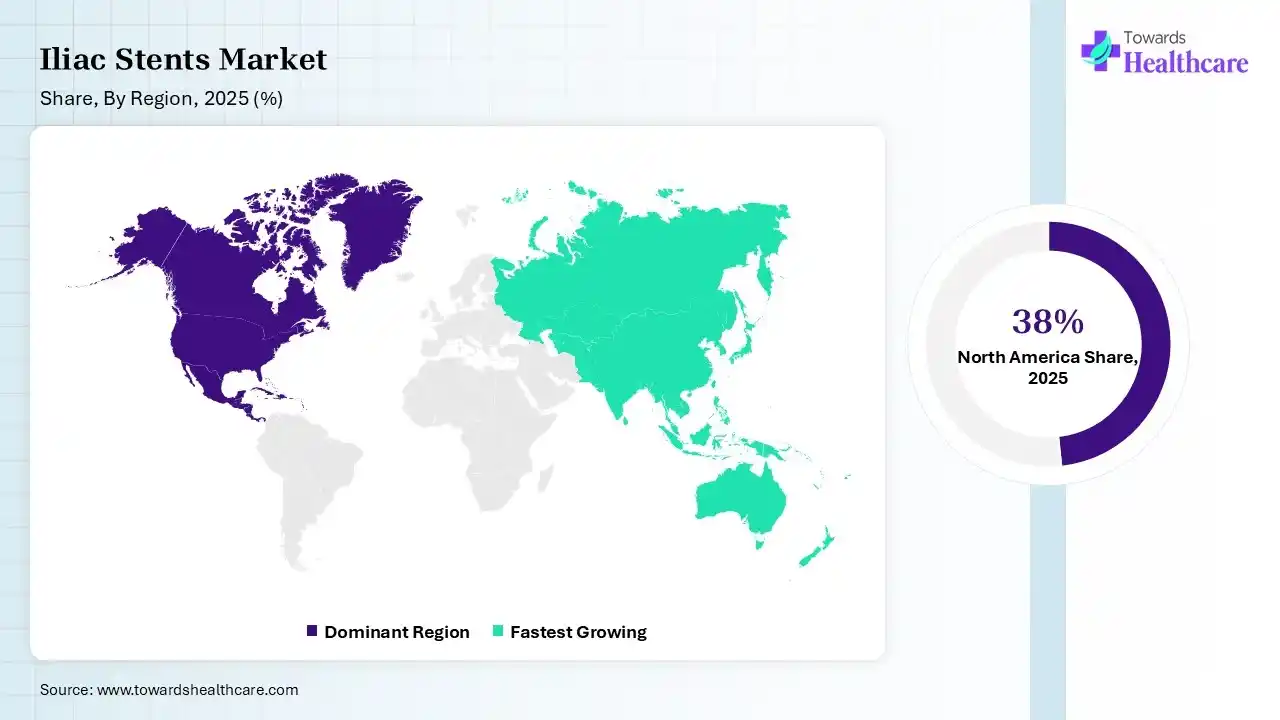
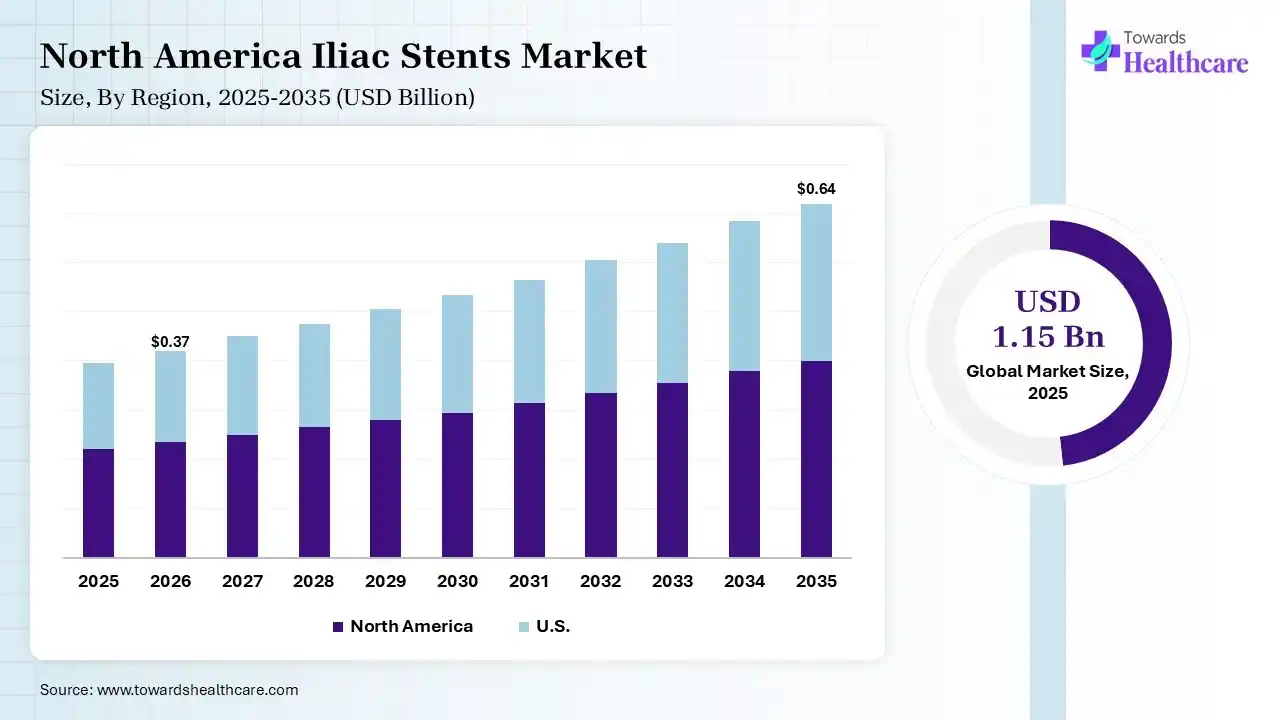
In 2025, North America dominated the iliac stents market with a share of 38% in 2025, because of the advancement in R-PCI with a focus on developments in robotic expertise, procedural difficulty, tele-stenting, and training processes. Significant industry players like Medtronic, Abbott, and W. L. Gore & Associates have strong production and research presences in the region.
For Instance,
The U.S. has a population of more than 330 million people and is supported by one of the most challenging healthcare systems in the world. This region has well-developed industrial countries. The region has a massive incidence of PAD, diabetes, and obesity, increasing demand for advanced iliac stents.
Asia Pacific held 22% share of the market. expected to experience the fastest growth with 8.2% CAGR during the forecast period, the rising CVD epidemics in this region are the consequence of challenging effects from interrelated changes in socioeconomics and living environments, demography, lifestyles, the prevalence of CVD risk factors, and the increasing demand for iliac stents. The quickly aging population in the Asia-Pacific region has the potential to create a severe health crisis, which contributes to the growth of the market.
India Market Trends
Peripheral arterial disease (PAD) is one of the underdiagnosed and undertreated vascular diseases despite its noteworthy burden in India. In India, the etiological aspects, diagnostic strategies, treatment modalities, and other preventive measures may diverge in different regions. Government hospitals often opt for domestic stents because of their affordability.
R&D:
Manufacturing Processes:
Patient Services:

| Company | Headquarters | Latest Update |
| Medtronic | Ireland | Medtronic has identified that a small subset of Abre stent lots contain stents with potential for a smaller-than-expected stent diameter due to a variation in Nitinol material properties. |
| Abbott Laboratories | United States | In September 2025, Abbott announced that Health Canada had authorized the Esprit BTK Everolimus Eluting Resorbable Scaffold System, a first-of-its-kind treatment innovation for people with chronic limb-threatening ischemia (CLTI) below-the-knee (BTK). |
| Boston Scientific Corporation | United States | In March 2025, Boston Scientific Corporation announced that the U.S. Food and Drug Administration (FDA) had approved its Express LD Iliac Premounted Stent System for use in iliac arteries. |
| Becton Dickinson | United States | In November 2025, BD announced full enrollment of the iliac artery patient cohort in its essential AGILITY investigational device exemption (IDE) research, which is assessing the Revello vascular covered stent for the treatment of peripheral artery disease (PAD). |
| W. L. Gore & Associates, Inc. | United States | In January 2026, W. L. Gore & Associates’ medical business (Gore) announced the FDA approval of the GORE VIABAHN FORTEGRA Venous Stent, previously known as the GORE VIAFORT Vascular Stent. |
Strengths
Weakness
Opportunities
Threat
By Product Type
By Material
By Indication
By End User
By Region
April 2026
April 2026
April 2026
April 2026