
The Latin America central lab market size reached US$ 289.89 million in 2025 and is anticipate to increase to US$ 322.51 million in 2026. By 2035, the market is forecasted to achieve a value of around US$ 773.25 million, growing at a CAGR of 10.20%.
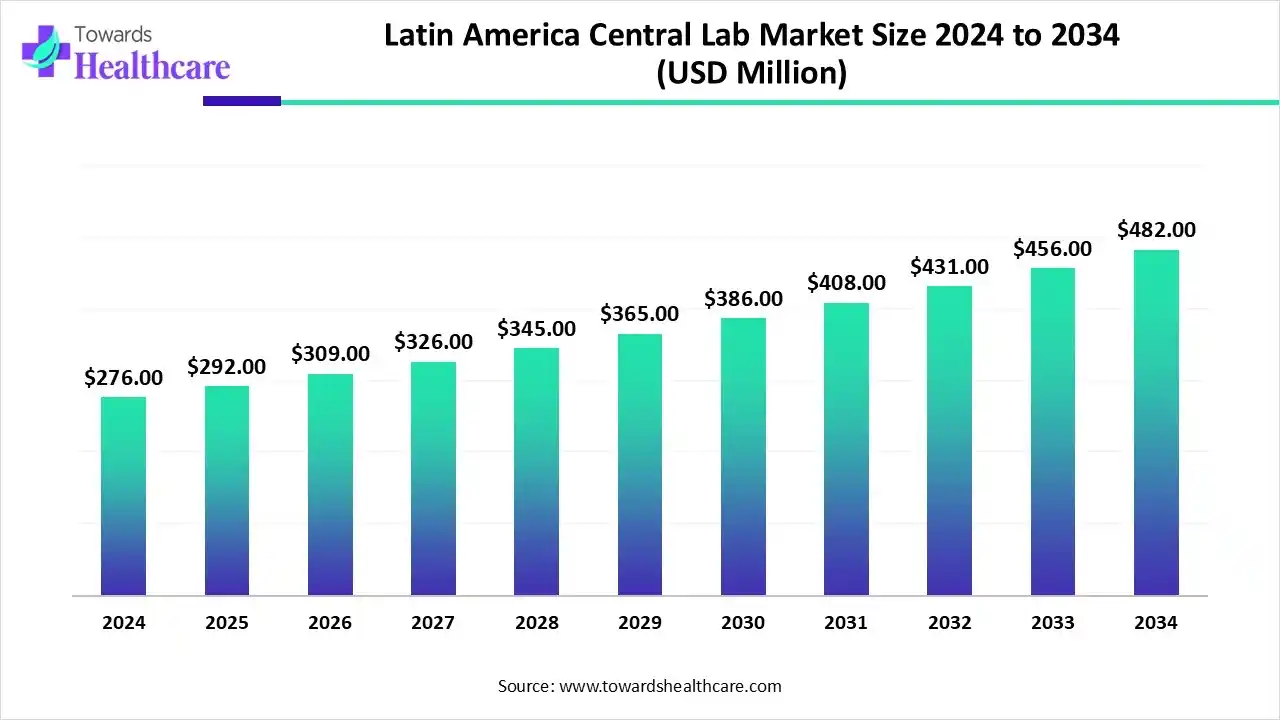
The Latin America central lab market is experiencing steady growth, driven by the rising number of clinical trials, expanding pharmaceutical and biotechnology sectors, and improved regulatory frameworks. Growing demand for high-quality diagnostic services and standardized testing supports this trend. Among regional countries, Brazil dominates the market due to its advanced healthcare infrastructure, skilled workforce, and strong government initiatives to promote clinical research and innovation.
The Latin America central lab market is expanding as global pharma companies increasingly outsource laboratory services to the region for cost efficiency and access to a diverse patient population. It is a network of specialized laboratories that support clinical trials by providing standardized testing, data management, and analytical services across the region.
The market is increasing due to its cost-effective operations, growing pool of treatment-naïve patients, and improving regulatory standards that attract global clinical trials. The region offers diverse patient demographics, faster recruitment rates, and expanding research infrastructure, making it an appealing destination for pharmaceutical and biotech companies. Additionally, increasing collaboration between international CROs and local labs further enhances efficiency and quality in clinical trial support services.
AI is transforming the Latin America central lab market by enhancing data accuracy, automating sample analysis, and improving turnaround times for clinical trial results. It enables predictive analytics, real-time monitoring, and efficient data management, reducing human error and operational costs. Additionally, AI-driven platforms support faster decision-making in drug development and patient safety assessment, positioning Latin America as a competitive hub for global clinical research and laboratory innovation.
| Company Name | Headquarters | Recent Product Focus |
| QIAGEN | Venlo, Netherlands | Focuses on the automated PCR system and its syndromic testing platforms |
| OPKO Health, Inc. | Miami, Florida, U.S. | Focus on the 4Kscore test, which helps to assess the risk for prostate cancer. |
| Charles River Laboratories | Wilmington, Massachusetts, U.S. | Focuses on advanced therapies like cell and gene therapy and accelerated digital-enabled drug development. |
| Genomics Health | Redwood City, California, U.S. | Major focus on targeted discovery and drug design for DNA and RNA therapies, and AI-powered solutions for genomic medicine |
| Quest Diagnostics | Secaucus, New Jersey, U.S. | The major focus area is oncology includes MRD and liquid biopsy tests, infectious tests, neurological biomarkers, and cardiometabolic panels. |
| Table | Scope |
| Market Size in 2026 | USD 322.51 Million |
| Projected Market Size in 2035 | USD 773.25 Million |
| CAGR (2025 - 2034) | 10.20% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Service Type, By Therapeutic Area, By Clinical Trial Phase, By Modality, By End-User |
| Top Key Players | OPKO Health, Inc., Fresenius Medical Care AG & Co. KGaA, QIAGEN, Quest Diagnostics Incorporated, Charles River Laboratories, Laboratory Corporation of America Holdings (LabCorp), Genomic Health (Exact Sciences Corporation), DASA |
The biomarker assays segment held the largest market share with a revenue share in 2025 due to the rising demand for precision medicine and targeted therapies. Increasing adoption of biomarkers in oncology, cardiovascular, and neurological disease research has driven high utilization in clinical trials and diagnostics. Biomarker assays enable early disease detection, patient stratification, and treatment monitoring, improving therapeutic outcomes. Additionally, growing investments in R&D and supportive regulatory frameworks have further accelerated the adoption of biomarker-based testing across the pharmaceutical and biotechnology sectors globally.
The molecular & genetic testing segment is expected to grow at the fastest rate due to rising demand for early and accurate disease detection, personalized treatment plans, and advancements in technologies such as next-generation sequencing (NGS) and PCR. Increasing prevalence of genetic disorders, oncology-focused diagnostics, and expanding research in precision medicine are driving adoption, with hospitals, diagnostic centers, and research laboratories increasingly integrating molecular testing into routine healthcare and clinical trial workflows globally.
The clinical chemistry analyzers segment dominated the market in 2025, accounting for laboratory test usage globally, due to their ability to perform multiple routine biochemical tests efficiently. Hospitals and central labs, representing the largest end-users, rely on these analyzers for high-throughput testing, rapid results, and cost-effectiveness. With automation reducing human error and increasing productivity, the segment is projected to grow steadily over the next decade, supporting the rising demand for clinical diagnostics in both developed and emerging regions.
The molecular techniques segment is expected to grow at the fastest rate due to increasing adoption of PCR and next-generation sequencing (NGS) for early and precise disease detection. Molecular tests currently represent roughly 30% of global diagnostic test volumes, and this proportion is expected to rise as hospitals, research labs, and clinical trials expand their use. Growth is driven by applications in oncology, infectious diseases, and genetic disorder testing, along with technological advancements and rising demand for personalized medicine.
The hospitals & healthcare systems segment led the market with the revenue share in 2025 due to high patient volumes, advanced laboratory infrastructure, and demand for routine and specialized diagnostic testing. Hospitals perform the majority of clinical tests, representing roughly 50–55% of all diagnostic procedures globally, including oncology, infectious disease, and molecular diagnostics. The growing number of hospital admissions, increasing prevalence of chronic diseases, and integration of advanced diagnostic technologies continue to drive the dominance of hospitals and healthcare facilities in the central laboratory market.
The contract research organizations (CRO) segment is expected to grow at the fastest CAGR during the forecast period due to increasing outsourcing of clinical trials and laboratory testing by pharmaceutical and biotechnology companies. CROs currently handle an estimated 20–25% of global clinical trial testing volumes, a proportion expected to rise as drug development becomes more complex. Growth is driven by the demand for cost-effective, specialized laboratory services, faster trial timelines, and access to advanced molecular and biomarker testing technologies.
The blood/plasma/serum segment held the highest market share, accounting in Latin America due to its extensive use in clinical diagnostics. Blood samples are routinely used for biochemical, immunological, and molecular tests in hospitals and central laboratories. For example, Brazil’s Ministry of Health reported over 12 million routine blood tests conducted nationwide in 2023 for public health monitoring and disease detection. Their reliability and comprehensive diagnostic value continue to drive dominance in the region.
The saliva/swabs segment is expected to grow at the fastest CAGR in Latin America due to increasing adoption of non-invasive diagnostic methods. For instance, the Government of Brazil has authorized self-collected saliva specimens for SARS-CoV-2 detection, enhancing accessibility and patient comfort. This approach has been successfully implemented in various regions, demonstrating its potential to expand testing coverage and improve public health outcomes. The convenience and safety of saliva/swab testing drive its growing preference in the region.
The infectious diseases segment held the largest market share with the revenue share in 2025 due to the high prevalence of diseases like dengue, Zika, and tuberculosis in Latin America. For instance, Brazil's Ministry of Health reported over 1.5 million cases of dengue in 2023, highlighting the demand for diagnostic testing. Central laboratories play a crucial role in disease surveillance and outbreak management, driving the growth of this segment in the region.
The rare diseases & genetic screening segment is expected to grow at the fastest CAGR in Latin America due to increasing adoption of non-invasive diagnostic methods. For instance, the Government of Brazil has authorized self-collected saliva specimens for SARS-CoV-2 detection, enhancing accessibility and patient comfort. This approach has been successfully implemented in various regions, demonstrating its potential to expand testing coverage and improve public health outcomes. The convenience and safety of genetic screening drive its growing preference in the region.
The outsourced/third-party central labs segment held the largest market share, accounting in 2025 due to growing demand for specialized testing services and efficiency in handling large sample volumes. In Brazil, the Ministry of Health reported that over 2.1 million COVID-19 RT-PCR tests were processed by regional and outsourced laboratories in 2024. This demonstrates the reliance on third-party labs for high-throughput diagnostics, disease surveillance, and public health initiatives across Latin America.
The hybrid segment is expected to grow at the fastest pace during the forecast period. This growth is driven by the increasing demand for specialized testing, faster turnaround times, and scalable laboratory capacity. Hybrid models allow central labs to leverage local expertise while accessing advanced technologies and global standards. Their flexibility and efficiency make them increasingly preferred for diagnostic testing, disease surveillance, and public health initiatives across Latin America.
In 2025, Brazil's Ministry of Health implemented the Clinical Research Law, aiming to modernize the clinical research framework. This legislation reduces the approval process for clinical trials from up to six months to 30 days, enhancing efficiency and attracting international investments. With a population of approximately 214 million, Brazil's diverse genetic and cultural landscape offers unique opportunities for clinical research, positioning the country as a strategic hub for laboratory-based studies in Latin America.
How is Mexico Approaching the Latin America Central Lab Market?
Mexico is approaching the market by expanding partnerships with local laboratories and enhancing molecular and genomic testing capabilities. The company focuses on integrating advanced diagnostic technologies with regional laboratory networks to improve testing efficiency and accessibility. These initiatives aim to support public health programs, infectious disease monitoring, and research initiatives across Latin America. By leveraging local expertise and standardized protocols, MaxiCo seeks to strengthen centralized laboratory services and accelerate market growth.
How is Argentina Accelerating the Latin America Central Lab Market?
Argentina is accelerating the market by expanding its network of accredited central laboratories and promoting molecular and genomic testing across the country. The Ministry of Health has enhanced laboratory infrastructure to support infectious disease monitoring and public health programs, increasing testing coverage nationwide. These initiatives improve diagnostic capabilities, enable faster disease detection, and strengthen the country’s role as a key hub for centralized laboratory services in Latin America.
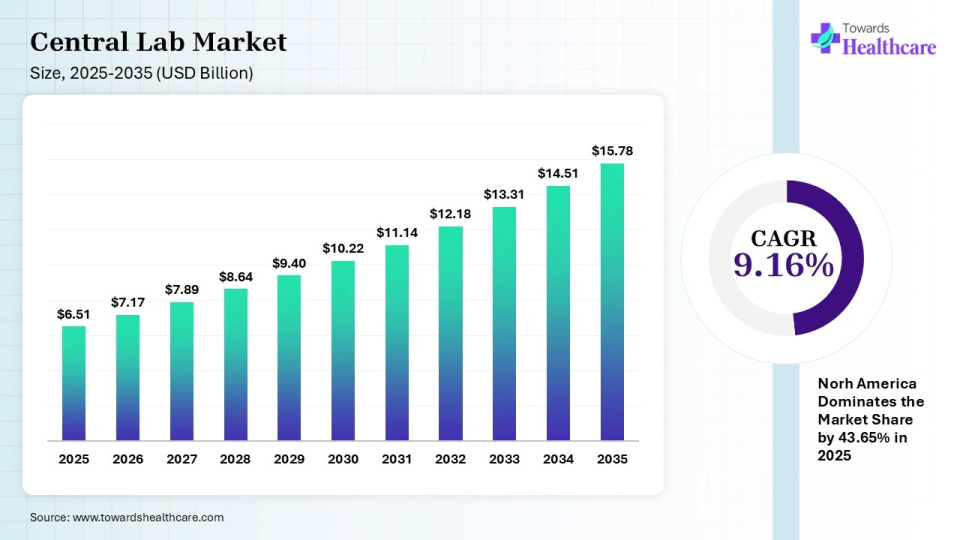
The global central laboratory market is valued at USD 7.17 billion in 2026 and is projected to reach USD 15.78 billion by 2035, growing at a CAGR of 9.16% between 2026 and 2035, driven by increasing investments in R&D and a growing demand for clinical trials.
Clinical Trials
Regulatory approvals
Patient Support and Services
By Service / Offering
By Test / Technology Type
By End-User / Customer
By Sample Type
By Therapeutic Area / Application
By Business / Operating Model
By Region