Learn About the Top Companies in the Acute Pancreatitis Market
- Pfizer
- Merck & Co.
- Baxter International
- Abbott Laboratories
- B. Braun SE
- CalciMedica Inc.
- Dynavax Technologies
- Fresenius SE & Co.
- GlaxoSmithKline
- Olympus Corporation
- SCM Lifescience
- Boston Scientific Corporation
- AnGes
- Mitsubishi Tanabe Pharma
- Medtronic
- Ixaka Ltd (formerly Rexgenero)
- Mercator MedSystems
- Hemostemix Inc.
- Caladrius Biosciences
- Reven Pharmaceuticals
What is Acute Pancreatitis?
The global acute pancreatitis market comprises diagnostics, therapeutics, supportive care solutions, hospital services, and medical technologies used in the management of acute pancreatitis. The market covers pharmaceutical treatments (analgesics, antibiotics, IV fluids, enzyme inhibitors), nutritional support, endoscopic and surgical interventions, imaging diagnostics (CT, MRI, ultrasound), monitoring tools, and hospital-based critical care services.
Growth is driven by the rising prevalence of gallstones, increasing alcohol consumption, higher incidence of hypertriglyceridemia-induced pancreatitis, improved diagnostic imaging, and demand for minimally invasive procedures (ERCP, EUS). Hospitals dominate patient management, while advancements in biologics and anti-inflammatory therapies represent emerging growth areas.
Market Growth
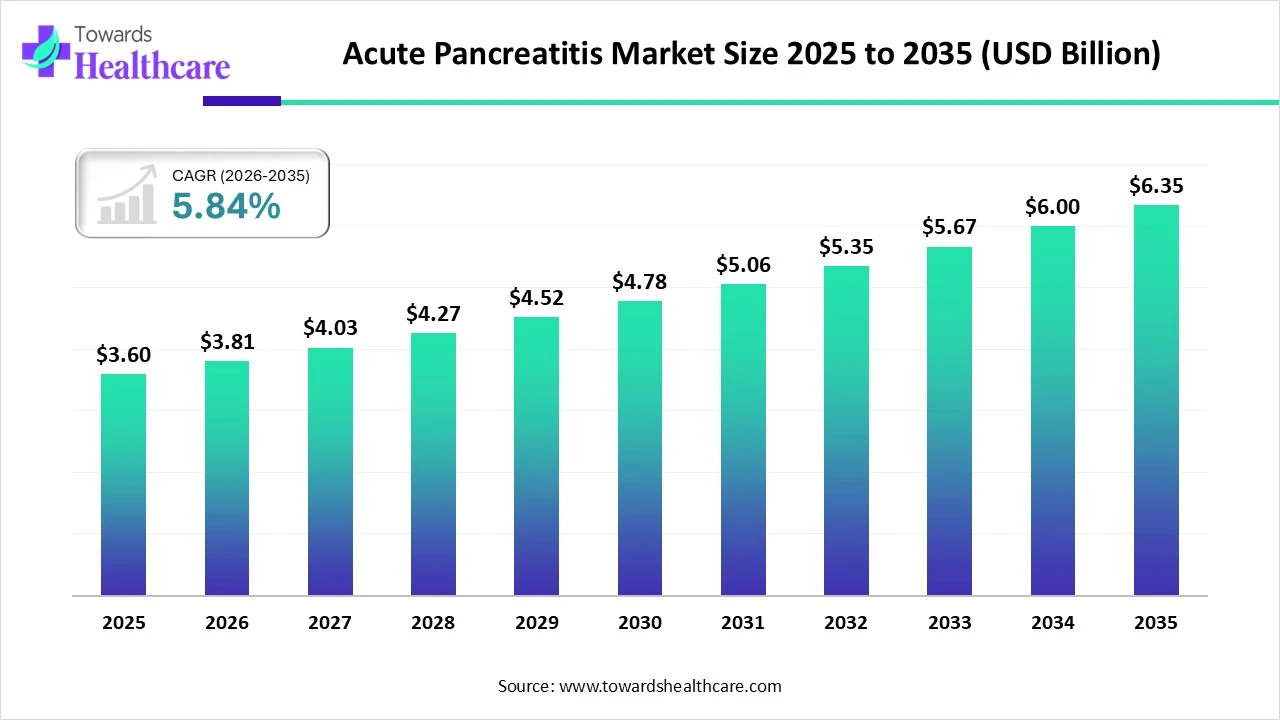
The global acute pancreatitis market size is calculated at US$ 3.6 in 2025, grew to US$ 3.81 billion in 2026, and is projected to reach around US$ 6.35 billion by 2035. The market is expanding at a CAGR of 5.84% between 2026 and 2035.
Top Vendors in the Acute Pancreatitis Market & Their Offerings
1. Pfizer
-
Offerings in Acute Pancreatitis: Pain management medications (analgesics), antibiotics, and sterile injectables.
-
Contribution to Market: Provides essential supportive care drugs fundamental to standard treatment protocols in hospitals.
-
Focus Area: Supportive Care
2. Abbott Laboratories
-
Offerings in Acute Pancreatitis: IV fluids, nutritional products (enteral/parenteral), diagnostic tools/assays.
-
Contribution to Market: Supplies critical supportive nutrition and aids in diagnostics to assess severity and manage complications.
-
Focus Area: Diagnostics & Nutrition
3. B. Braun SE
-
Offerings in Acute Pancreatitis: IV solutions, infusion pumps, pain therapy systems, and surgical products.
-
Contribution to Market: Offers comprehensive solutions for fluid management and precise drug delivery within the acute care setting.
-
Focus Area: Fluid & Pain Mgmt
4. CalciMedica Inc.
-
Offerings in Acute Pancreatitis: Auxora (CRAC channel inhibitor drug candidate).
-
Contribution to Market: Developing a novel therapeutic to target the root cause of organ failure in severe cases through clinical trials.
-
Focus Area: Novel Therapeutics
5. Dynavax Technologies
-
Offerings in Acute Pancreatitis: Adjuvants for vaccine development (no direct AP product).
-
Contribution to Market: No direct offerings for acute pancreatitis treatment or diagnostics; they specialize in adjuvant systems for vaccines.
-
Focus Area: (Not Applicable)
Company Landscape
Merck & Co., Inc. (MSD outside the U.S. and Canada)
Company Overview
- Corporate Information:
- Headquarters: Rahway, New Jersey, United States
- Year Founded: 1891 (as Merck & Co.)
- Ownership Type: Public (NYSE: MRK)
- History and Background:
- Began as the U.S. subsidiary of the German Merck KGaA.
- Separated during WWI and became an independent American company.
- Grew into a major global pharmaceutical company through decades of drug discovery and development across various therapeutic areas.
- Key Milestones/Timeline:
- 1891: Founded in the U.S.
- 1933: Established the Merck Research Laboratories (MRL).
- 2009: Completed the merger with Schering-Plough Corporation.
- 2021: Spun off its Organon & Co. business focused on women's health, biosimilars, and established brands, optimizing its focus on high-growth areas like oncology and vaccines.
- Business Overview:
- Focuses on prescription medicines, vaccines, biological therapies, and animal health products.
- Business Segments/Divisions:
-
- Pharmaceutical (includes oncology, vaccines, infectious diseases, cardiovascular, diabetes, and other therapeutic areas, which house the supportive care drugs used in acute pancreatitis).
- Animal Health.
- Geographic Presence:
- Global presence with operations in over 140 countries.
- Significant revenue generation in the United States, Europe, and the Asia Pacific.
- Key Offerings:
- Acute Pancreatitis Relevant: Analgesics (e.g., opioids for pain management), antibiotics (for infection and sepsis complications), and other supportive care medications.
- Core Portfolio: Keytruda (oncology), Gardasil (HPV vaccine), and various other vaccines and specialty drugs.
- End-Use Industries Served:
- Hospitals and Clinics (Primary end-users for acute care treatment).
- Retail and Online Pharmacies.
- Government/Public Health Sector (Vaccines).
- Veterinary Medicine (Animal Health division).
- Key Developments and Strategic Initiatives:
- Mergers & Acquisitions: Focused on smaller, strategic acquisitions to bolster pipeline in high-growth areas like oncology and cardiovascular disease.
- Partnerships & Collaborations: Extensive R&D collaborations with academic institutions and biotechs, particularly in immuno-oncology and infectious disease.
- Product Launches/Innovations: Continual launch of new indications for existing blockbuster drugs and pipeline medications.
- Capacity Expansions/Investments: Ongoing capital investment in manufacturing facilities to support growing demand for key products.
- Regulatory Approvals: Consistent stream of FDA and international regulatory approvals for new drug indications and pipeline assets.
- Distribution Channel Strategy:
- Primarily through wholesale distributors to retail pharmacies, hospitals, and clinics.
- Direct sales force targeting key healthcare providers and systems.
- Government and public sector contracts for vaccine distribution.
- Technological Capabilities/R&D Focus:
- Core Technologies/Patents: Expertise in small molecule drugs, biologics, and vaccine development. Holds numerous patents across its diversified portfolio.
- Research & Development Infrastructure: Operates Merck Research Laboratories (MRL) globally, one of the largest and most successful research organizations in the pharmaceutical industry.
- Innovation Focus Areas: Oncology, vaccines, infectious diseases (including HIV and emerging pathogens), cardiovascular and metabolic diseases.
- Competitive Positioning:
- Strengths & Differentiators: Strong, diversified portfolio with market-leading oncology and vaccine franchises. Robust R&D pipeline. Significant financial resources.
- Market presence & ecosystem role: A global pharmaceutical giant, a dominant player in oncology, and a major supplier of essential hospital medications, including supportive drugs used in acute pancreatitis care.
- SWOT Analysis:
- Strengths: Market leadership in key therapeutic areas; strong patent protection on high-revenue drugs; global reach.
- Weaknesses: Reliance on a few blockbuster drugs for a large portion of revenue; increasing competition in the biosimilars space.
- Opportunities: Expansion of oncology and vaccine pipeline; focus on emerging markets; utilization of AI and machine learning in drug discovery.
- Threats: Patent expirations (loss of exclusivity); competitive pressures from other large pharma/biotech companies; regulatory scrutiny.
- Recent News and Updates:
- Press Releases (2024/2025): Continuously announces positive Phase 3 clinical trial results for pipeline drugs and key oncology/vaccine products. For example, announcements regarding new indications for Keytruda.
- Industry Recognitions/Awards: Frequently recognized as a top R&D spender and a leader in corporate social responsibility (CSR) initiatives related to global health.
Baxter International Inc.
Company Overview
- Corporate Information:
- Headquarters: Deerfield, Illinois, United States
- Year Founded: 1931
- Ownership Type: Public (NYSE: BAX)
- History and Background:
- Pioneered the first commercially prepared intravenous (IV) solution in 1931.
- Focused initially on intravenous fluids and blood transfusion products, expanding over time into medical devices, hospital products, and renal care.
- The core business of supportive care products is highly relevant to acute pancreatitis treatment, which primarily involves aggressive IV hydration.
- Key Milestones/Timeline:
- 1931: Founded by Dr. Don Baxter.
- 1956: Began international expansion.
- 2015: Spun off its BioScience division to form Baxalta (later acquired by Shire/Takeda).
- 2021: Acquired Hillrom to significantly expand its connected care and digital health offerings.
- Business Overview:
- Provides a broad portfolio of essential healthcare products, including acute and critical care therapies, sterile IV solutions, hospital products, and advanced surgical tools.
- Business Segments/Divisions:
- Medical Products and Therapies: Includes IV solutions, administration sets, drug reconstitution systems, and surgical care products (highly relevant for acute pancreatitis supportive care).
- Patient Support and Monitoring: Focuses on devices for connected care, patient monitoring, and smart beds (expanded significantly with the Hillrom acquisition).
- Renal Care: Products for peritoneal dialysis and hemodialysis.
- Geographic Presence:
- Sells products in more than 100 countries.
- Major markets include North America, Europe, and the Asia Pacific.
- Key Offerings:
- Acute Pancreatitis Relevant: Large Volume Parenterals (IV Fluids) (e.g., Lactated Ringer's, Normal Saline - essential for initial, aggressive hydration), sterile IV administration sets, and pre-mixed injectables (analgesics and antibiotics).
- Core Portfolio: Medication delivery systems, acute and chronic dialysis products, and advanced patient monitoring technologies.
- End-Use Industries Served:
- Hospitals (Inpatient, ICU, ORs - largest segment).
- Home Healthcare.
- Dialysis Centers.
- Specialty Clinics.
- Key Developments and Strategic Initiatives:
- Mergers & Acquisitions: 2021 Hillrom Acquisition: A pivotal move to become a global leader in connected care and digital health solutions, expanding their presence from the ICU to the patient's room.
- Partnerships & Collaborations: Collaborates with tech firms and health systems to integrate its medical devices and digital solutions.
- Product Launches/Innovations: Launch of new IV fluid bags and delivery systems, including non-PVC offerings, and new connected care monitoring platforms.
- Capacity Expansions/Investments: Investments in upgrading and expanding global manufacturing capacity for critical supply chain products like IV fluids to mitigate shortages.
- Regulatory Approvals: Ongoing regulatory clearances for new connected medical devices and software, as well as essential drug delivery products.
- Distribution Channel Strategy:
- Direct sales force and distribution network to hospitals and healthcare systems (dominant channel).
- Partnerships with major healthcare distributors (e.g., McKesson, Cardinal Health).
- Direct-to-patient model for home renal care.
- Technological Capabilities/R&D Focus:
- Core Technologies/Patents: Expertise in sterile manufacturing, fluid delivery systems, and acute renal replacement therapy. Growing portfolio of patents in connected care and digital monitoring.
- Research & Development Infrastructure: Focuses R&D on improving medication delivery, patient monitoring, and expanding acute therapy options.
- Innovation Focus Areas: Connected care (smart beds, remote monitoring), advanced surgical devices, and enhancing the safety/efficiency of drug compounding and delivery.
- Competitive Positioning:
- Strengths & Differentiators: Dominant position in essential hospital products, particularly IV fluids, which are irreplaceable for acute pancreatitis supportive care. Deep integration within hospital workflows. Global manufacturing scale.
- Market presence & ecosystem role: A foundational supplier for virtually every hospital worldwide, critical for managing acute, life-threatening conditions like acute pancreatitis, sepsis, and renal failure.
- SWOT Analysis:
- Strengths: Market leadership in essential supportive care products (IV fluids); strong global supply chain; bolstered technology portfolio via Hillrom acquisition.
- Weaknesses: Historically lower R&D spend compared to pure-play biopharma; complex integration post-Hillrom acquisition.
- Opportunities: Leveraging connected care portfolio to improve outcomes in critical care settings; expanding use of IV solutions in emerging markets; addressing global IV fluid shortages.
- Threats: Supply chain disruptions; pricing pressure on generic hospital supplies; competition from contract manufacturers; high debt load from recent acquisitions.
- Recent News and Updates:
- Press Releases (2024/2025): Focus on the integration of Hillrom and the launch of new digital health and patient monitoring solutions. Announced expansion of smart bed technology rollouts.
- Industry Recognitions/Awards: Often recognized for its leadership in the essential hospital supplies and medical technology segments. Recognized for supply chain resilience in critical care products.
-
Acute Pancreatitis Market Value Chain Analysis
R&D
R&D focuses on finding novel therapeutic agents, imaging methods (such as sophisticated AI analysis of CT/MRI scans), and biomarkers, from laboratory studies to clinical hypotheses.
Key Companies: Siemens Healthineers, Olympus Corporation, Cook Group Incorporated, Neusoft Medical System, Bioseutica B.V., Lamassu Pharma, LLC, GNT Pharma Co., etc.
Clinical Trials and Regulatory Approvals
It involves proving the safety and effectiveness of novel diagnostics and treatments through human trials, which can be difficult because of the high placebo response in pain. This is followed by gaining FDA or comparable agency market authorization.
Key Companies: Arctx Medical, AcelRx Pharmaceuticals, LipimetiX Development, Angion Biomedica, SCM Lifescience, Panafina, Inc., etc.
Patient Support and Services
It includes lifestyle counseling, long-term management to avoid recurrence, supportive care (IV fluids, pain management, nutrition), and putting patients in touch with advocacy organizations such as the National Pancreas Foundation.
Key Companies: Abbott Laboratories, Baxter International Inc., Fresenius SE & Co. KGaA, Medtronic, Boston Scientific, etc.
Recent Developments in the Acute Pancreatitis Market
- In September 2025, Olezarsen's pivotal Phase 3 CORE and CORE2 studies in patients with severe hypertriglyceridemia (sHTG) showed encouraging topline results, according to Ionis Pharmaceuticals, Inc. In the trials, olzarsen showed favorable safety and tolerability, along with highly statistically significant reductions of 85% in acute pancreatitis events and up to 72% in fasting triglycerides compared with placebo.
- In August 2025, Arctx Medical announces new leadership and receives FDA IDE approval for a pivotal clinical trial of its ACC kit for the treatment of acute pancreatitis.
Become a valued research partner with us, please feel free to contact us at sales@towardshealthcare.com
Keypoints
- Company Overview
- Locations Subsidiaries/Geographic reach
- Key Executives
- Company Financials
- Patents registered
- SWOT Analysis
- Applications Catered
- Strategic collaborations
- Recent Developments
- Competitive Benchmarking
