May 2026

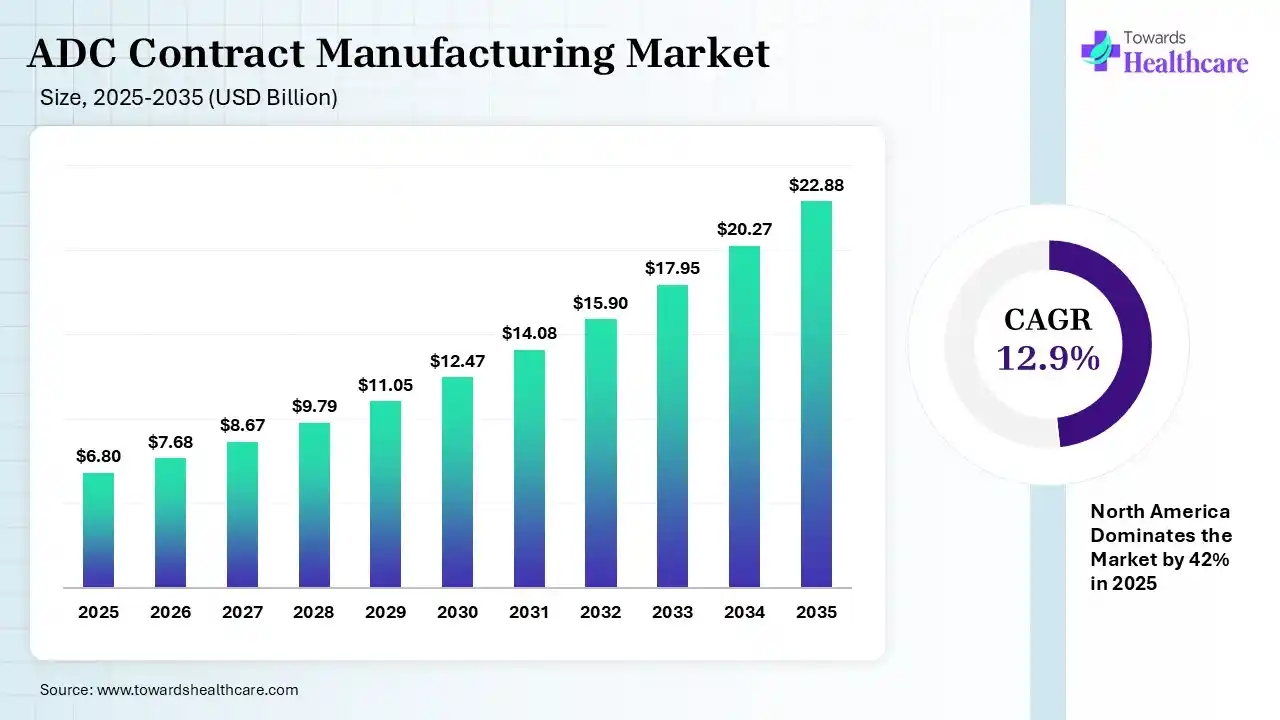
The global ADC contract manufacturing market size was estimated at USD 6.8 billion in 2025 and is predicted to increase from USD 7.68 billion in 2026 to approximately USD 22.88 billion by 2035, expanding at a CAGR of 12.9% from 2026 to 2035. The growing cancer burden globally is increasing the demand for ADC products, driving the adoption of ADC contract manufacturing. Growing R&D activities, funding, government support, outsourcing trends, technological advancements, and collaborations are enhancing the market growth.
The ADC contract manufacturing market is driven by growing technical complexity and extreme capital requirements. The ADC contract manufacturing refers to the antibody-drug conjugate outsourcing to the specialized third-party companies for their development and production. They offer various services such as ADC production, commercial manufacturing, quality testing, formulation optimization, management of cytotoxic payloads, and clinical trial support.
AI offers a wide range of applications in the market, including process optimization and predictive analytics, reducing the chances of product or batch failures. It also helps in maintaining a consistent drug antibody ratio, which also helps in detecting any anomalies, ensuring quality control. AI also helps in the development of personalized ADC products and offers automation, reducing the chances of human errors and accelerating product development.
Blooming Outsourcing Trends
A rise in the ADC drug pipeline is increasing the outsourcing trends, promoting the use of ADC contract manufacturing services. They are being preferred to avoid the use of expensive facilities and complex technologies and promote faster, higher-quality, and safety products development.
Rising Adoption of End-To-End Services
The lack of manufacturing capacities and skilled personnel is increasing the shift towards ADC contract manufacturing to leverage their end-to-end services. Their one-stop solution offering antibody production, conjugations, payload synthesis, and fill-finish solutions is also attracting the client.
Technological Advancements
Growing technological advancements are increasing the adoption of new technologies, driving collaborations with ADC contract manufacturing organizations. Their site-specific conjugation technologies, improved linker chemistry, and continuous and automated manufacturing are also increasing their demand.
| Table | Scope |
| Market Size in 2026 | USD 7.68 Billion |
| Projected Market Size in 2035 | USD 22.88 Billion |
| CAGR (2026 - 2035) | 12.9% |
| Leading Region | North America by 42% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Service Type, By Payload Type, By Linker Type, By Conjugation Technology, By Scale of Operation, By End User, By Region |
| Top Key Players | Lonza Group, Axplora (Novasep), WuXi Biologics, MilliporeSigma (Merck KGaA), Samsung Biologics, Cambrex, Catalent, Inc., Sterling Pharma Solutions, Piramal Pharma Solutions, Abzena |
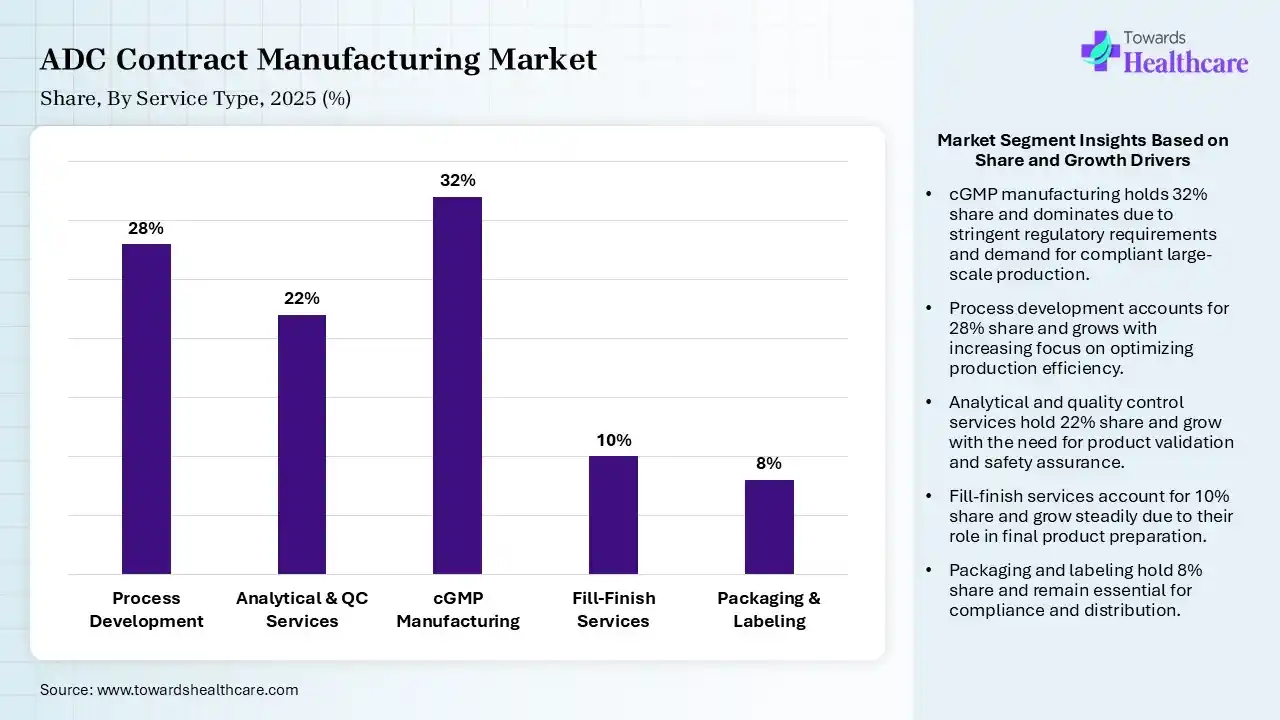
| Segment | Share 2025 (%) |
| Process Development | 28% |
| Analytical & QC Services | 22% |
| cGMP Manufacturing | 32% |
| Fill-Finish Services | 10% |
| Packaging & Labeling | 8% |
The cGMP Manufacturing Segment Dominated the Market With 32% in 2025
The cGMP manufacturing segment accounted for the highest revenue share of 32% of the ADC contract manufacturing market in 2025, due to a rise in the large-scale ADC production demand driven by growth in their approvals. High-capital facilities also pushed outsourcing to CDMOs. Growth in the commercialization pipelines also increased their demand.
The process development segment held the second-largest share of 28% of the market in 2025 and is expected to show the highest growth during the forecast period, due to the increasing complexity of ADC design, which drives demand for optimized processes. They are also used for early-stage outsourcing to boost innovation efficiency. Rising pipeline also fuels development contracts, increasing their use.
The analytical & quality control services segment held 22% of the ADC contract manufacturing market share in 2025, and is expected to grow at the fastest CAGR during the forecast period, driven by regulatory scrutiny, which increases the need for robust analytical validation, driving their adoption rates. Complex ADC characterization is also expanding testing demand. Biopharma outsourcing also accelerates QC services.
The fill-finish services segment held 10% of the market share in 2025, due to a rise in the sterile handling requirements, which boost specialized fill-finish demand. Growth in injectable oncology drugs also increases their utilization. Expansion of biologics packaging capacity is also supporting their growth.
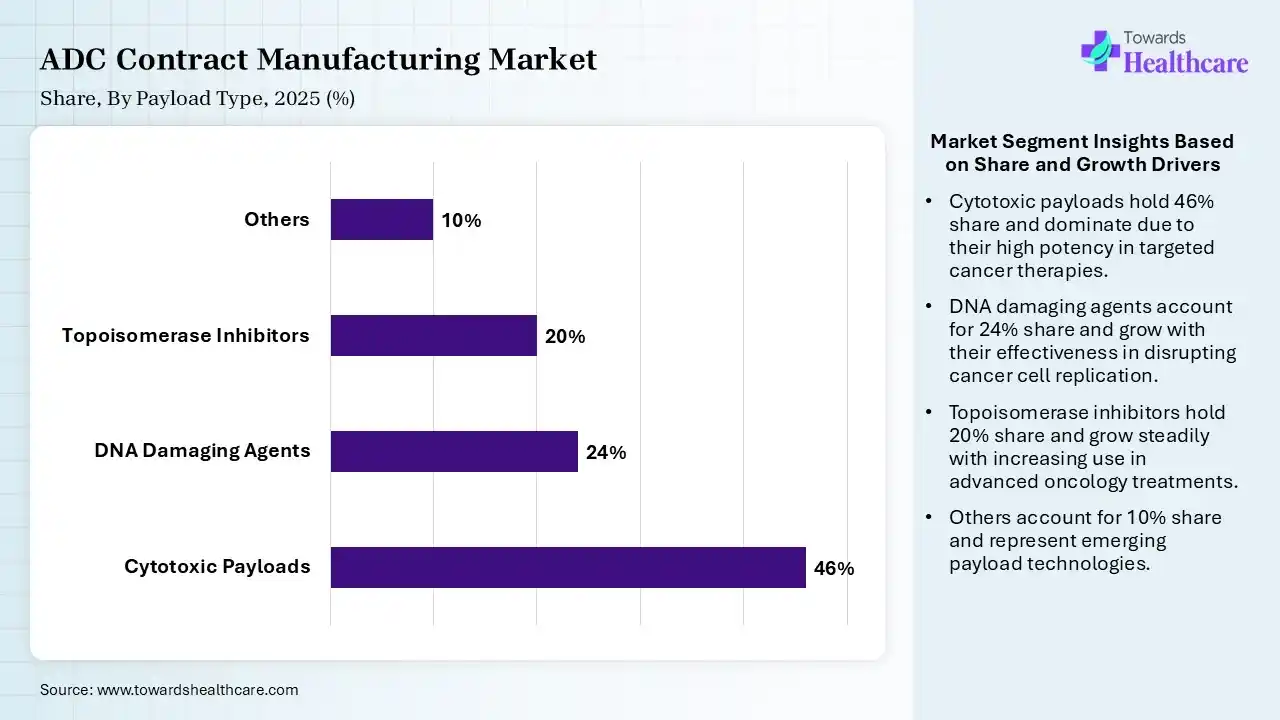
| Segment | Share 2025 (%) |
| Cytotoxic Payloads | 46% |
| DNA Damaging Agents | 24% |
| Topoisomerase Inhibitors | 20% |
| Others | 10% |
The Cytotoxic Payloads Segment Dominated the Market With 46% in 2025
The cytotoxic payloads segment held a major revenue share of 46% of the ADC contract manufacturing market in 2025, due to established clinical success, which contributed to their continued adoption. A strong pipeline of MMAE-based ADCs also increased the demand for cytotoxic payloads. Proven efficacy also ensured commercial dominance.
The DNA-damaging agents segment held the second-largest share of 24% of the market in 2025 and is expected to expand rapidly during the forecast period, due to their superior potency driving next-gen ADC development. Increased investment in PBD-based payloads also supports their use. Rising focus on targeted cancer therapies is also increasing their demand.
The topoisomerase inhibitors segment held 20% of the ADC contract manufacturing market share in 2025, due to expanding approvals, which are increasing their manufacturing demand. Their improved safety profiles are also promoting their adoption. Strong pipeline growth is also supporting segment expansion.
The others segment held 10% of the market share in 2025, due to growing innovation in novel payloads, which diversifies applications. A rise in the early-stage research also drives niche demand. Emerging technologies are also expanding their opportunities.
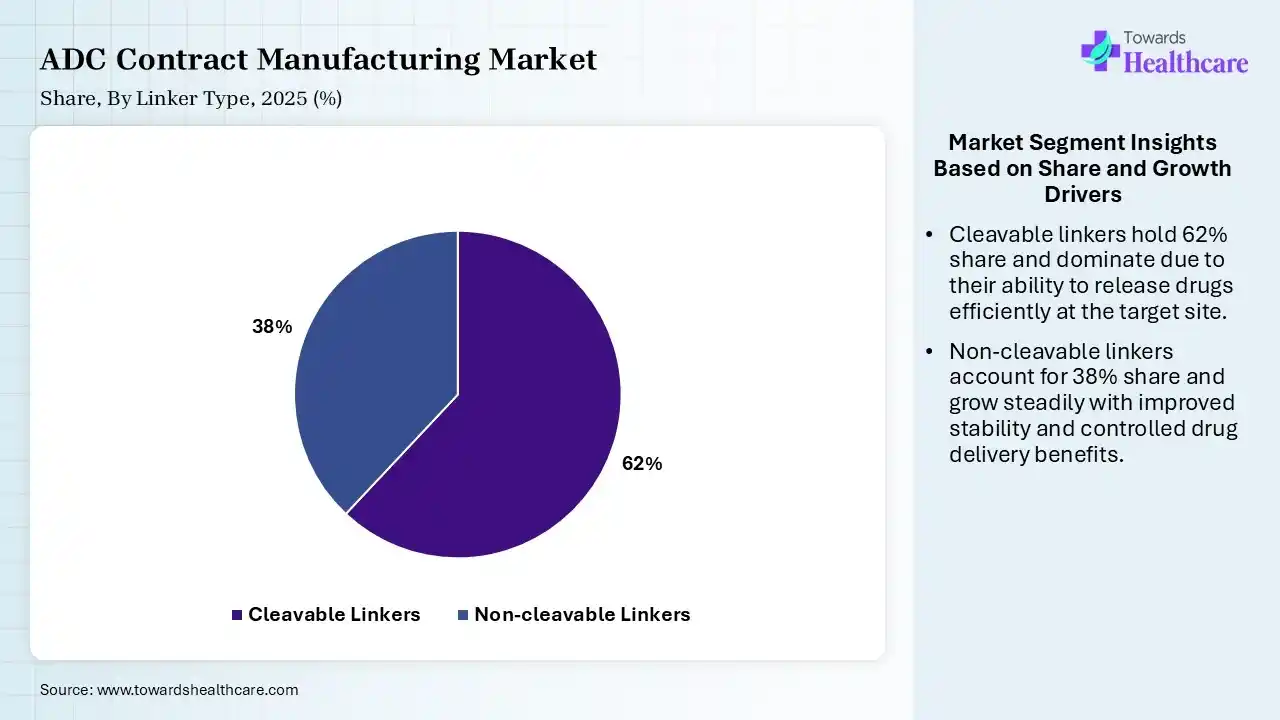
| Segment | Share 2025 (%) |
| Cleavable Linkers | 62% |
| Non-cleavable Linkers | 38% |
The Cleavable Linkers Segment Dominated the Market With 62% in 2025
The cleavable linkers segment held the largest revenue share of 62% of the ADC contract manufacturing market in 2025 and is expected to grow with the highest CAGR during the forecast period, due to enhanced tumor-specific drug release, which increased their adoption. Advances in linker chemistry improved their efficacy, which enhanced their use. Strong pipeline preference for cleavable systems also increased their use.
The non-cleavable linkers segment held the second-largest share of 38% of the market in 2025, driven by improved stability, which supports their continued usage. Established ADC products rely on non-cleavable systems, fueling their demand. Lower toxicity profiles also sustain their demand.
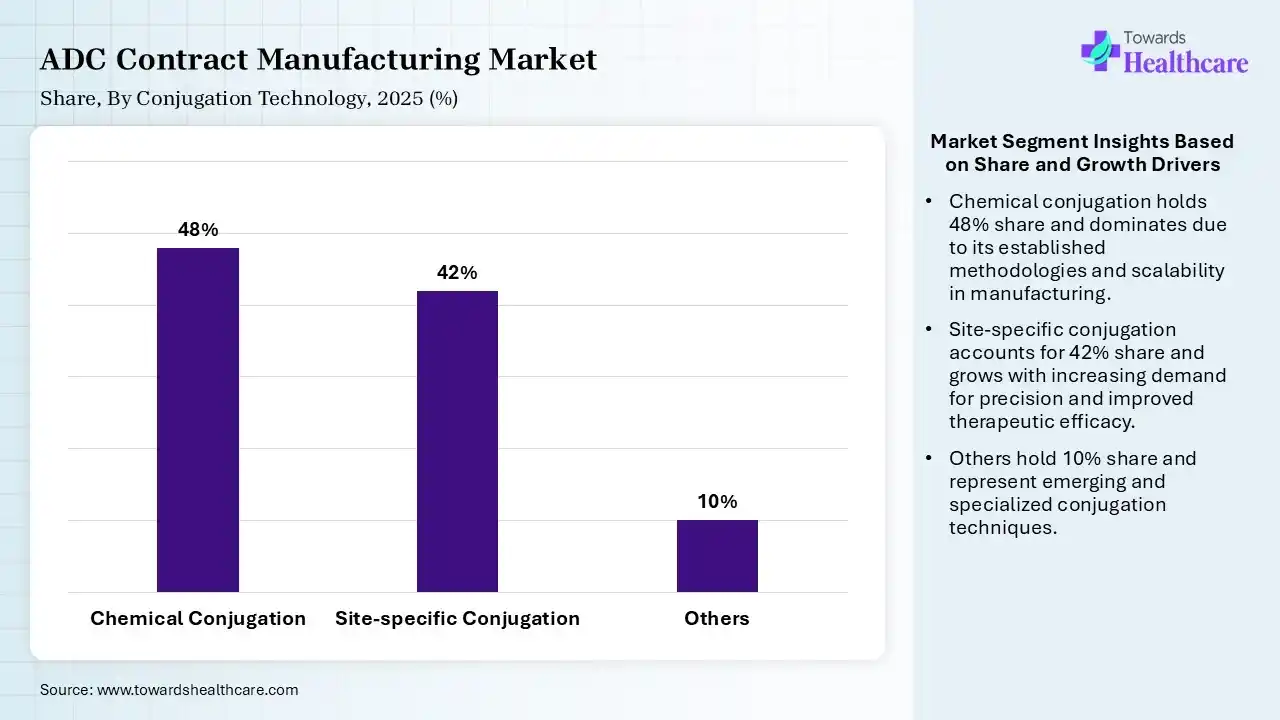
| Segment | Share 2025 (%) |
| Chemical Conjugation | 48% |
| Site-specific Conjugation | 42% |
| Others | 10% |
The Chemical Conjugation Segment Dominated the Market With 48% in 2025
The chemical conjugation segment contributed the biggest revenue share of 48% of the ADC contract manufacturing market in 2025, due to its widespread adoption driven by established protocols. Cost-effective manufacturing also increased their preference. The presence of legacy ADCs also maintained the segment strength.
The site-specific conjugation segment held the second-largest share of 42% of the market in 2025 and is expected to gain the highest share during the forecast period, driven by precision targeting that improves therapeutic index. Growing adoption of engineered antibodies is also increasing their demand. Innovation enhances product consistency, driving its adoption.
The others segment held 10% of the ADC contract manufacturing market share in 2025, due to emerging technologies that are supporting niche applications. Ongoing R&D activities also drive innovation, promoting their adoption. The growing development of new ADC products and focus on enhancing their efficacy, safety, and stability are also increasing their use.
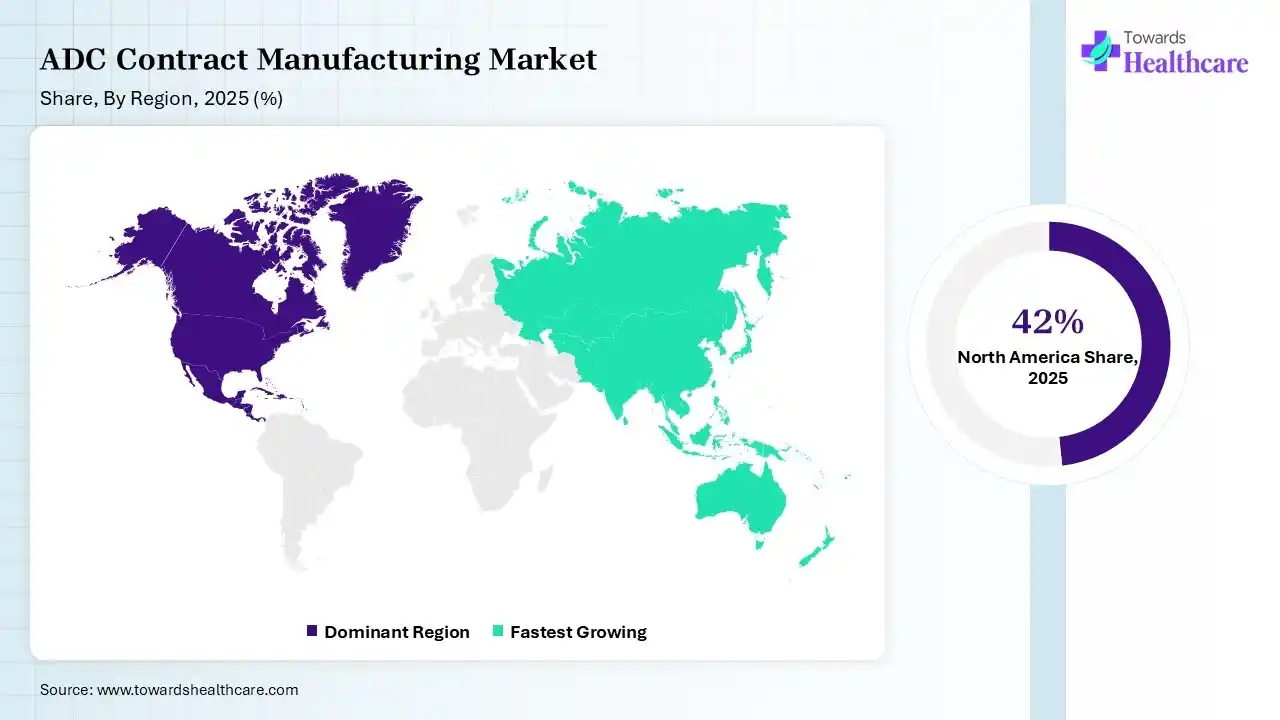
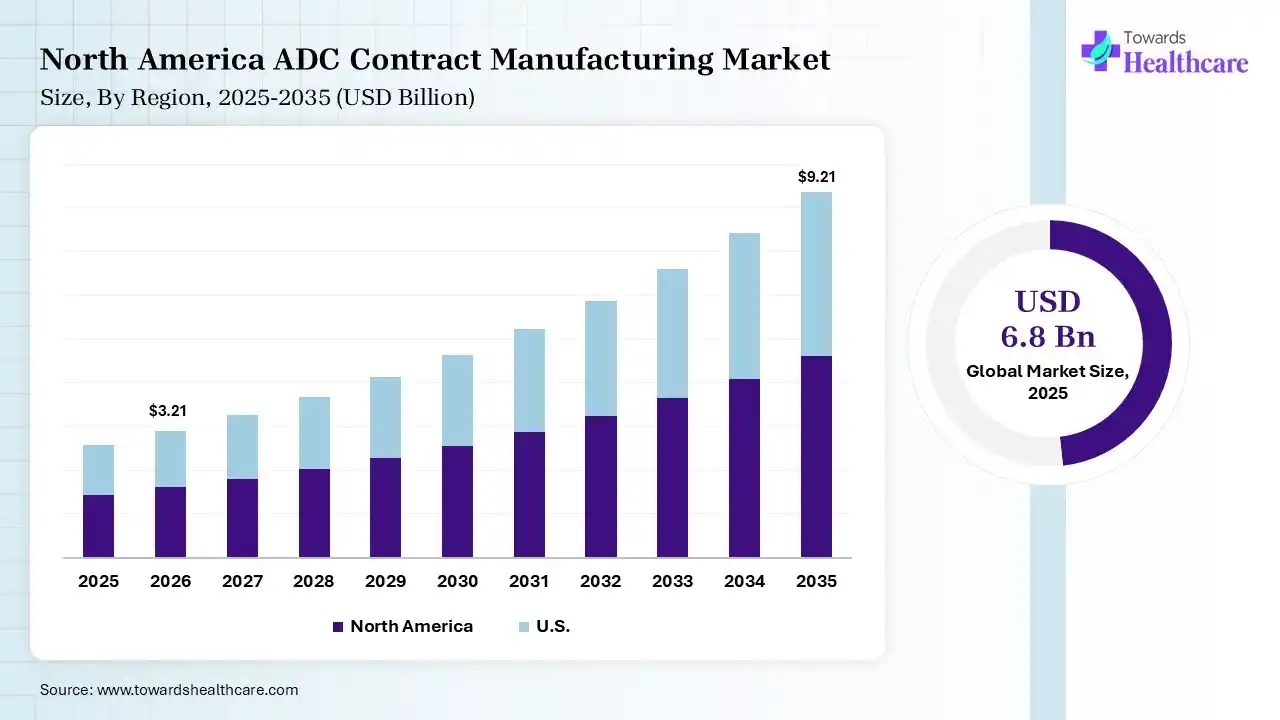
North America dominated the ADC contract manufacturing market with 42% in 2025, due to the strong presence of leading CDMOs and pharma firms. The presence of an advanced regulatory framework and a rise in the oncology drug demand also supported the growth of ADC contract manufacturing. Growth in funding, outsourcing trends, and advanced technology adoption also contributed to the market growth.
U.S. Market Trends
The presence of the largest ADC pipeline globally in the U.S. is driving the demand for ADC contract manufacturing. Strong CDMO presence enhances capacity, where high biologics spending also fuels growth. Growing outsourcing trends and R&D investments are also increasing their adoption rates.
Asia Pacific held 22% share of the ADC contract manufacturing market in 2025 and is expected to grow at the fastest CAGR during the forecast period, due to the rapid expansion of CDMO capabilities, which is driving the ADC contract manufacturing. Cost advantages are also attracting global outsourcing, promoting their use. A growing biotech ecosystem, increasing investments, and growing clinical trials are also accelerating their demand, enhancing the market growth.
China Market Trends
Rapid CDMO expansion and growing GMP infrastructure are boosting global outsourcing in China, increasing the demand for ADC contract manufacturing services. Strong government support and an increasing domestic ADC pipeline are accelerating their growth. Growing biotech investments and expanding clinical trials are also increasing their demand.
R&D
Formulation and Final Dosage Preparation
Packaging and Serialization
Distribution to Hospitals, Pharmacies
Patient Support and Services
| Companies | Headquarters | ADC Contract Manufacturing Services |
| Lonza Group | Basel, Switzerland | End-to-end services |
| Axplora (Novasep) | Raubling, Germany | ADC manufacturing services |
| WuXi Biologics | Wuxi, China | One-stop shop platform for payloads, dug products, and linker discovery, CMC, and GMP manufacturing. |
| MilliporeSigma (Merck KGaA) | Darmstadt, Germany | Comprehensive CDMO services |
| Samsung Biologics | Incheon, South Korea | Large-scale antibody manufacturing |
| Cambrex | East Rutherford, U.S. | Expanded large-scale capacity for ADC payloads and linker |
| Catalent, Inc. | Somerset, U.S. | Bioconjugation, fill-finish, and analytical testing |
| Sterling Pharma Solutions | Cramlington, UK | ADC bioconjugation and development |
| Piramal Pharma Solutions | Mumbai, India | Integrated ADC services |
| Abzena | San Diego, U.S. | ThioBridge conjugation technology |
Strengths
Weaknesses
Opportunities
Threats
By Service Type
By Payload Type
By Linker Type
By Conjugation Technology
By Scale of Operation
By End User
By Region
May 2026
May 2026
April 2026
March 2026