May 2026

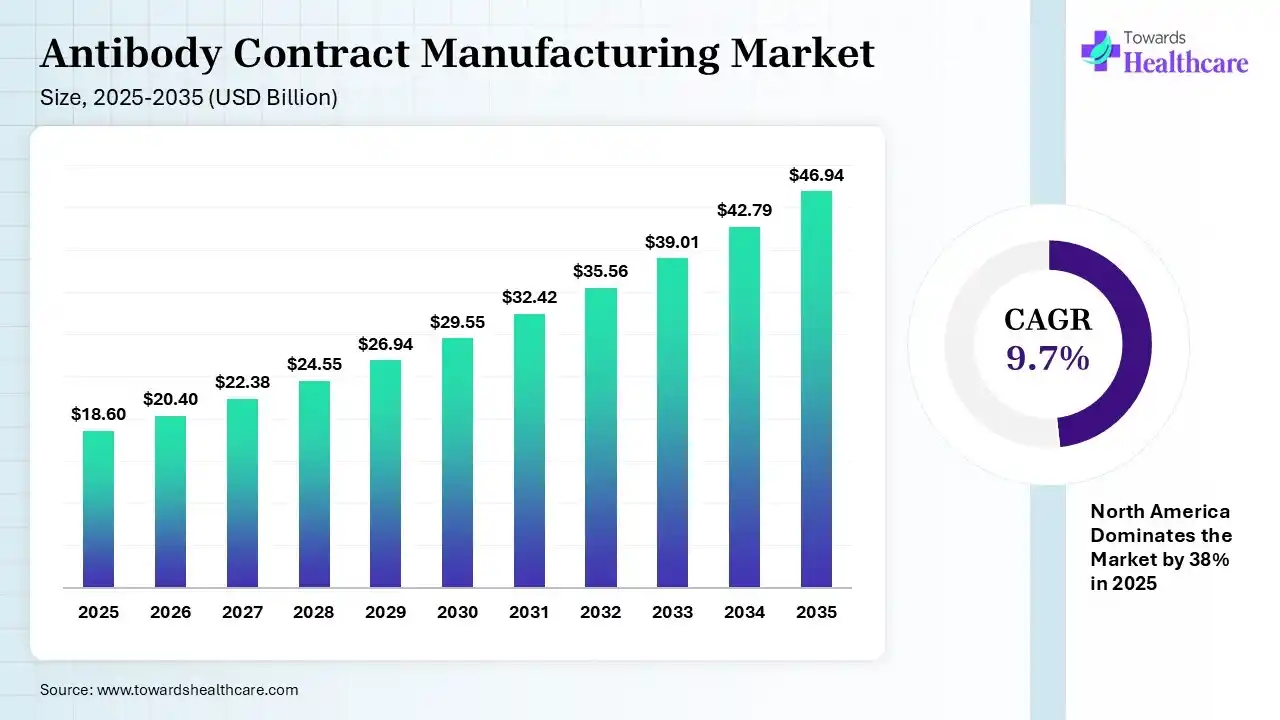
The global antibody contract manufacturing market size was estimated at USD 18.6 billion in 2025 and is predicted to increase from USD 20.4 billion in 2026 to approximately USD 46.94 billion by 2035, expanding at a CAGR of 9.7% from 2026 to 2035. A rise in the chronic disease burden globally is increasing the demand for antibody contract manufacturing services. Growing R&D activities, healthcare investments, advancements in biosimilars, strategic partnerships, technological innovations, and new platform launches are also enhancing the market growth.
The antibody contract manufacturing market is driven by increasing outsourcing trends and a surge in the demand for biologics. The antibody contract manufacturing refers to the outsourcing of therapeutic antibodies to specialized third-party companies for their production. They are preferred for large-scale production and faster drug development due to their affordable services, quality assurance, expertise, and regulatory support.
AI offers process optimization, improving production efficiency and predictive maintenance, reducing the chances of errors, and increasing its adoption in antibody contract manufacturing. It also helps in monitoring process deviation and data analysis, ensuring quality control, and consistent yield of the products. AI also helps in accelerating the antibody manufacturing, workflow automation, and real-time monitoring, which increases their adoption.
Biologics on Rise
The growth in the demand for biologics is increasing the adoption of antibody contract manufacturing services to develop products with high specificity and effectiveness. This increases the use of monoclonal antibodies to drive the development of new biologics for a wide range of disease treatment.
Blooming Advanced Antibody Modalities
The growing R&D activities and collaborations are increasing the development of next-generation antibody-based products, driving the demand for antibody contract manufacturing services. This is increasing the development of antibody-drug conjugates and fragment-based antibodies, creating new opportunities.
Shift Towards Personalized Medicines
A rise in health awareness is driving the demand for patient-specific antibody therapies. This, in turn, is increasing the use of antibody contract manufacturing services for small batch production and flexible manufacturing systems.
| Table | Scope |
| Market Size in 2026 | USD 20.4 Billion |
| Projected Market Size in 2035 | USD 46.94 Billion |
| CAGR (2026 - 2035) | 9.7% |
| Leading Region | North America by 38% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Service Type, By Antibody Type, By Expression System, By Scale of Operation, By End User, By Application, By Region |
| Top Key Players | Lonza Group, Samsung Biologics, FUJIFILM Diosynth Biotechnologies, WuXi Biologics, Siegfried Holding, Thermo Fisher Scientific (Patheon), AGC Biologics, Catalent, Inc, Merck KGaA (MilliporeSigma), Boehringer Ingelheim (BioXcellence) |
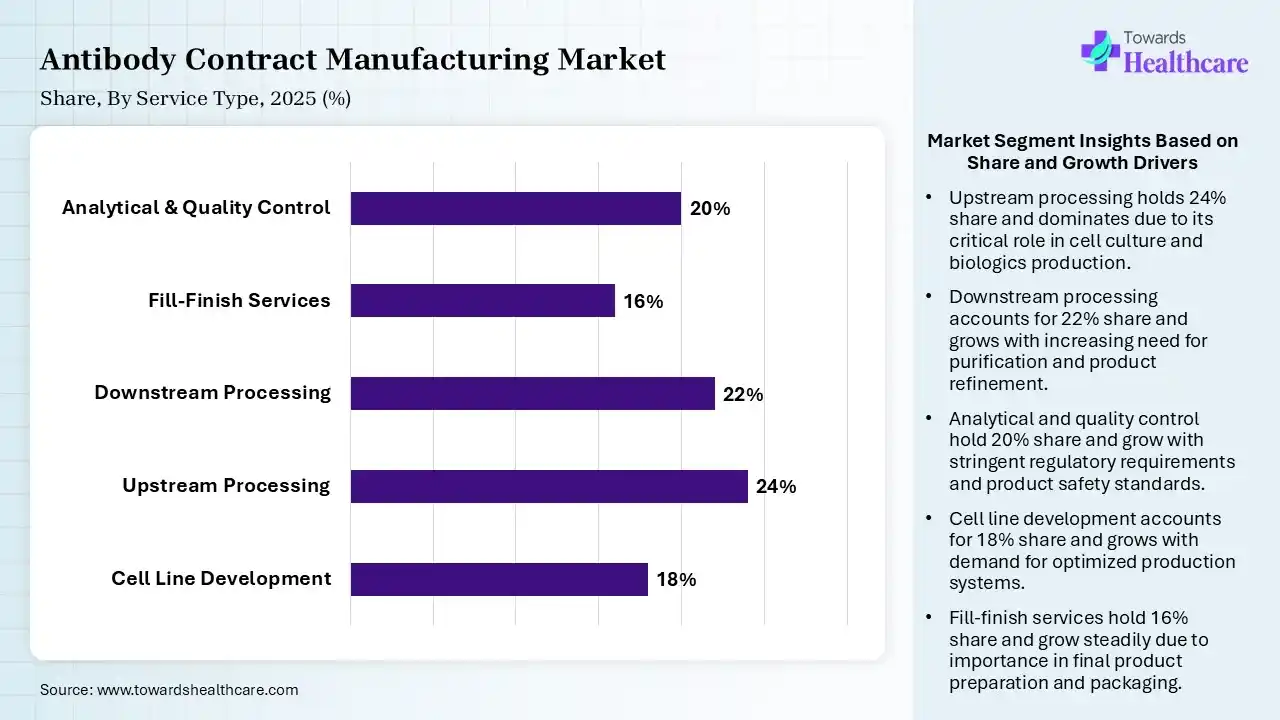
| Segment | Share 2025 (%) |
| Cell Line Development | 18% |
| Upstream Processing | 24% |
| Downstream Processing | 22% |
| Fill-Finish Services | 16% |
| Analytical & Quality Control | 20% |
The Upstream Processing Segment Dominated the Market With 24% in 2025
The upstream processing segment led the antibody contract manufacturing market with 24% share in 2025, due to a growth in the biologics pipeline, which increased the large-scale cell culture needs. Continuous bioprocessing improves efficiency, which also increases the use of antibody contract manufacturing services. Expansion of the CDMOs' investments in bioreactor capacity also increased their adoption.
The downstream processing segment held the second-largest share of 22% of the market in 2025 and is expected to witness the fastest growth with a CAGR of 10.4% during the forecast period, due to the complexity of antibody purification, which drives their demand. High purity standards require advanced chromatography solutions, which promote their use. Automation enhances scalability and consistency, driving their adoption.
The analytical & quality control segment held 20% of the antibody contract manufacturing market share in 2025, due to stringent regulatory requirements driving analytical testing demand. They are also being used in stability studies to ensure product safety. Increasing biosimilar development boosts QC services, increasing their use.
The cell line development segment held 18% of the market share in 2025, due to increasing demand for high-yield cell lines, which drives their outsourcing. Companies seek stable and scalable production platforms, increasing the use of antibody contract manufacturing services. The presence of advanced genetic engineering also accelerates adoption.
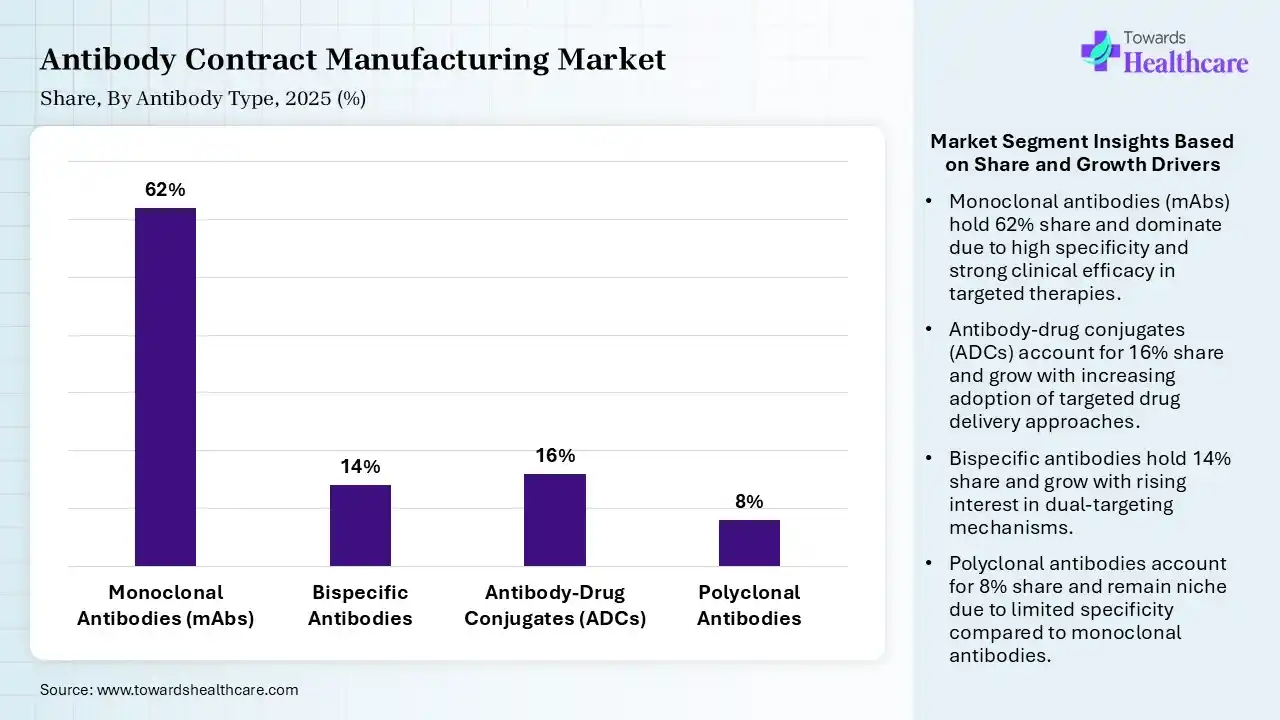
| Segment | Share 2025 (%) |
| Monoclonal Antibodies (mAbs) | 62% |
| Bispecific Antibodies | 14% |
| Antibody-Drug Conjugates (ADCs) | 16% |
| Polyclonal Antibodies | 8% |
The Monoclonal Antibodies (mAbs) Segment Dominated the Market With 62% in 2025
The monoclonal antibodies (mAbs) segment accounted for the highest revenue share of 62% of the antibody contract manufacturing market in 2025, due to a strong pipeline of approved biologics, which sustained their demand. Their wide use in oncology and immunology has also increased their use. Continuous innovation to enhance their therapeutic efficacy also contributed to their advancements.
The antibody-drug conjugates (ADCs) segment held the second-largest share of 16% of the market in 2025, due to their increasing oncology applications, fueling their demand. Advanced linker technologies improve efficacy, which drives their adoption. Manufacturing complexity favors CDMOs, promoting a rise in their use.
The bispecific antibodies segment held 14% of the antibody contract manufacturing market share in 2025 and is expected to show the highest growth with a CAGR of 11.2% during the forecast period, due to rising interest in dual-target therapies, which drives their adoption. Clinical success increases investment, which boosts their advancements. Complex manufacturing also increases outsourcing needs.
The polyclonal antibodies segment held 8% of the market share in 2025, due to their niche applications, sustaining moderate demand. They are also being used in diagnostics and research settings. Their strong binding affinity and detection of complex antigens are also increasing their use.
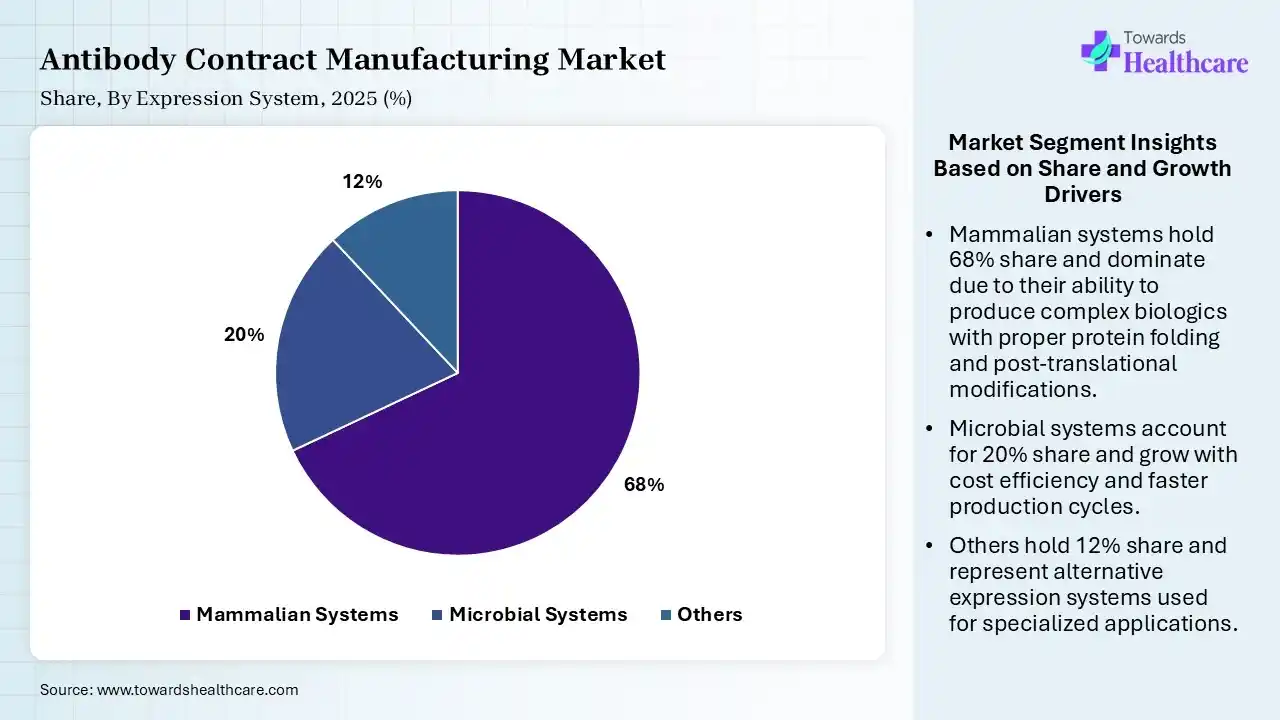
| Segment | Share 2025 (%) |
| Mammalian Systems | 68% |
| Microbial Systems | 20% |
| Others | 12% |
The Mammalian Systems Segment Dominated the Market With 68% in 2025
The mammalian systems segment held a major revenue share of 68% of the antibody contract manufacturing market in 2025, due to their increased preference for complex protein expression. High product quality and regulatory acceptance also increased their adoption rates. Advances in CHO cell engineering, boosting productivity also increased their use.
The microbial systems segment held the second-largest share of 20% of the market in 2025, due to their cost-effective production, which supports early-stage research. Faster growth cycles improve turnaround time, promoting their use. Simple production and high yield potential also drive their demand.
The others segment held 12% of the antibody contract manufacturing market share in 2025 and is expected to expand rapidly with a CAGR of 10.7% during the forecast period, due to a rise in emerging systems like insect and plant cells. Lower production costs are also attracting innovators. Additionally, they are suitable for specialized antibody formats, which drives their adoption.
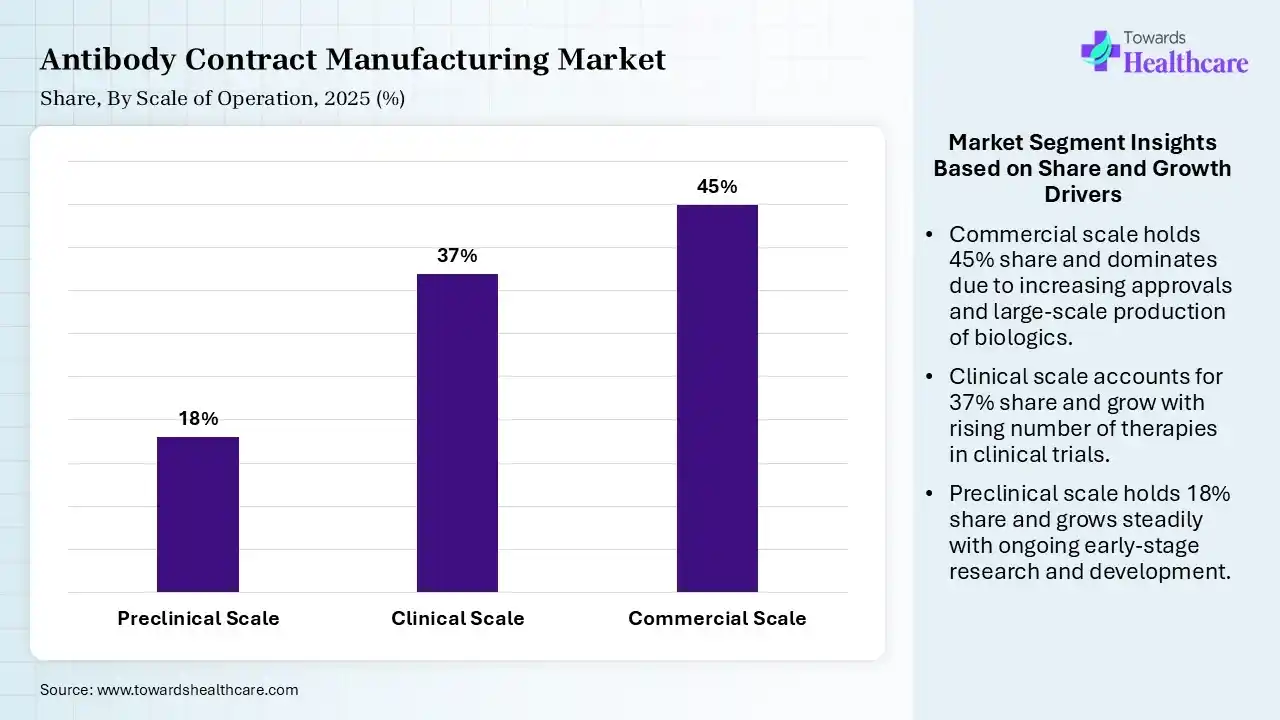
| Segment | Share 2025 (%) |
| Preclinical Scale | 18% |
| Clinical Scale | 37% |
| Commercial Scale | 45% |
The Commercial Scale Segment Dominated the Market With 45% in 2025
The commercial scale segment contributed the biggest revenue share of 45% of the antibody contract manufacturing market in 2025 and is expected to gain the highest share with a CAGR of 10.5% during the forecast period, driven by a growth in biologics approvals, which increased the demand for large-scale production. Capacity constraints also contributed to the outsourcing of the products. Long-term manufacturing contracts also supported their growth.
The clinical scale segment held the second-largest share of 37% of the market in 2025, due to growing clinical trials, which increase the production demand. Flexible manufacturing also supports multiple phases, promoting their use. Regulatory approvals accelerate scale-up needs, fueling their demand.
The preclinical scale segment held 18% of the antibody contract manufacturing market share in 2025, driven by growing early-stage R&D activities, which drive their demand. Small batch production also supports the demand for screening technologies. Increasing biotech startups also boost their usage.
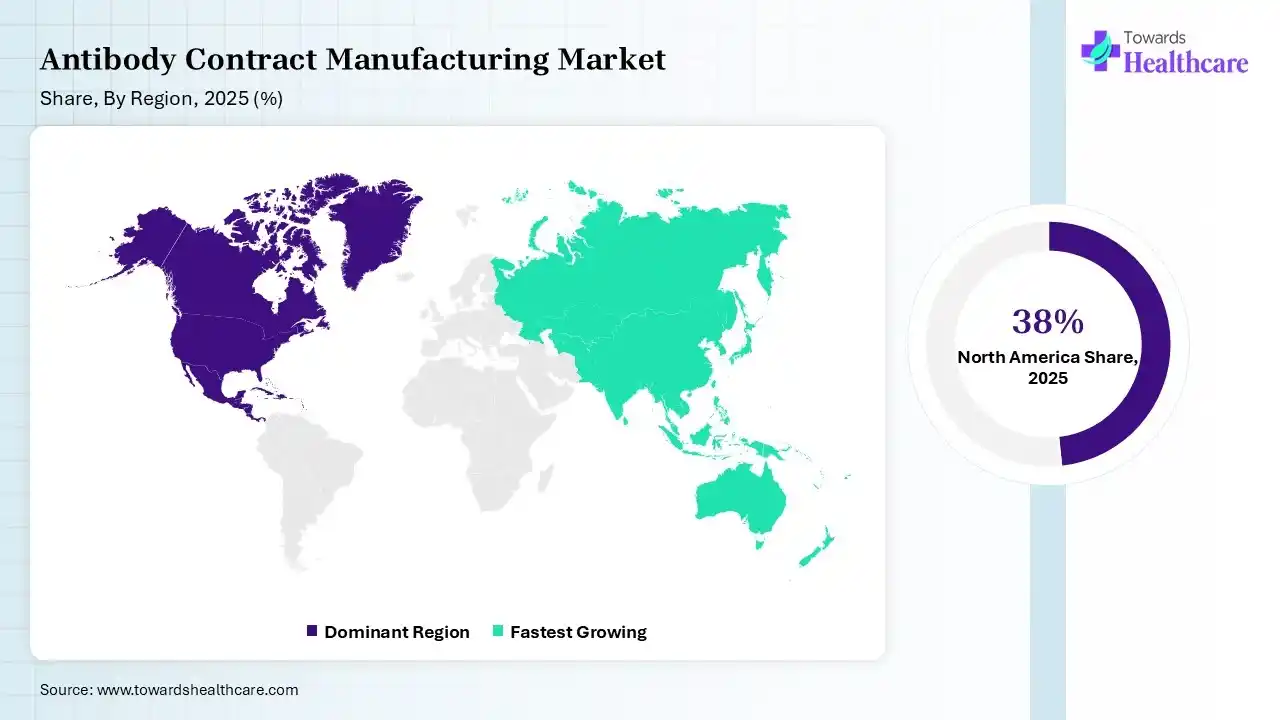
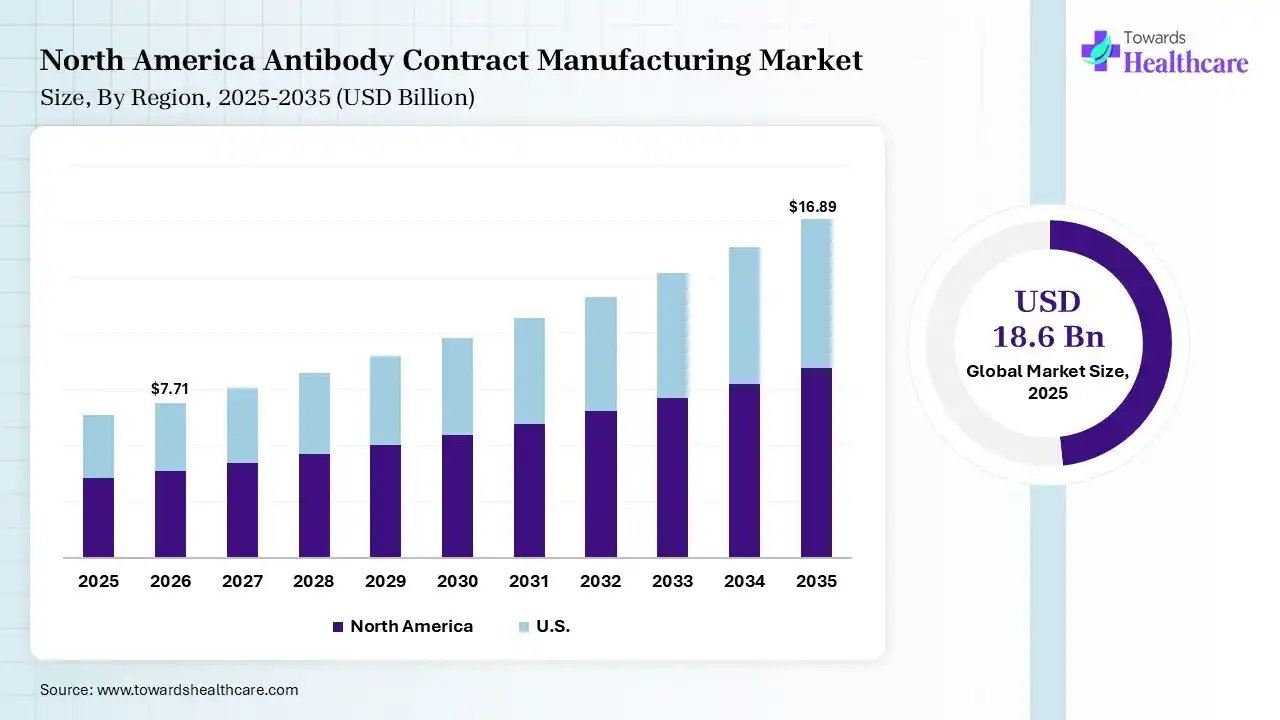
North America dominated the antibody contract manufacturing market with 38% in 2025, due to strong biopharma presence, which increased the demand for antibody contract manufacturing services. Advanced manufacturing infrastructure also supported their growth. High R&D investment also accelerated their innovation, which contributed to the market growth.
U.S. Market Trends
The large biologics market supporting outsourcing trends across the U.S. is increasing the use of antibody contract manufacturing services. The presence of leading CDMOs, biotech industries, and pharmaceutical companies is also driving their growth. The presence of a robust regulatory framework also increases their use to ensure quality standards.
Asia Pacific held 23% share of the antibody contract manufacturing market in 2025 and is expected to grow at the fastest CAGR of 11.2% during the forecast period, due to cost advantages that attract outsourcing. Expanding manufacturing capacity also supports antibody contract manufacturing growth. Rising biotech investments also accelerate their demand, enhancing the market growth.
China Market Trends
Rapid expansion of CDMO capabilities across China drives the growth of antibody contract manufacturing services. Increasing government support and demand for biologics also drive their adoption. Expanding biotech companies, clinical trials, and outsourcing trends are also increasing their use.
R&D
Formulation and Final Dosage Preparation
Patient Support and Services

| Companies | Headquarters | Antibody Contract Manufacturing Solutions |
| Lonza Group | Basel, Switzerland | Ibex Solutions |
| Samsung Biologics | Incheon, South Korea | S-DUAL |
| FUJIFILM Diosynth Biotechnologies | College Station, Tokyo | Specialized one-stop shop for ADC production |
| WuXi Biologics | Wuxi, China | WuXiBody bispecific platform |
| Siegfried Holding | Zofingen, Switzerland | Antibody drug substance and sterile drug product manufacturing |
| Thermo Fisher Scientific (Patheon) | Waltham, U.S. | Accelerator Drug Development Platform |
| AGC Biologics | Seattle, U.S. | Diverse mammalian and microbial production systems |
| Catalent, Inc. | Bridgewater, U.S. | GPEx Lightning cell line technology and SMARTag ADC platform |
| Merck KGaA (MilliporeSigma) | Burlington, Darmstadt | Specialized CDMO services for ADC bioconjugation and biosafety testing solutions |
| Boehringer Ingelheim (BioXcellence) | Ingelheim, Germany | Integrated DNA-to-finished-dosage services |
Strengths
Weaknesses
Opportunities
Threats
By Service Type
By Antibody Type
By Expression System
By Scale of Operation
By End User
By Application
By Region
May 2026
April 2026
April 2026
March 2026