May 2026

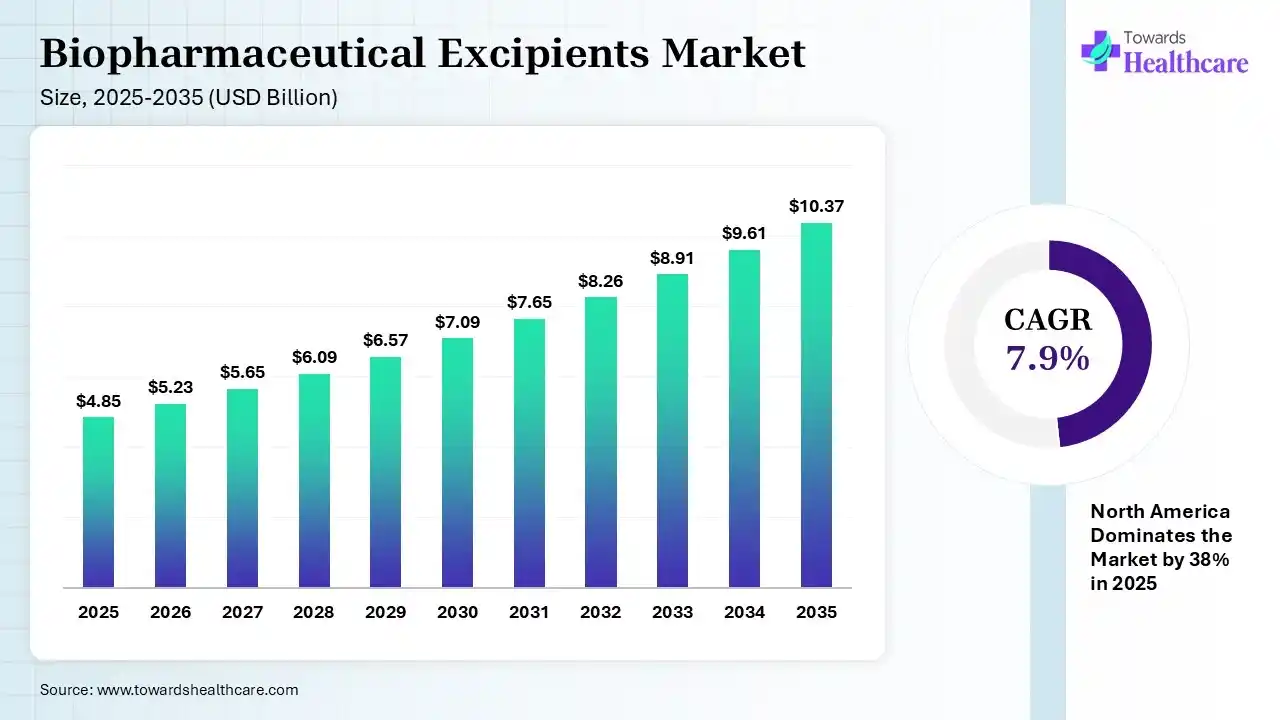
The global biopharmaceutical excipients market size was estimated at USD 4.85 billion in 2025 and is predicted to increase from USD 5.23 billion in 2026 to approximately USD 10.37 billion by 2035, expanding at a CAGR of 7.9% from 2026 to 2035. The biopharmaceutical excipients market is increasing due to it plays a significant role in safeguarding the safety, quality, and efficiency of medications. Excipients undergo severe testing to meet government standards.
Pharmaceutical excipients are materials other than the active pharmaceutical ingredient (API) that have been suitably evaluated for safety and are intentionally encompassed in a drug delivery system. Excipients allow the drug substance to be used by the patient in the right form and support the way and place of action without being active themselves. Excipients available in a wide range, from inert and simple to active and complex substances, which are problematic to characterize. Major excipients are potential toxicants at high doses in animals, however safe in humans at healing doses, involving commonly used excipients like dextran, cyclodextrins, and polyethylene glycol. Excipients used in pharmaceutical preparations not only affect the physical form but also have a major impact on the therapeutic efficacy and safety.
Incorporation of AI-driven technology in the biopharmaceutical excipients change-control technologies lessens cycle times, reducing human error rates, improves document completeness, and strengthens leading readiness. Incorporation of AI-driven technology in formulation design, smart drug delivery systems (DDSs), and maintainable pharmaceutics accentuates its role in increasing development, increasing personalization, and helping environmental responsibility. AI-driven technology, including machine learning, deep learning, Bayesian optimization, reinforcement learning, and digital twins, allows accurate prediction of significant quality attributes, reproductive discovery of excipients, and closed-loop optimization with negligible experimental input. AI-based models, involving neural networks, predict excipient-API interactions under different environmental conditions to lower degradation, like oxidation or hydrolysis.
High-Purity and Specificity:
High-purity and specific biopharmaceutical excipients are significant for the stabilization, formulation, and delivery of sensitive biological drugs, such as monoclonal antibodies, vaccines, and recombinant proteins.
Multifunctional Excipients:
Multi-functional or co-processed excipients are effective and efficient ways to enhance the production process of a drug product. These excipients provide enhanced productivity, lowered operating expenses, and enhanced product quality.
Biosimilar Expansion:
Growing requirement for tailored excipients to ensure biosimilar stability, effectiveness, and safety. Biosimilars extend beyond direct cost savings, allowing medical care systems to reallocate resources, increasing treatment access, and maintaining long-term sustainability.
| Table | Scope |
| Market Size in 2026 | USD 5.23 Billion |
| Projected Market Size in 2035 | USD 10.37 Billion |
| CAGR (2026 - 2035) | 7.9% |
| Leading Region | North America by 38% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product Type, By Functionality, By Formulation, By End User, By Region |
| Top Key Players | BASF SE, Evonik Industries AG, Merck KGaA, Roquette Frères, Avantor, Inc. |
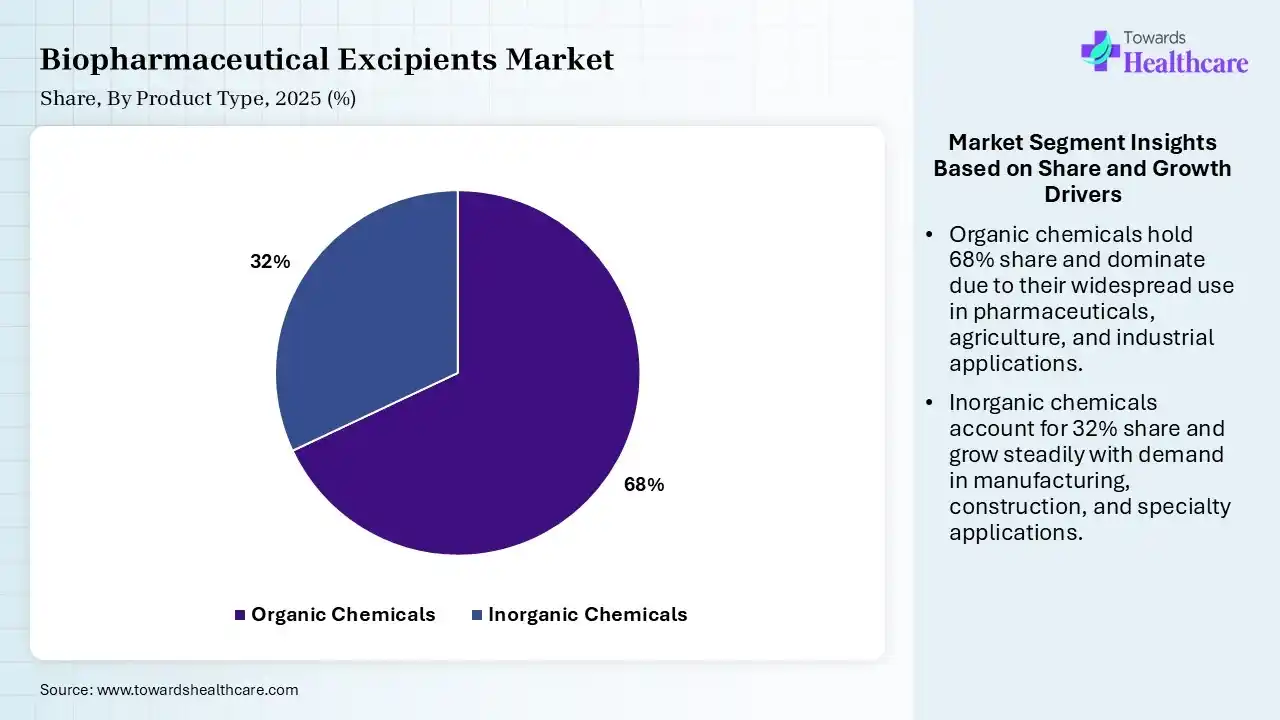
| Segment | Share 2025 (%) |
| Organic Chemicals | 68% |
| Inorganic Chemicals | 32% |
The Organic Chemicals Segment Led the Biopharmaceutical Excipients Market in 2025
The organic chemicals segment contributed the largest market share of 68% in 2025, as it is capable of creating a broad range of products with changing characteristics and functionalities. Organic chemicals serve as the significant building blocks for the combination of more multifaceted compounds. Major organic chemicals are resulting from renewable resources, contributing to ecological sustainability.
The inorganic chemicals segment held a significant share of 32% in the market, and is estimated to be the fastest-growing during the forecast period, as inorganic excipients are usually more chemically stable and inert than organic materials; they are less likely to interact negatively with the active pharmaceutical ingredient (API). Important inorganic materials, like colloidal silicon dioxide, act as outstanding anti-caking agents and glidants.
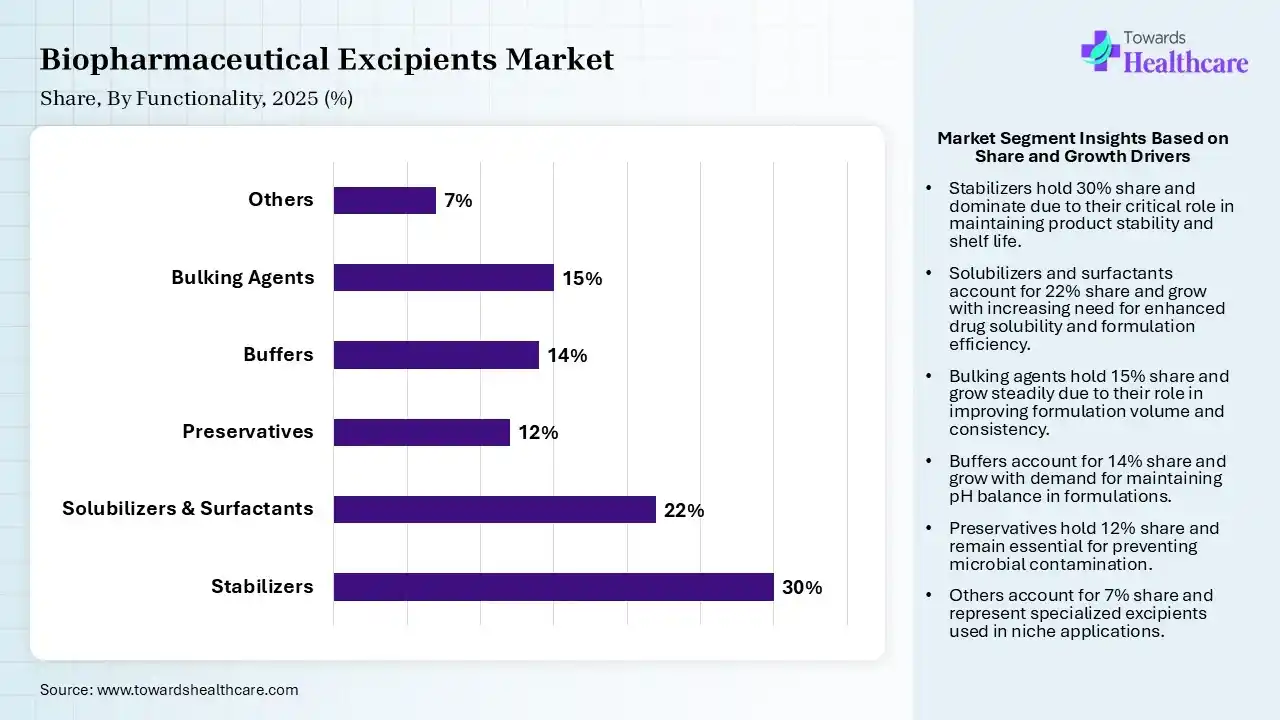
| Segment | Share 2025 (%) |
| Stabilizers | 30% |
| Solubilizers & Surfactants | 22% |
| Preservatives | 12% |
| Buffers | 14% |
| Bulking Agents | 15% |
| Others | 7% |
Stabilizers Segment Led the Biopharmaceutical Excipients Market in 2025
The stabilizers segment contributed the largest market share of 30%, stabilizers are used to support the active pharmaceutical ingredient (API) and maintain the desirable characteristics of the product until it is expelled by the patient. Stabilizers also support maintaining the physical and chemical characteristics of the product, like texture, appearance, and pH, to ensure product effectiveness and safety.
The solubilizers & surfactants segment held a significant share of 22% of the market, and is expected to witness the fastest growth of 8.6% during the forecast period, as surfactants are extremely multipurpose and strategically valuable excipients in the formulation of poorly water-soluble medicines. Solubilizers are excipients used to improve the solubility of poorly soluble medicines in the formulation. Surfactants prevent crystallisation and help to stabilise the shapeless form of a drug, and supports to enhance dissolution rate by preserving a higher concentration of dissolved drug.
The bulking agents segment held a significant share of 15% of the market, as bulking agents are substances that add fiber to stool to enhance its consistency and transit, which is significant for patients with compromised colon function. The bulking agent supports the urethra to make a watertight seal and prevent minute leaking from the bladder.
The buffers segment held a significant share of 14% of the market, as buffer solutions serve as the core factor in achieving the intended composition. Applications a buffer with higher buffer capacity offers for a more significant process deprived of requiring huge quantities of buffers.
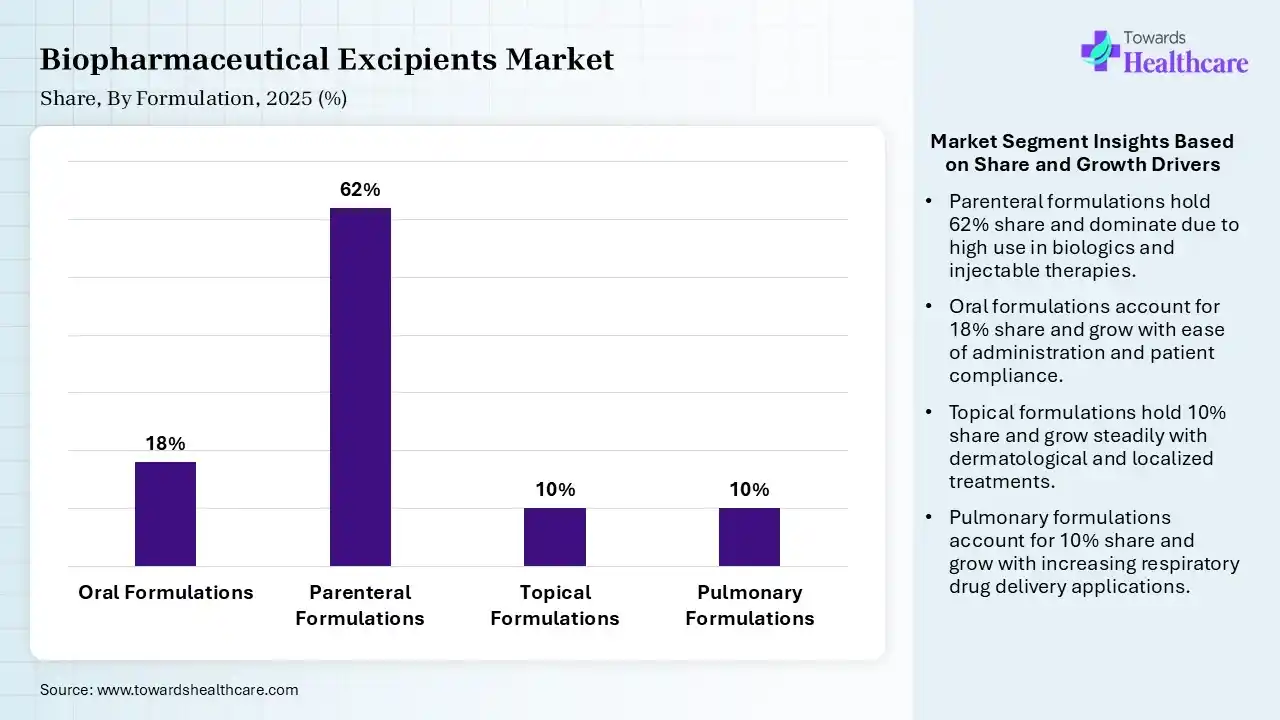
| Segment | Share 2025 (%) |
| Oral Formulations | 18% |
| Parenteral Formulations | 62% |
| Topical Formulations | 10% |
| Pulmonary Formulations | 10% |
Parenteral Formulations Segment Led the Biopharmaceutical Excipients Market in 2025
The parenteral formulations segment contributed the largest market share of 62%, and is expected to grow at the fastest rate of 8.2% CAGR during the forecast period, as parenteral formulations are appreciated in emergency conditions when a rapid onset of action is required. The parenteral route, also referred to as the injection route, is the rapid and the second most common route of drug delivery.
The oral formulations segment held a significant share of 18% in the market, as oral dosage forms play a significant role in treatment and wellness by carrying appropriate quantities of active pharmaceutical ingredients (APIs) efficiently and conveniently. Oral dosage forms are pharmaceutical formulations intended for administration via the mouth, driving the delivery of APIs in the body.
The topical formulations segment held a significant share of 10% in the biopharmaceutical excipients market, as topical delivery is specifically suited for dermatological situations. Skin diseases such as eczema, psoriasis, and acne often require direct application of medications to the affected region. Topical delivery is adaptable and suitable for different patient populations, including paediatric and geriatric patients.
The pulmonary formulations segment held a significant share of 10% in the market, as pulmonary drug delivery has the benefits of being rapid, effective, and well-targeted, with few systemic adverse effects. Pulmonary drug delivery is its capability to evade first-pass hepatic metabolism.
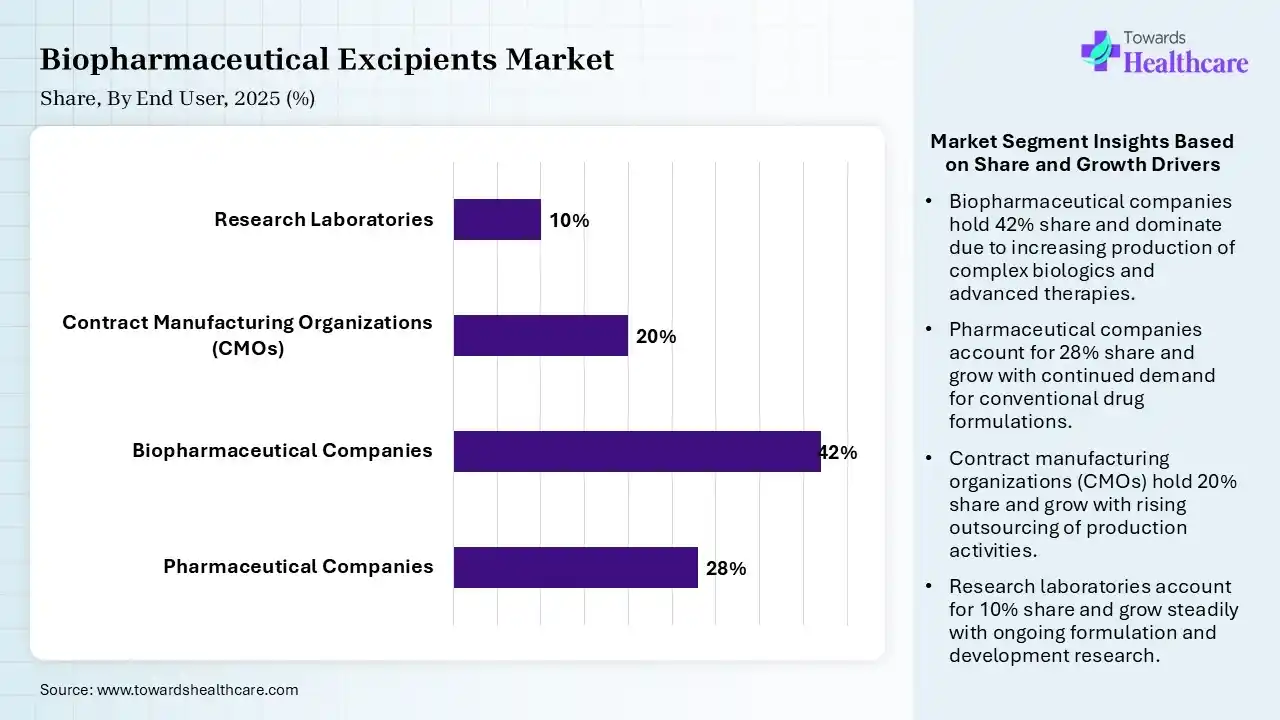
| Segment | Share 2025 (%) |
| Pharmaceutical Companies | 28% |
| Biopharmaceutical Companies | 42% |
| Contract Manufacturing Organizations (CMOs) | 20% |
| Research Laboratories | 10% |
The biopharmaceutical companies segment led the Biopharmaceutical Excipients Market in 2025
The biopharmaceutical companies segment contributed the largest market share of 42%, expected to grow at the fastest rate of 8.4% during the forecast period, as applications of the right excipients rationalize the manufacturing technology and enhance yields, thus lowering overall expenses. Pharmaceutical excipients are significant, non-active components that enhance drug formulation, stability, and patient compliance.
The pharmaceutical companies segment held a significant share of 28% in the market, as pharmaceutical excipients are significant, functional, and non-active components that ensure drug stability, efficacy, and safety. They improve bioavailability, allow controlled release, enhance manufacturability, such as binding, lubrication, and enhance patient compliance through taste-masking or coloring.
The contract manufacturing organizations (CMOs) segment held a significant share of 20% in the biopharmaceutical excipients market, as CMOs allow pharma organizations to streamline drug manufacturing and scale efficiently. Contract Manufacturing Organizations (CMOs) for excipients offer important advantages, including significantly lower capital expenditure (CapEx) for production facilities.
The research laboratories segment held a significant share of 10% in the market, as excipients play a diversity of roles in pharmaceutical manufacturing, involving modifying solubility and API bioavailability, enhancing the stability of the active ingredient in dosage forms, assisting the active ingredient in preserving preferred polymorphic forms or conformations, and acting as a disintegrant, binder, lubricant, and filler.
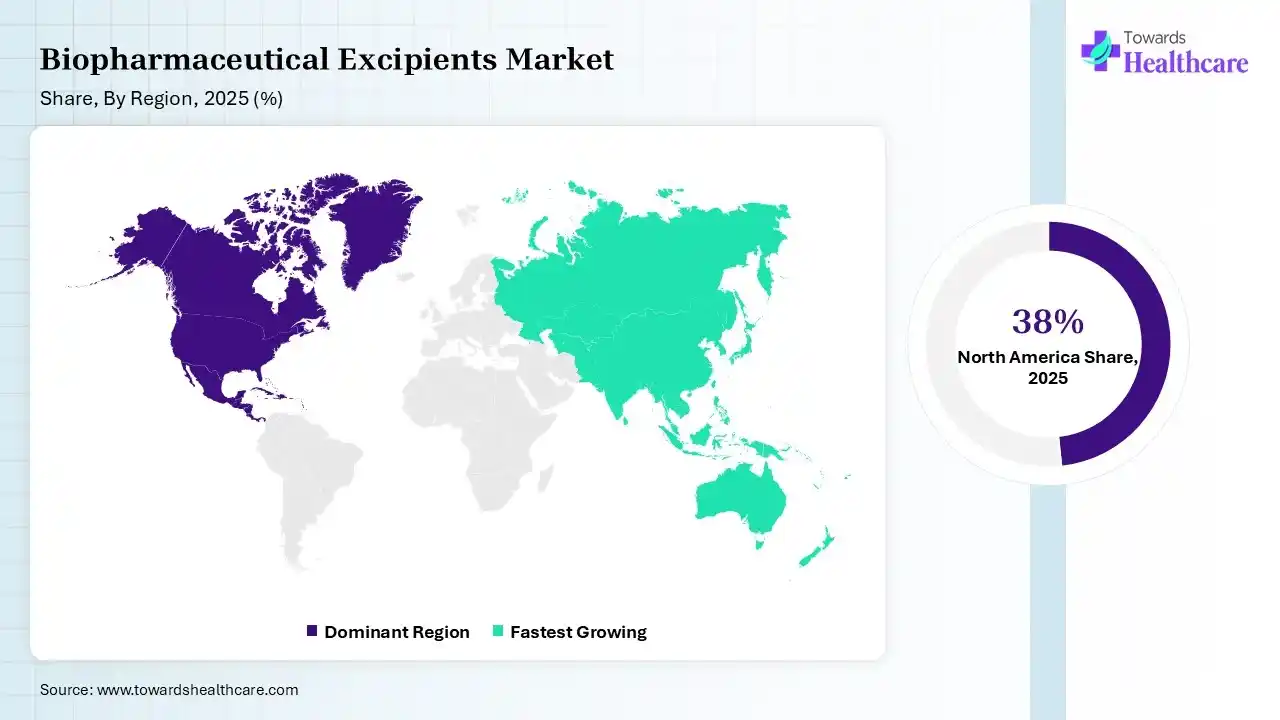
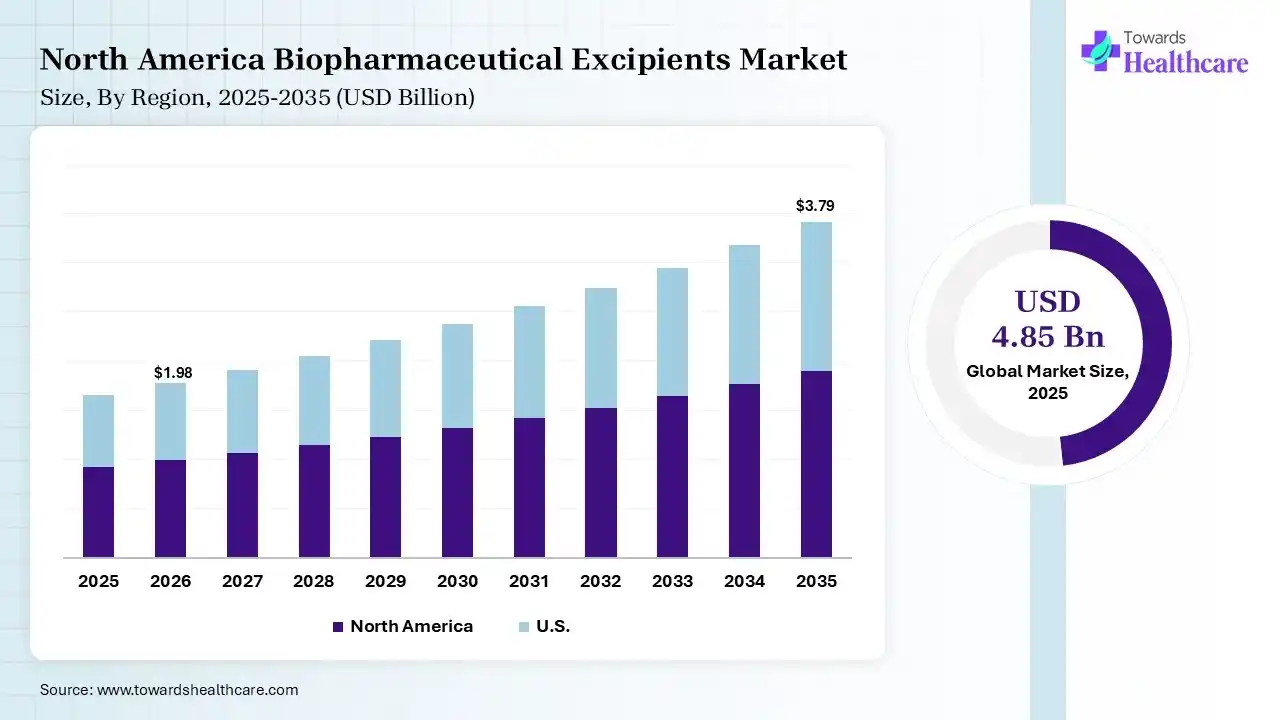
In 2025, North America dominated the biopharmaceutical excipients market with a share of 38% in 2025, due to pharmaceutical firms and a diverse network of qualified contract development and manufacturing organizations (CDMOs) and contract manufacturing organizations (CMOs). FDA is firming its focus on challenging drug products, necessitating detailed, specialized classification for excipients used in topical and transdermal preparations, which contributes to the growth of the market.
For Instance,
U.S. Market Trends
The U.S. hosts an increasing concentration of biopharmaceutical organizations worldwide, with over 1,400 biotechnology organizations and an established contract manufacturing infrastructure, helping drug development. Significant spending in advanced drug delivery technologies and collaborative research among academia and industry accelerates the advancement of novel excipients.
Asia Pacific held 24% share of the biopharmaceutical excipients market, and is expected to have the fastest growth of 9.2% CAGR during the forecast period, as this region is a leader in the next generation of production, driven by its commitment to novelty, trade, and spending. This region is evolving as a worldwide manufacturing hub as organizations diversify production to improve resilience and lower costs, which contributes to the growth of the market.
India Market Trends
Indian manufacturers have the advantages of manufacturing scale and an integrated supply chain. Indian and international contract manufacturers often have well-established export networks, allowing pharma companies. Advanced technologies like IoT, AI, robotics, and digital twins are renovating manufacturing processes.
R&D:
Manufacturing Processes:
Patient Services:

| Company | Headquarters | Latest Update |
| BASF SE | United States | In June 2025, BASF opened its novel good manufacturing practice (GMP) solution center in Wyandotte, Michigan. This spending underscores the continuing commitment of BASF to offering innovative services in the biopharma and pharmaceutical ingredients industries. |
| Evonik Industries AG | Germany | In September 2025, Evonik launched MaxiPure Polysorbate 80, a highly pure surfactant intended for injectable and biopharmaceutical applications. |
| Merck KGaA | Germany | Merck KGaA demonstrated strong, resilient growth, precisely driven by its process solutions business, which serves as a major supplier of pharmaceutical excipients. |
| Roquette Frères | France | In February 2025, Roquette launched a novel coatings platform, integrating advanced performance with eco-consciousness, intended to meet the increasing demand. |
| Avantor, Inc. | United States | Avantor, Inc. focused on increasing its biopharmaceutical abilities by investing in manufacturing infrastructure, improving its single-use systems, and launching sustainable services for biomanufacturing. |
Strengths
Weakness
Opportunities
Threat
By Product Type
By Functionality
By Formulation
By End User
By Region
May 2026
May 2026
May 2026
May 2026