April 2026

The global pulmonary function testing systems market size was estimated at USD 5.3 billion in 2025 and is predicted to increase from USD 5.66 billion in 2026 to approximately USD 10.23 billion by 2035, expanding at a CAGR of 6.8% from 2026 to 2035. The worldwide massive rise in environmental pollution, rising smoking rates & geriatric population are highly susceptible to different respiratory diseases like COPD, asthma, etc. For these cases, leading companies are putting efforts into the development of portable, handheld devices, coupled with AI solutions & home care approaches.
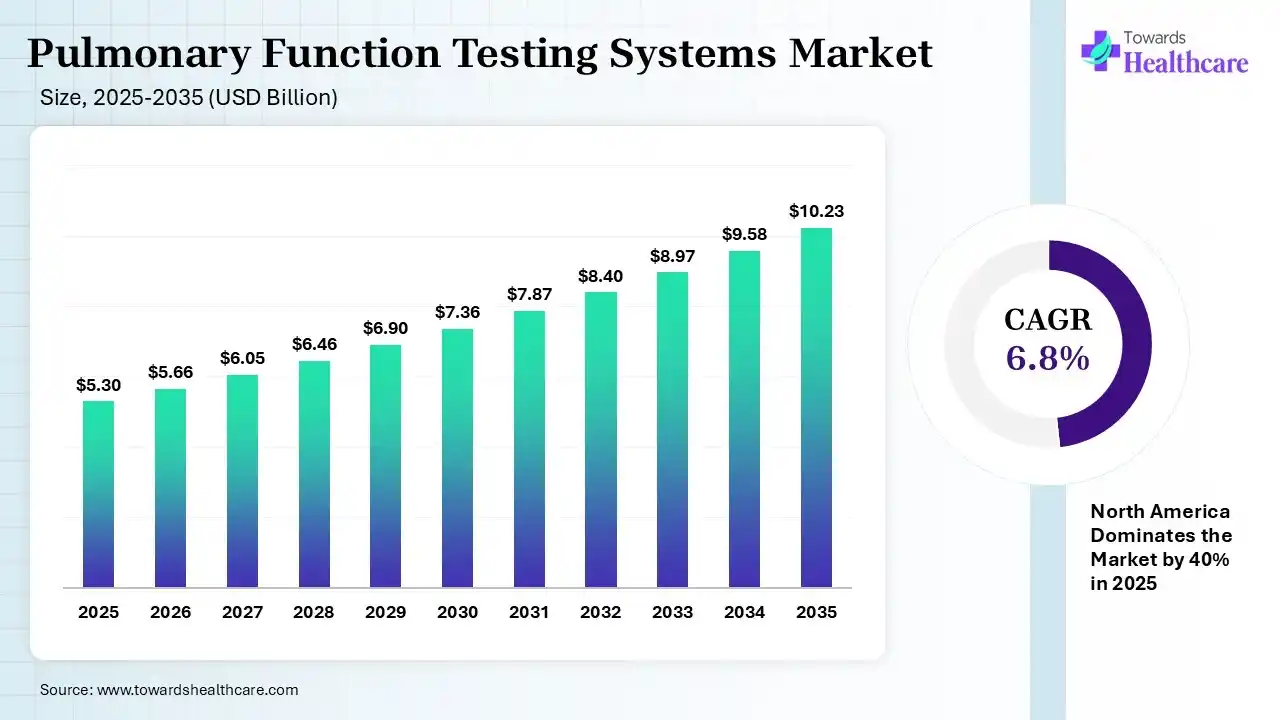
The global pulmonary function testing systems market covers non-invasive diagnostic tools to record how well lungs take in & exhale air, and how efficiently they transfer oxygen to the blood. The market progression is propelled by the growing incidence of diverse chronic respiratory diseases, rise in geriatric population, broader air pollution, & immersive, fastest adoption of portable, AI-powered diagnostic systems for prior, extensive lung function evaluation.
Primarily, the use of AI assists in ensuring tests are operated appropriately, with immediate identification of technical flaws, which offer instant feedback to patients & technicians. However, eventual advancements in AI algorithms, specifically deep learning is fostering the analysis of PFT results to categorize patterns. Due to the automated, continuous interpretations, AI lowers the diversity & subjectivity linked with manual interpretation of PFT results.
Bolstering Portability & Home Testing
Gradually, the firms are stepping toward compact, portable PFT systems to employ outside hospitals, like point-of-care, primary care, & home settings.
Spurring Digital Integration & Connectivity
Incorporation of modern systems features cloud connectivity, enabling instant data analysis, remote monitoring, & perfect electronic health record (EHR) integration.
Promoting Innovative Techniques
The globe is pushing the Forced Oscillation Technique (FOT/Resistive Oscillometry) technique to measure lung function without the need for forced breathing, which makes it favourable for children & patients with severe, restrictive, or obstructed lung diseases.
| Table | Scope |
| Market Size in 2026 | USD 5.66 Billion |
| Projected Market Size in 2035 | USD 10.23 Billion |
| CAGR (2026 - 2035) | 6.8% |
| Leading Region | North America by 40% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Type, By End-User, By Application, By Region |
| Top Key Players | MGC Diagnostics Corporations, Vyaire Medical, Inc., ndd Medizintechnik AG, COSMED Srl, Vitalograph, Schiller AG, KoKo PFT, Midmark Corporation, Geratherm Medical AG |
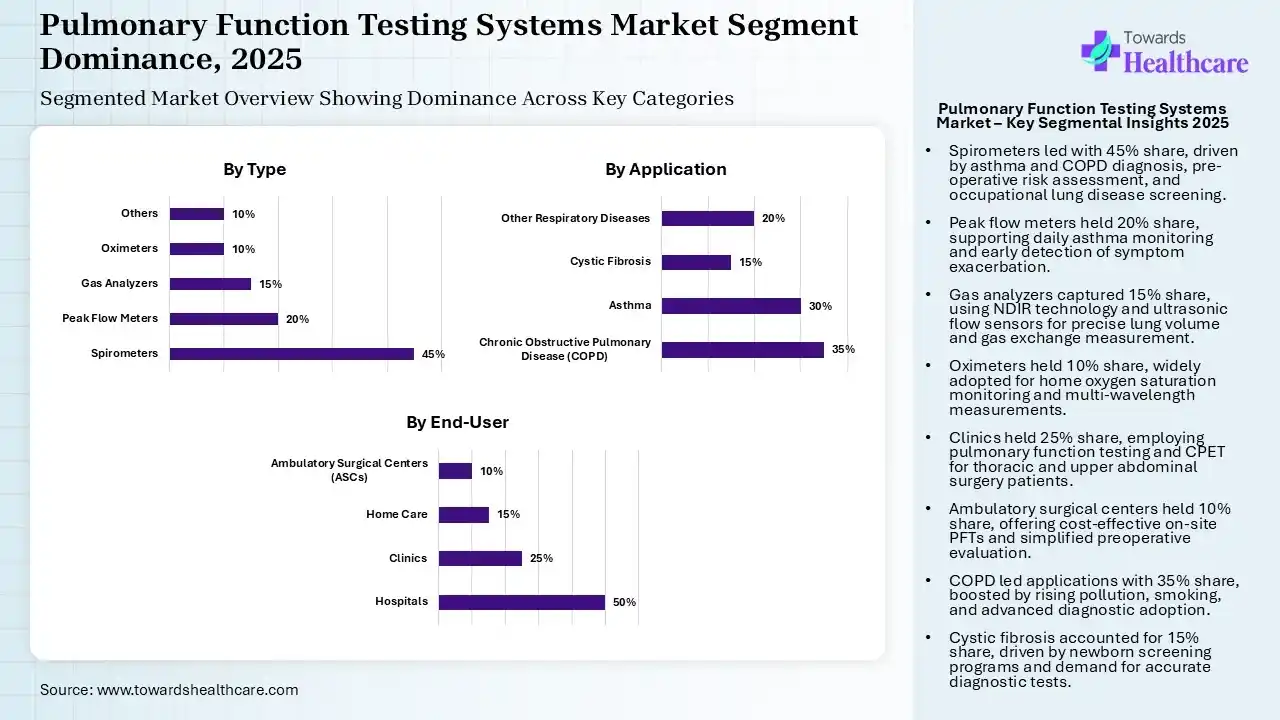
| Segment | Share 2025 (%) |
| Spirometers | 45% |
| Peak Flow Meters | 20% |
| Gas Analyzers | 15% |
| Oximeters | 10% |
| Others | 10% |
The Spirometers Segment Led the Market in 2025
The spirometers segment held 45% share of the pulmonary function testing systems market in 2025. Dominance is driven by its wider adoption in the diagnosis of issues, including asthma & COPD, with assessment of pre-operative risk, & screening for occupational lung diseases. The trend is exploring the use of ultrasonic and Venturi-based flow sensors, strict ATS/ERS 2022 standards, and leveraging Global Lung Function Initiative (GLI) reference equations.
The handheld spirometers sub-segment is predicted to expand at 7.20% CAGR. Accelerating demand for home-based monitoring, & consistent technological breakthroughs, such as Bluetooth-connected, AI-enhanced, & user-friendly devices, are fueling the overall progression.
The peak flow meters segment held the second-largest share of 20% in 2025, due to the increasing prevalence of asthma, which enables patients to monitor their asthma daily, understanding early signs of worsening symptoms, especially for those who struggle to recognize exacerbations.
The gas analyzers segment captured 15% share of the pulmonary function testing systems market. This type has extensive application in both diagnostic & research applications for pulmonary health. The market is executing Non-Dispersive Infrared (NDIR) technology for multi-gas analysis & ultrasonic flow sensors for precise, maintenance-free, & real-time study of gas exchange (DLCO) & lung volumes.
The oximeters segment held 10% share in 205, due to the surging adoption for monitoring oxygen saturation, specifically in home care. Alongside, the globe is widely using multi-wavelength technology to capture more than just oxygen saturation.
| Segment | Share 2025 (%) |
| Hospitals | 50% |
| Clinics | 25% |
| Home Care | 15% |
| Ambulatory Surgical Centers (ASCs) | 10% |
The Hospitals Segment Dominated the Market in 2025
In 2025, the hospitals segment was dominant with 50% share of the market. As per the survey, there were approximately 400 million people with COPD, & 260 million people have asthma issues, which has propelled the expansion of hospitals. Hospitals are providing assessment of respiratory risk before surgeries, often initiating from surgical areas.
The clinics segment held 25% share of the pulmonary function testing systems market. They are increasingly employing PFTs to study risks for patients undergoing thoracic or upper abdominal surgeries. Also, implementing Cardiopulmonary Exercise Testing (CPET) in the analysis of exercise-induced challenges & mortality risk in COPD patients.
Moreover, the home care segment held 15% share in 2025 & is estimated to expand at 8.2% CAGR. Catalysts are a growth in demand for remote patient monitoring, lowering hospital visits, improving affordability, & the need for daily symptom tracking. Leading companies are fostering miniaturized, user-friendly, & accurate handheld devices for precise tests at home.
The ambulatory surgical centers (ASCs) segment captured 10% share, due to the 30-50% reduction in procedure spending compared to hospitals, with emphasis on PFTs on-site that simplifies the patient experience, avoids hospital risks, & supports handling preoperative evaluation expenditures.
| Segment | Share 2025 (%) |
| Chronic Obstructive Pulmonary Disease (COPD) | 35% |
| Asthma | 30% |
| Cystic Fibrosis | 15% |
| Other Respiratory Diseases | 20% |
The Chronic Obstructive Pulmonary Disease (COPD) Segment Led the Market in 2025
In 2025, the chronic obstructive pulmonary disease (COPD) segment dominated with 35% share of the pulmonary function testing systems market. A substantial rise in environmental pollution, smoking rates, & notably rising COPD cases are fueling the demand for advanced testing approaches. Ongoing developments are embracing forced oscillation techniques, advanced imaging, & AI to foster diagnosis.
The asthma segment captured 30% share in 2025 & is predicted to expand at 7.5% CAGR. Elevating awareness is focusing on airway inflammation, reversible airflow obstruction, & Small Airway Dysfunction (SAD). Apart from home care, the market rolled out FeNO as a beneficial marker of eosinophilic airway inflammation & assists in evaluating treatment responsiveness, mainly in children.
The cystic fibrosis segment held 15% share of the pulmonary function testing systems market, due to the escalating newborn screening for cystic fibrosis, which ensures earlier diagnosis, & further demand for accurate diagnostic tests, including sweat tests & PFTs.

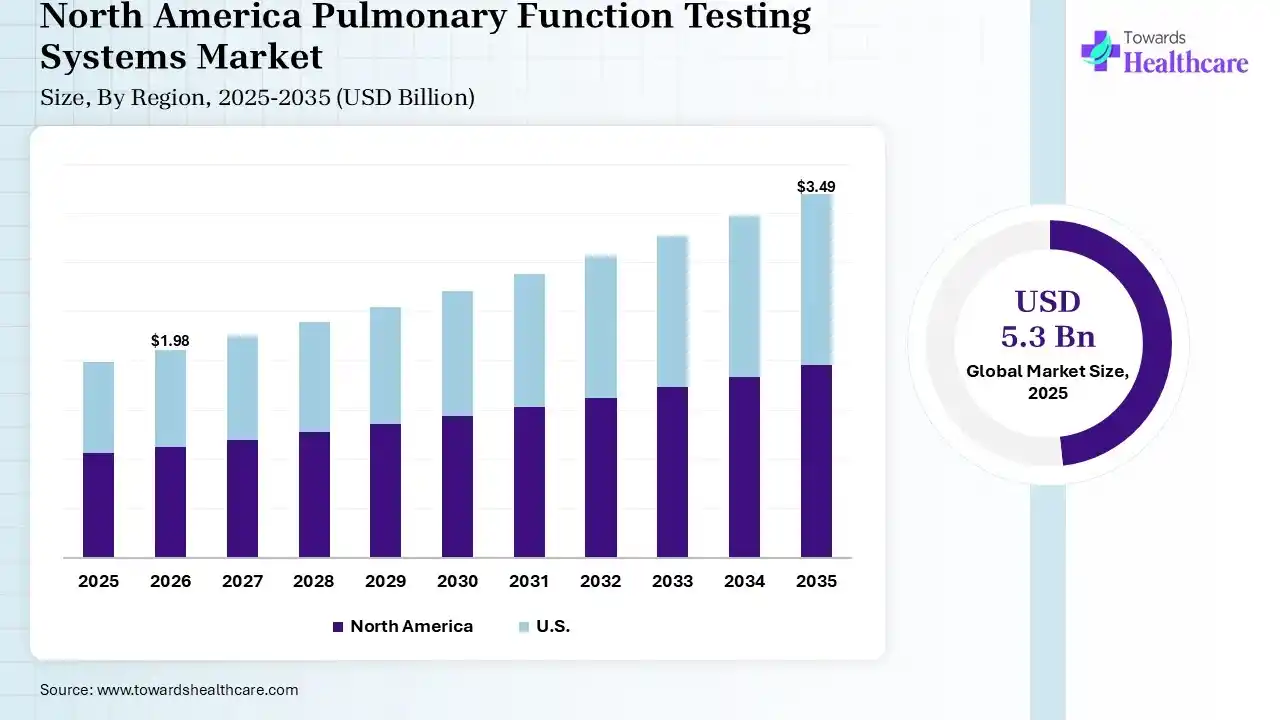
North America led with 40% share in 2025, due to the higher healthcare expenditure & possession of robust medical infrastructure. The regional systems are moving towards portable, handheld, & wireless-enabled PFT systems. Novelty comprises Canada's foremost mobile Lung Health Check PFT unit in Alberta to elevate access to screening in rural, remote, & Indigenous communities.
U.S. Market Trends
The pulmonary function testing systems market in the U.S. captured a major share of 35% & is actively enforcing calibration-free spirometers with AI quality checks & broader adoption of TeleSpirometry in VA clinics for COPD management.
For instance,
Asia Pacific held 20% of the market share and is anticipated to witness rapid growth at 7.00%, due to the surging ageing population, coupled with faster urbanization, which speeds up cases of various lung-related disorders & pushes the need for advanced systems. In addition, the APAC is boosting innovation in technologies, like compact, AI-assisted, & portable spirometers, prominently in primary care & remote areas.
China Market Trends
China’s pulmonary function testing systems market is rapidly growing at 8.0% CAGR with immersive government efforts, including the Happy Breathing Program, which accelerates the active deployment of spirometers to community healthcare centers to allow for early screening & management of chronic respiratory diseases.
In 2025, Europe held 30% share of the market, which is estimated to expand at a lucrative CAGR of 6.00% in the coming era. This has been impelling due to the enforcement of the latest European Respiratory Society (ERS)/American Thoracic Society (ATS) standards for spirometry, lung volumes, & diffusing capacity, which focuses on strict quality control & automated grading for accurate testing.
For instance,
UK Market Trends
Whereas, the UK’s pulmonary function testing systems market is anticipated to grow at 6.50% CAGR, with rigorous emphasis on the detection of issues early to prevent prospective risks. Also, the UK firms are leveraging cloud-connected PFT analytics, automated body-box operations, & handheld devices to offer clinical-grade diagnostics & also support the NHS's shift toward community-based testing.

| Company | Description |
| MGC Diagnostics Corporations | Its offerings cover Meridian Series, Ultima PFX, SpiroAir, etc. |
| Vyaire Medical, Inc. | Its portfolio facilitates Vyntus BODY, Vyntus ONE, & Vyntus SPIRO. |
| ndd Medizintechnik AG | A firm has explored TrueFlow ultrasound technology & TrueCheck technology. |
| COSMED Srl | This leader provides pulmonary function testing (PFT) systems, i.e., from portable spirometers to advanced body plethysmographs. |
| Vitalograph | Its offerings comprise VitaloPFT Series, led by ComPAS2 software& the Compact Expert workstation. |
| Schiller AG | This firm offers diversity from portable spirometers to sophisticated, stationary laboratory equipment. |
| KoKo PFT | This has unveiled the KoKo Px series for complete diagnostics & the KoKo Smart/SX1000 spirometers. |
| Midmark Corporation | A company focuses on digital spirometry solutions, especially for point-of-care testing in primary care settings. |
| MIR (Medical International Research) | It offers portable, desktop, and app-based spirometers & oximeters. |
| Geratherm Medical AG | This provides various pulmonary function testing (PFT) systems for clinical & professional applications. |
Strengths
Weaknesses
Opportunities
Threats
By Type
By End-User
By Application
By Region
April 2026
March 2026
March 2026
February 2026