April 2026

The global pharmaceutical microbiology QC testing market size was estimated at USD 4.07 billion in 2025 and is predicted to increase from USD 4.6 billion in 2026 to approximately USD 13.84 billion by 2035, expanding at a CAGR of 13.02% from 2026 to 2035.
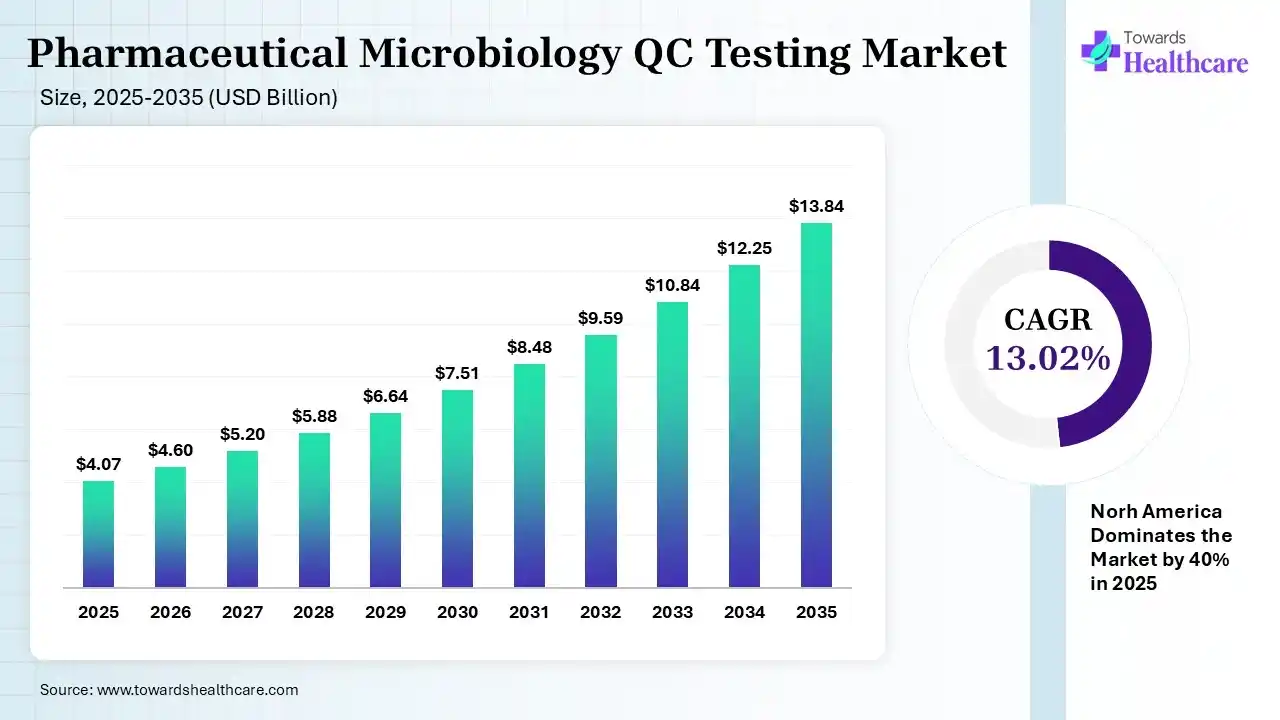
The market is growing steadily, fueled by stricter drug safety regulations, increasing biologics production, and the adoption of advanced rapid testing methods, enabling faster, accurate detection of microbial contamination in pharmaceutical products globally.
Pharmaceutical microbiology QC testing is the process of analyzing drugs, biologics, and raw materials to detect, quantify, and control microbial contamination, ensuring product safety, sterility, and compliance with regulatory standards. The pharmaceutical microbiology QC testing market is growing due to increasing demand for safe and sterile drugs, rising biologics and vaccine production, and stricter regulatory requirements. Adoption of rapid and automated microbial testing methods, along with the need to minimize contamination risks and ensure product quality, is driving market expansion globally. Growing awareness of drug safety and technological advancements further fuels the market’s steady growth.
| Year | Adoption of Rapid Microbiological Methods (%) |
| 2023 | 28% |
| 2024 | 35% |
| 2025 | 40% |
Artificial intelligence (AI) can significantly transform the market by enabling faster and more accurate microbial detection, predictive analysis, and automated data management. AI-powered systems reduce human error, enhance efficiency, and support regulatory compliance. Integration of AI in QC processes accelerates testing timelines, optimizes resource utilization, and improves overall product safety, making it a key driver for innovation and market growth in pharmaceutical microbiology testing.
| Table | Scope |
| Market Size in 2026 | USD 4.6 Billion |
| Projected Market Size in 2035 | USD 13.84 Billion |
| CAGR (2026 - 2035) | 13.02% |
| Leading Region | North America by 40% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product, By Test Type, By Technique, By Application, By Region |
| Top Key Players | Thermo Fisher Scientific, Inc., Merck KGaA, BD, bioMérieux, Danaher Corporation, Sartorius AG, Rapid Micro Biosystems, Charles River Laboratories |
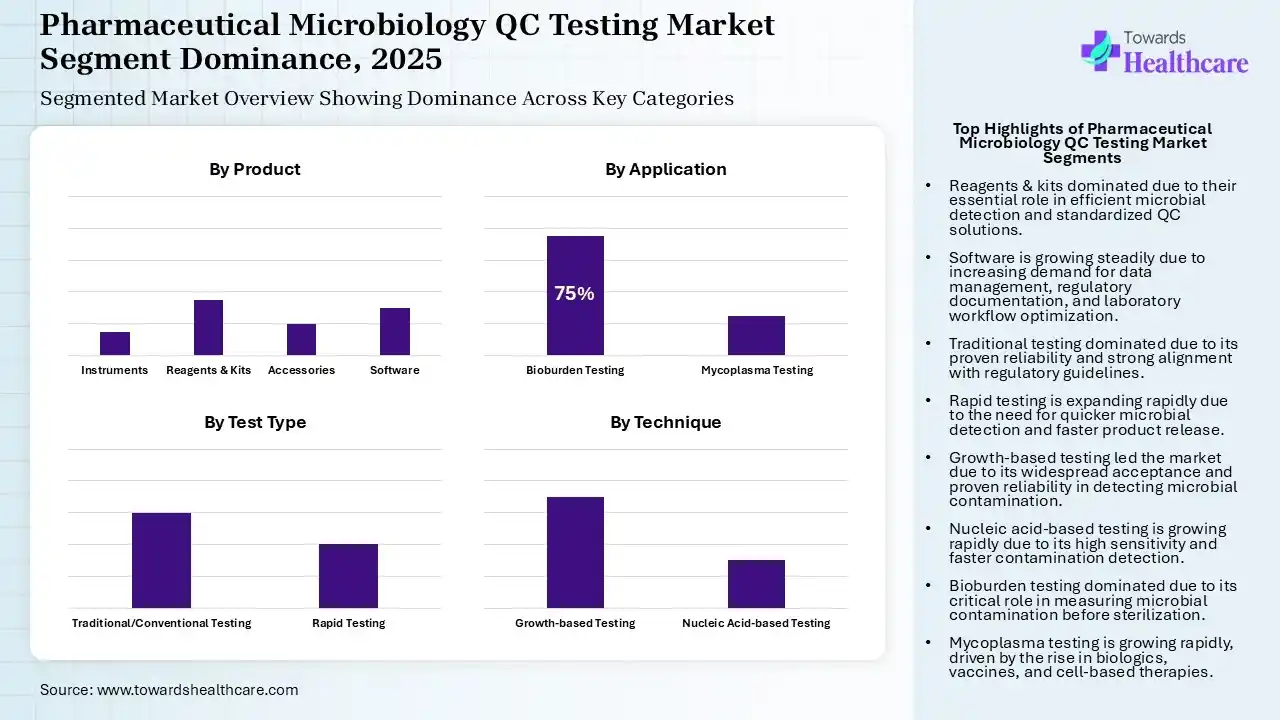
Why Did the Reagents & Kits Segment Dominate in the Market in 2025?
The reagents & kits segment dominated the pharmaceutical microbiology QC testing market by 15% in 2025 due to their essential role in accurate and efficient microbial detection. Ready-to-use kits simplify testing, reduce preparation time, and ensure consistent results, while reagents support a wide range of assays. Growing demand for standardized, reliable, and rapid QC solutions across pharmaceutical and biologics manufacturing is driving drone storage adoption, maintaining the segment’s market leadership.
Software
The software segment is expected to grow at a notable rate in the market due to the increasing need for efficient data management, regulatory documentation, and laboratory workflow optimization. Advanced laboratory software helps track testing results, ensure compliance with quality standards, and improve traceability. As pharmaceutical companies focus on digital record-keeping and streamlined quality control operations, demand for specialized microbiology testing software is expected to rise steadily.
What Made the Traditional Testing Segment Dominant in the Market in 2025?
The traditional testing segment dominated the pharmaceutical microbiology QC testing market by 60% in 2025 due to its widespread acceptance, proven reliability, and strong alignment with established regulatory guidelines. Methods such as culture-based microbial detection remain the industry standards for sterility and contamination testing. Many pharmaceutical manufacturers continue using these techniques because they are well-validated, cost-effective, and trusted by regulatory authorities, ensuring consistent quality control across drug manufacturing processes.
Rapid Testing
The rapid testing segment is expected to grow at the fastest CAGR due to the increasing need for quicker microbial detection and faster product release in pharmaceutical manufacturing. Rapid methods significantly reduce testing time compared to traditional culture techniques while maintaining accuracy. Growing demand for efficient quality control, higher production of biologics, and the need to minimize contamination risks are encouraging pharmaceutical companies to adopt rapid microbiological testing solutions.
How did the Growth-Based Testing Segment Dominate the Market in 2025?
The growth-based testing segment dominated the pharmaceutical microbiology QC testing market by 70% in 2025 because these methods are widely accepted and validated for detecting microbial contamination in pharmaceutical products. Techniques such as culture-based testing are strongly supported by regulatory guidelines and are commonly used for sterility, microbial limits, and environmental monitoring. Their reliability, established protocol, and continuous cost-effectiveness make growth-based methods a preferred choice in pharmaceutical quality control laboratories.
Nucleic Acid-Based Testing
The nucleic acid-based testing segment is expected to grow at the fastest CAGR due to its ability to detect microorganisms quickly and with high sensitivity. These techniques identify microbial genetic material directly, reducing testing time compared to conventional methods. Increasing demand for rapid contamination detection, rising biologics manufacturing, and the need for precise microbial identification are encouraging pharmaceutical companies to adopt nucleic acid-based testing technologies in quality control processes.
How Did the Bioburden Testing Segment Dominate the Market in 2025?
The bioburden testing segment dominated the pharmaceutical microbiology QC testing market by 75% in 2025 because it is essential for measuring microbial contamination levels in raw material, components, and finished products before sterilization. Pharmaceutical manufacturing relies heavily on bioburden testing to maintain product quality and comply with strict regulatory standards. Its routine use in manufacturing processes and environmental monitoring made it a critical step in ensuring safe and contamination-free pharmaceutical production.
Mycoplasma Testing
The mycoplasma testing segment is expected to grow at the fastest CAGR due to the rising production of biologics, vaccines, and cell-based therapies, which are highly vulnerable to mycoplasma contamination. Detecting these microorganisms is critical for ensuring product safety and quality. Increasing regulatory focus on contamination control and the need for reliable detection methods in biopharmaceutical manufacturing are further driving the growing demand for mycoplasma testing in quality control processes.
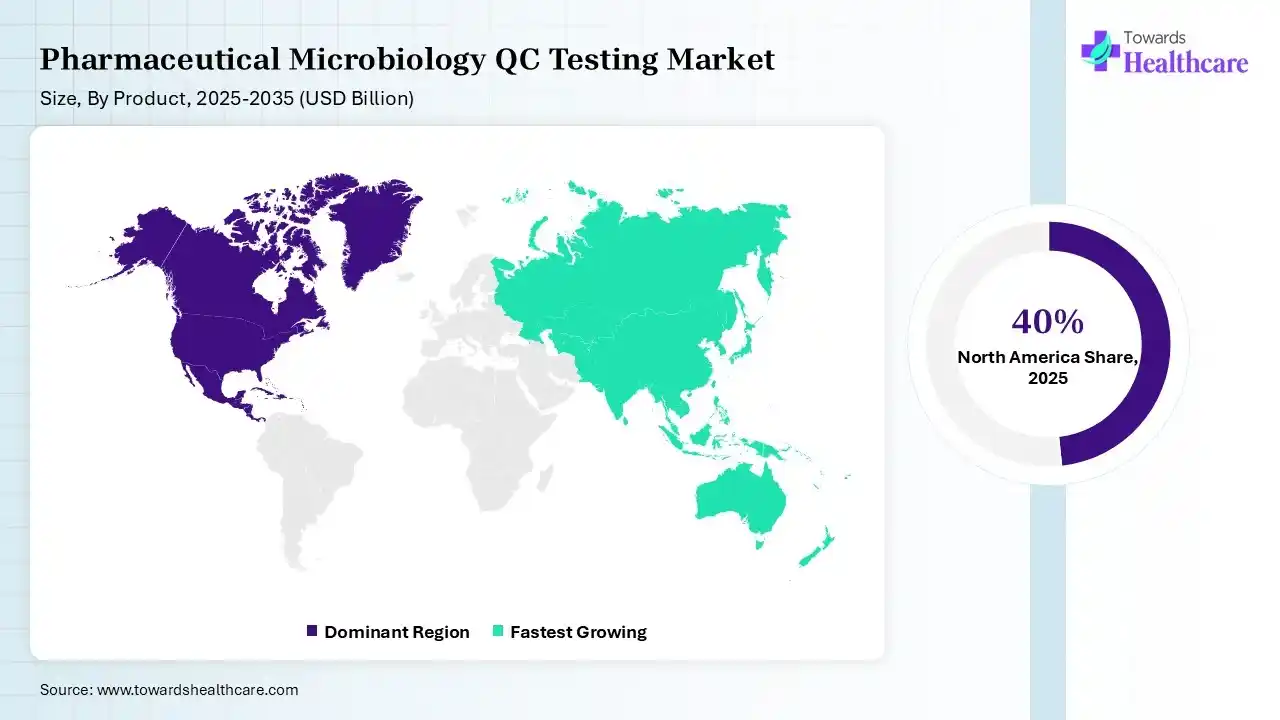
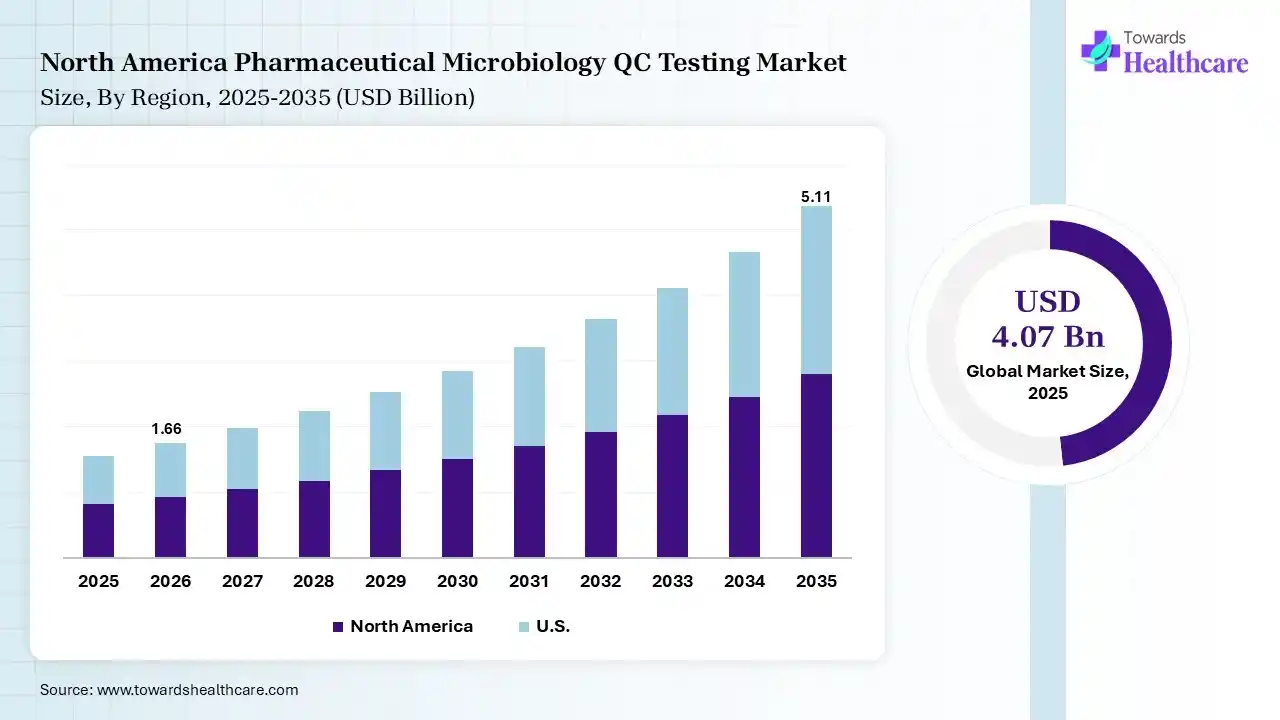
North America dominated the pharmaceutical microbiology QC testing market by 40% in 2025 due to its well-established pharmaceutical and biotechnology industries, strict regulatory standards, and strong focus on drug quality and safety. The presence of leading pharmaceutical companies, advanced laboratory infrastructure, and significant investments in research and development further supported the region's leadership in microbiological quality control testing.
Growth Momentum in the U.S. Pharmaceutical Microbiology QC Testing Market
The U.S. market is expanding due to the strong presence of pharmaceutical and biotechnology companies, increasing production of biologics and vaccines, and strict regulatory oversight for drug safety. Growing investment in advanced laboratory technologies and quality control processes further supports market growth, as manufacturers focus on ensuring contamination-free production and maintaining high standards of pharmaceutical product quality.
Asia Pacific is anticipated to grow at the fastest CAGR in the pharmaceutical microbiology QC testing market due to the rapid expansion of pharmaceutical manufacturing, increasing investments in biotechnology, and growing contract manufacturing activities. Improving healthcare infrastructure, rising demand for high-quality medicine, and strengthening regulatory standards across countries such as China, India, and South Korea are further accelerating the adoption of microbiological quality control testing in the region.
India Emerging as a Rapidly Expanding Market
India is expected to grow at the fastest CAGR in the pharmaceutical microbiology QC testing market due to the rapid expansion of its pharmaceutical manufacturing sector and increasing global demand for generic drugs and vaccines. Rising investments in biotechnology, growing contract research and manufacturing services, and improving regulatory standards for drugs are further driving the adoption of advanced microbiological quality control testing across pharmaceutical production facilities.
Europe is anticipated to grow at a notable rate in the pharmaceutical microbiology QC testing market due to its strong pharmaceutical manufacturing base and strict regulatory standards for drug safety. Increasing investments in biologics and vaccine development, along with growing adoption of advanced microbiological testing, are supporting market expansion. Additionally, the presence of leading pharmaceutical companies and well-established research infrastructure further contributes to regional growth.
UK Emerging as a Growing Hub for Microbiology QC Testing
The United Kingdom is expected to grow at a significant CAGR in the pharmaceutical microbiology QC testing market due to its strong biopharmaceutical research ecosystem and increasing investment in advanced drug development. Rising biologics production, expanding clinical research activities, and strict regulatory standards for drug quality are encouraging pharmaceutical companies to adopt advanced microbiological quality control testing across manufacturing and research facilities.
Research and development in pharmaceutical microbiology QC is centered on designing quicker and more precise microbial detection techniques. New approaches such as ATP bioluminescence, PCR-based testing, and metabolic heat monitoring help accelerate testing processes and reduce dependence on slower culture-based methods.
Key Players: Thermo Fisher Scientific, Merck KGaA, bioMérieux, Danaher Corporation, Sartorius AG, and Charles River Laboratories.

| Companies | Headquarters | Offerings |
| Thermo Fisher Scientific, Inc. | Massachusetts, U.S. | Provides microbial culture media, sterility testing kits, PCR-based detection systems, laboratory reagents, and environmental monitoring solutions used in pharmaceutical quality control laboratories. |
| Merck KGaA | Darmstadt, Germany | Supplies microbiology media, filtration and sterility testing tools, reagents, and analytical solutions that help pharmaceutical manufacturers detect microbial contamination. |
| BD | New Jersey, U.S. | Develops microbiological culture media, sample preparation products, and laboratory instruments used for microbial identification and contamination monitoring. |
| bioMérieux | France | Offers rapid microbial detection platforms, identification systems, and environmental monitoring technologies designed for pharmaceutical manufacturing and quality testing. |
| Danaher Corporation | D.C., U.S. | Through life-science subsidiaries, it provides filtration technologies, molecular diagnostic systems, and laboratory tools that support microbial testing and contamination control. |
| Sartorius AG | Göttingen, Germany | Provides sterility testing systems, microbial monitoring equipment, filtration products, and laboratory instruments used in pharmaceutical microbiology labs. |
| Rapid Micro Biosystems | Massachusetts, U.S. | Specializes in automated rapid microbial detection platforms that help pharmaceutical companies shorten testing timelines and improve quality control efficiency. |
| Charles River Laboratories | Massachusetts, U.S. | Offers contract microbiology testing services, including sterility, endotoxin, and microbial identification testing for pharmaceutical and biologics manufacturing. |
By Product
By Test Type
By Technique
By Application
By Region
April 2026
April 2026
April 2026
April 2026