
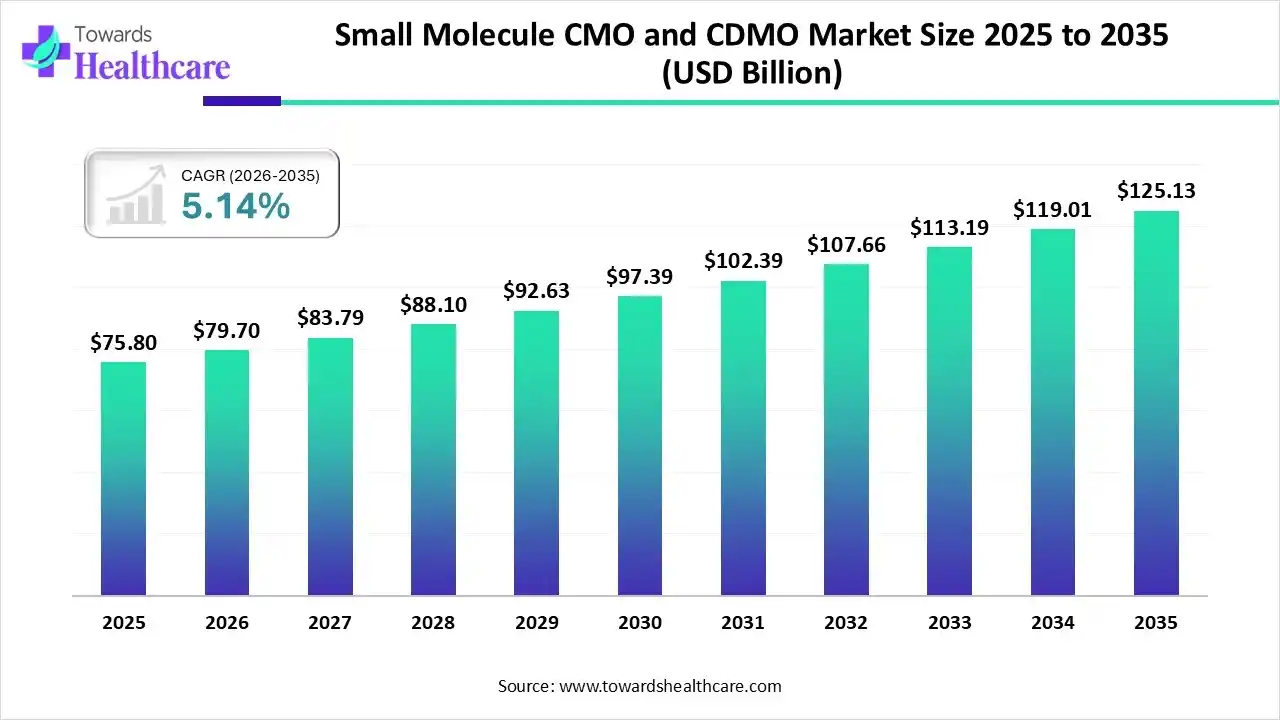
The global small molecule CMO and CDMO market size was estimated at USD 75.8 billion in 2025 and is predicted to increase from USD 79.7 billion in 2026 to approximately USD 125.13 billion by 2035, expanding at a CAGR of 5.14% from 2026 to 2035.
The growing advancements in the complex small molecule development are increasing the use of small molecule CMO and CDMO services. Increasing AI integration, R&D activities, outsourcing trends, and new launches are also promoting the market growth.
The small molecule CMO and CDMO market is driven by a surge in targeted oncology therapies, complexities of high potency APIs (HPAPIs), and the transition from simple contract manufacturing towards integrated lifecycle partnership. The small molecule CMO and CDMO refer to the companies responsible for the manufacturing and/or development of the low molecular weight chemical compounds. The pharmaceutical and biotech companies rely on them for faster production, enhanced scalability, cost efficiency, and regulatory compliance.
The use of AI in the small molecule CMO and CDMO market is increasing for process optimization and quality control. It also helps in process monitoring, equipment maintenance, batch failure prediction, automated data analysis, and deviation investigation. At the same time, it also offers risk assessment, inventory optimization, and automated documentation, which is increasing its adoption rates.
Advancements in Specialty API
The growing interest in oncology and rare diseases is increasing the demand for small molecules, which is driving the development of specialty APIs that are chemically complex, encouraging collaborations with small molecule CMOs and CDMOs.
Growing Outsourcing Trends
The growing small molecule R&D activities and expanding biotech startups are increasing the demand for the CMO and CDMO to leverage their R&D and manufacturing services along with regulatory expertise.
Technological Innovations
The growing technological advancements are increasing the adoption of digital technologies, delivery technologies, and continuous manufacturing, promoting sustainable chemistry and automation in small molecule CMO and CDMO.
Infographics
| Table | Scope |
| Market Size in 2026 | USD 79.7 Billion |
| Projected Market Size in 2035 | USD 125.13 Billion |
| CAGR (2026 - 2035) | 5.14% |
| Leading Region | North America by 40% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product Type, By Drug Type, By Therapeutic Area (Application), By Stage Type, By Region |
| Top Key Players | Lonza, Thermo Fisher Scientific, Catalent, WuXi AppTec, Samsung Biologics, Piramal Pharma Solutions, Cambrex, Recipharm, Siegfried Holding |
| Segments | Shares % |
| Active Pharmaceutical Ingredients (API) | 61.8% |
| Finished Drug Products (FDP) | 38.2% |
Why Did the Active Pharmaceutical Ingredients (API) Segment Dominate in the Market in 2025?
The active pharmaceutical ingredients (API) segment led the small molecule CMO and CDMO market with an approximate 61.8% share in 2025, as APIs are the "core" of the small molecules. The growing demand for generic drugs also increased their outsourcing. Their complex synthesis and demand for expertise also increased the collaborations with CMOs and CDMOs.
Finished Drug Products (FDP)
The finished drug products (FDP) segment is expected to show the highest growth during the upcoming years, due to growing outsourcing of complex formulation, such as sterile injectables and specialized oral solids. Moreover, growing complex development processes are also increasing the demand for small molecule CMO and CDMO services.
| Segments | Shares % |
| Innovators | 55% |
| Generics | 45% |
How Innovators Segment Dominated the Market in 2025?
The innovators segment held a dominant position in the small molecule CMO and CDMO market with a share of approximately 55% in 2025, due to a robust pipeline of new molecular entities (NMEs) in oncology and neurology. Additionally, their complex production and regulatory compliance also increased the demand for small molecule CMO and CDMO.
Generics
The generics segment is expected to show the fastest growth rate during the upcoming years, due to a massive wave of patent expirations (the "patent cliff") and global government pressure for affordable medicine. This is increasing their production volume, driving the collaborations with the small molecule CMOs and CDMOs.
| Segments | Shares % |
| Oncology | 39% |
| Neurology/CNS | 27% |
| Others | 34% |
Which Therapeutic Area (Application) Type Segment Held the Dominating Share of the Market in 2025?
The oncology segment held the dominating share of approximate 39% in the small molecule CMO and CDMO market in 2025, due to growth in the small molecule inhibitors and targeted therapies for cancer. Moreover, the growth in their incidence rates and investments also encouraged the small molecule CMO and CDMO partnerships.
Neurology/CNS
The neurology/CNS segment is expected to expand rapidly during the predicted time, due to a surge in R&D for Alzheimer’s, Parkinson's, and chronic pain, which is creating high demand for specialized small molecule manufacturing. Additionally, faster regulatory approvals are also increasing the use of small molecule CMO and CDMO services.
| Segments | Shares % |
| Clinical Phase (Phase I–III) | 56% |
| Commercial Manufacturing | 44% |
What Made the Clinical Phase (Phase I–III) the Dominant Segment in the Market in 2025?
The clinical phase (phase I–III) segment held the largest revenue share of approximate 56% in the small molecule CMO and CDMO market in 2025, due to the vast number of drugs in the mid-to-late stage "funnel" which required constant CDMO support. Moreover, growth in outsourcing trends also increased the collaboration with small molecule CMO and CDMO.
Commercial Manufacturing
The commercial manufacturing segment is expected to show the highest growth with the highest CAGR during the predicted time, due to growth in the small molecules transition from trials to the market, and scaling up of high-volume commercial production. The small molecule CMO and CDMO were also preferred for their affordable services and regulatory expertise.
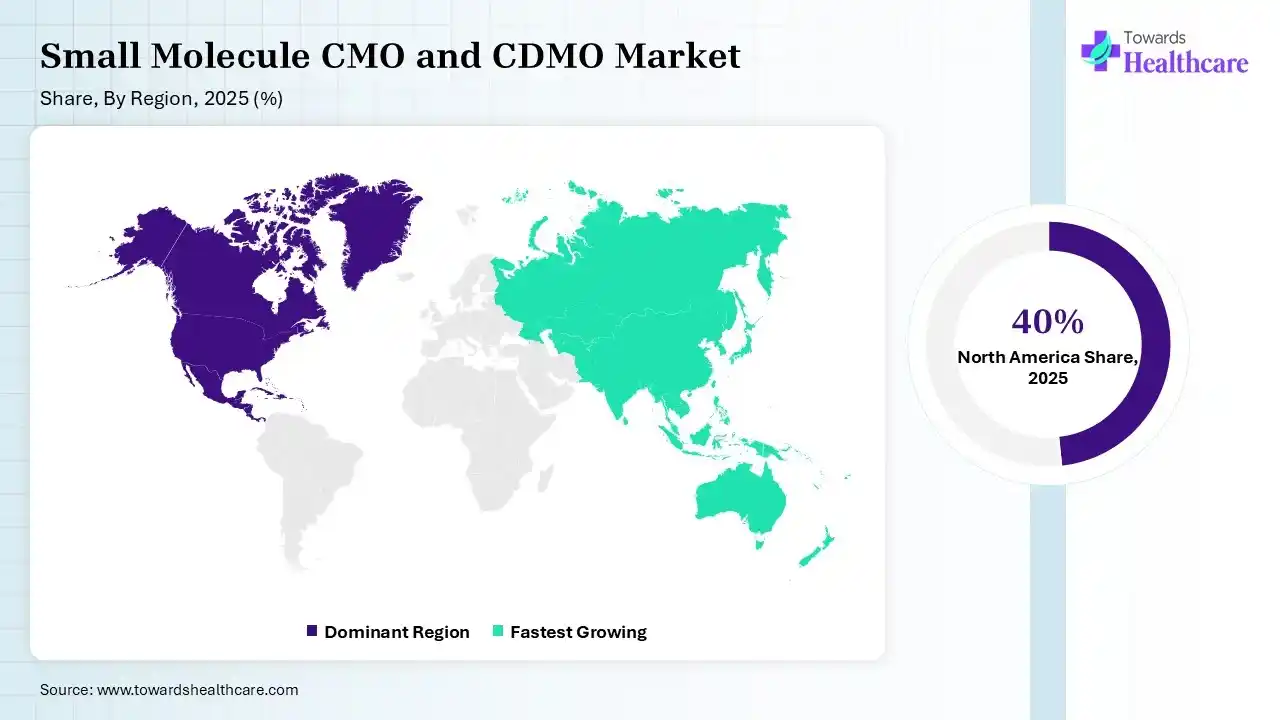
North America dominated the small molecule CMO and CDMO market with approximate 40% in 2025, due to the presence of advanced pharmaceutical and biotech industries along with well-established CMO and CDMO infrastructure. The growth in the R&D investments and stringent regulations also increased the collaboration with the small molecule CMO and CDMO, which contributed to the market growth.
U.S. Market Trends
The growth in the R&D investments in the U.S. is driving the adoption of advanced technologies and collaborations with small molecule CMO and CDMO to accelerate the development of complex small molecules. Moreover, the presence of regulatory and technical expertise is also attracting the companies.
Asia Pacific is expected to host the fastest-growing small molecule CMO and CDMO market during the forecast period, due to the presence of affordable manufacturing services, which encouraging outsourcing of small molecules. The rapid expansion of the industries and a growing demand for generics are also increasing the use of small molecule CMOs and CDMOs, enhancing the market growth.
China Market Trends
China is experiencing a rapid expansion in the pharmaceutical industry and adoption of advanced technologies, which is accelerating R&D activities. This is increasing the demand for small molecule CMO and CDMO for the development of affordable small molecule generic products.
Europe is expected to grow significantly in the small molecule CMO and CDMO market during the forecast period, due to growing focus on specialty drugs, which is increasing the collaborations with the small molecule CMO and CDMO. The presence of robust pharmaceutical companies and growing innovations is also increasing their demand, which is driving the market growth.
UK Market Trends
The presence of a pharmaceutical and biotechnology hub in the UK is increasing the R&D activities focused on small molecule development, which are leading to new partnerships among the small molecule CMOs and CDMOs. Stringent regulations and government policies are also encouraging their use.
R&D
Clinical Trials and Regulatory Approvals
Patient Support and Services
| Companies | Headquarters | Solutions |
| Lonza | Basel, Switzerland | Full-lifecycle CDMO services |
| Thermo Fisher Scientific | Waltham, U.S. | End-to-end development services |
| Catalent | Somerset, U.S. | Provides soft gel technologies and clinical supply services |
| WuXi AppTec | Shanghai, China | Comprehensive chemistry services |
| Samsung Biologics | Incheon, South Korea | Offer advanced chemical synthesis and integrated manufacturing |
| Piramal Pharma Solutions | Mumbai, India | Integrated services |
| Cambrex | East Rutherford, U.S. | Provides small-molecule API development, generic APIs, and specialized drug substance manufacturing |
| Recipharm | Stockholm, Sweden | High volume oral solid production and inhalation drug delivery expertise |
| Siegfried Holding | Zofingen, Switzerland | Complex chemical synthesis, sterile filling, and finished dosage forms of small molecule injectables |
| Euroapi | Paris, France | Developed APIs and intermediates of generic and specialty molecules |
Strengths
Weaknesses
Opportunities
Threats
By Product Type
By Drug Type
By Therapeutic Area (Application)
By Stage Type
By Region