April 2026

The U.S. contraceptive market size was estimated at USD 10.8 billion in 2025 and is predicted to increase from USD 11.52 billion in 2026 to approximately USD 20.66 billion by 2035, expanding at a CAGR of 6.7% from 2026 to 2035. The U.S. contraceptive market is growing due to its support for individuals to avoid unwanted pregnancies and plan births, which lowers pregnancy-related health challenges, specifically for adolescent girls.
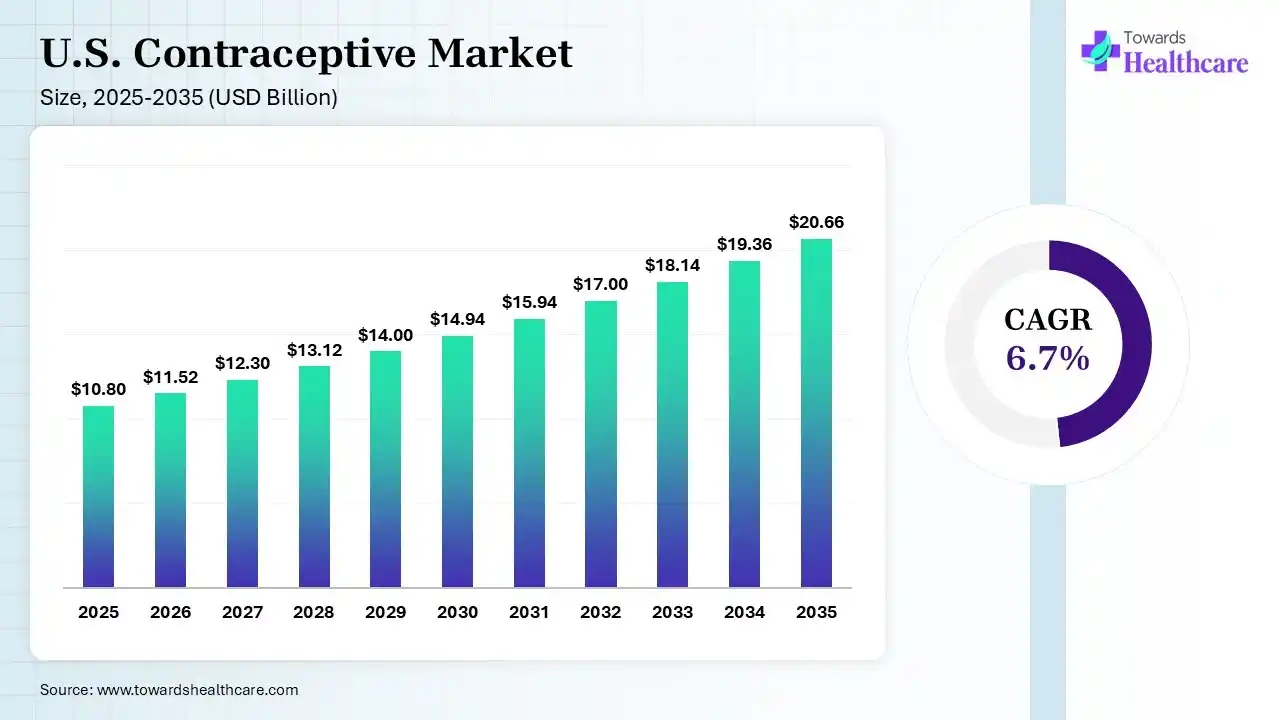
The U.S. contraceptive market is growing, as efficient contraception advantages both mothers and children through lessening morbidity and mortality, enhancing the social and economic status of women, and improving the relationship of the mother with her offspring. The United States has rates of unintentional pregnancy and abortion more approaching those of an emerging country than those of other industrialized nations. In the U.S., couples, 40% select male or female sterilization as their technique of contraception. Novel contraceptive methods probably to become available in the US soon will increase the number of efficient, reversible contraceptive choices for US couples. Increasing access to a wide variety of methods will increase contraceptive applications.
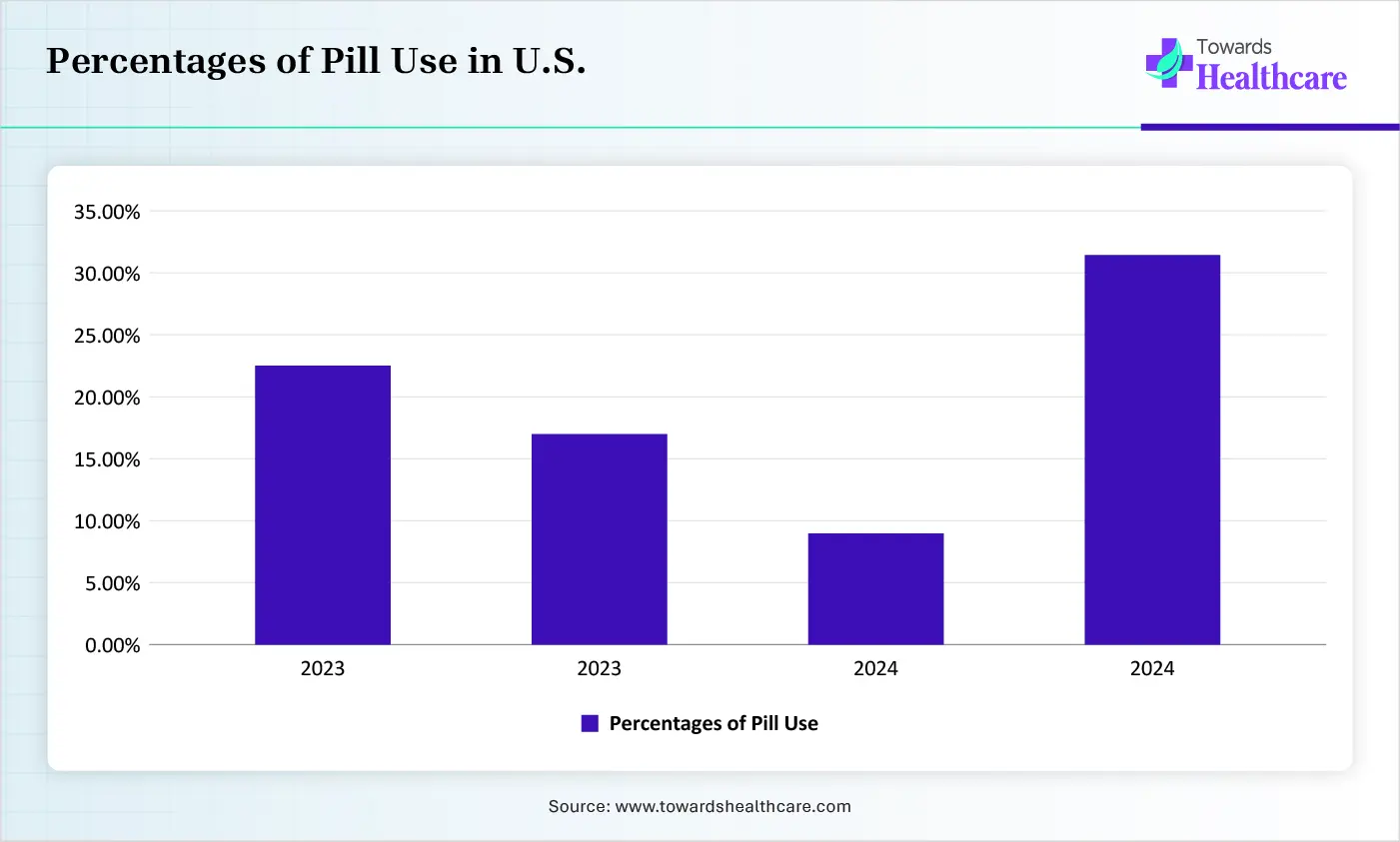
| Age Group | Percentages of Pill Use |
| 15-24 | 22.4% |
| 25 -34 | 16.9% |
| 35-44 | 8.7% |
| Older | 31% |
AI-based technology integrated into sexual reproductive health, involving enhancing care, offering high-quality contraception and infertility solutions, removing unsafe abortions, and allowing the prevention and treatment of sexually transmitted infections. AI-based technology analyzes to understand trends in sexual behavior, contraceptive use, and attitudes toward reproductive health. Government spending in AI-based initiatives, through collaboration of public-private organizations and intended funding streams, improves sustainability and encourages broad adoption.
Growth of Non-Hormonal Options:
Non-hormonal contraceptives are technologies that prevent pregnancy without intervening in the body’s hormonal cycle. Because they do not contain synthetic hormones like progestin or estrogen, they are used in women who cannot use hormonal contraceptives due to health reasons.
Rise in Male Contraception R&D:
The advancement of male contraception has been massively researched over the past few years. Different hormonal and non-hormonal technologies are being developed to offer better contraception with negligible or no disadvantages.
Dual-Protection Focus on Contraception:
A dual contraceptive technology is the utilization of any modern contraceptive technology along with male or female condoms, which decreases sexually transmitted diseases (STDs) and unwanted pregnancy.
| Table | Scope |
| Market Size in 2026 | USD 11.52 Billion |
| Projected Market Size in 2035 | USD 20.66 Billion |
| CAGR (2026 - 2035) | 6.7% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product Type, By Gender, By Age Group, By Distribution Channel |
| Top Key Players | Agile Therapeutics, Inc., Piramal Healthcare & Sun Pharma, HRA Pharma, Bayer AG, Pfizer Inc. |
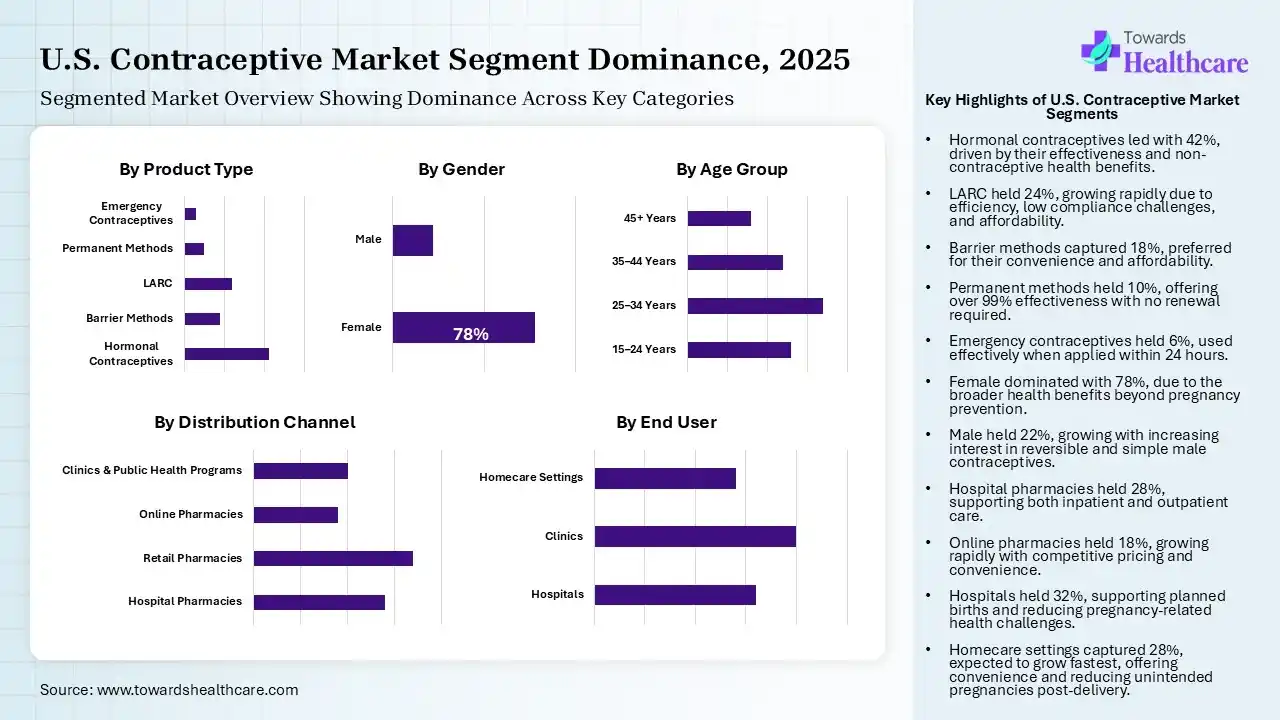
Hormonal Contraceptives Segment Led the U.S. Contraceptive Market in 2025
| Segment | Share 2025 (%) |
| Hormonal Contraceptives | 42% |
| Barrier Methods | 18% |
| LARC | 24% |
| Permanent Methods | 10% |
| Emergency Contraceptives | 6% |
The hormonal contraceptives segment contributed the largest market share of 42% in 2025, as hormonal contraceptives are efficient in preventing pregnancy; they contain a progestin, which inhibits ovulation. Hormonal contraceptives have been shown to offer different noncontraceptive advantages. Hormonal contraceptives reduce the risks of endometrial cancer, colorectal cancer, ovarian cancer, and osteoporosis.
The LARC segment held the second-largest share of 24% of the market in 2025 and is expected to grow at the fastest CAGR of 8.40% during the forecast period, as LARC offers an efficient process of contraception with lower compliance challenges by the consumer and prevents unwanted pregnancies and their challenges. These technologies are the most active modern contraceptive processes for preventing unintended pregnancy. They are long-acting, reliable, safe, and affordable.
Whereas the barrier methods segment held a notable share of 18% in the U.S. contraceptive market in 2025, as barrier contraception stops pregnancy by stopping the male's sperm from coming into contact with the female's ovum. Barrier contraceptives are affordable and convenient. Many women use them to avoid health challenges related to hormonal contraceptives.
The permanent methods segment held a notable share of 10% of the market in 2025, as permanent processes are more than 99% efficient at preventing pregnancy. Unlike short-term or long-acting processes, they never need to be renewed or replaced. Permanent contraception includes a surgical procedure that reliably prevents future pregnancies.
The emergency contraceptives segment held a notable share of 6% of the market in 2025, as emergency contraception is efficient when applied early, with only 3% of women becoming pregnant if used within 24 hours of unprotected sexual intercourse. Emergency contraception (EC) prevents up to over 95% of pregnancies when taken within 5 days after intercourse.
Female Segment Led the U.S. Contraceptive Market in 2025
| Segment | Share 2025 (%) |
| Female | 78% |
| Male | 22% |
The female segment contributed the largest market share of 78% in 2025, as female contraceptives provide significant benefits beyond pregnancy prevention, including highly efficient family planning, regulation of menstrual cycles, lower menstrual pain (dysmenorrhea), and reduced symptoms of PMS/PMDD.
The male segment held the second largest share of 22% of the U.S. contraceptive market in 2025 and is estimated to achieve 7.50% CAGR during 2026-2035, as male contraceptives lower the number of ejaculated fertile sperm inflowing the vagina to levels that reliably prevent fertilization. They have the strength to be simple to apply and reversible. Testosterone functions as a male contraceptive by suppressing the secretion of LH and FSH from the pituitary through negative feedback.
The Retail Pharmacies Segment held the Largest Share in the Market in 2025
| Segment | Share 2025 (%) |
| Hospital Pharmacies | 28% |
| Retail Pharmacies | 34% |
| Online Pharmacies | 18% |
| Clinics & Public Health Programs | 20% |
The retail pharmacies segment contributed the largest market share of 34% in 2025, as retail pharmacies play an important role in medical providers by offering convenient access to medications, a broad range of products, and significant additional solutions. Retail pharmacies are a significant part of medical care systems and, in major countries, are responsible for dispensing a huge proportion of medical care products.
The hospital pharmacies segment held the second largest share of 28% of the U.S. contraceptive market in 2025, as hospital pharmacies supply drugs to other entities performing medical actions, inpatient facilities offering 24-hour medical services, or outpatient health solutions.
The clinics & public health programs segment held a notable share of 20% of the market in 2025, as the family planning (KB) program aims to enhance the welfare of a family by limiting the birth rate or number of children born. These programs not only avert unintended pregnancies, lowering maternal and infant mortality rates, but also offer significant, comprehensive care.
The online pharmacies segment held a notable share of 18% of the market in 2025 and is expected to be the fastest-growing segment with 9.10% CAGR, as online pharmacies often provide competitive pricing, discounts, and generic substitutes, leading to potential price savings for patients. This affordability is predominantly relevant for those with chronic conditions who need long-term medicine.
The Clinics Segment held the Largest Share of the Market in 2025
| Segment | Share 2025 (%) |
| Hospitals | 32% |
| Clinics | 40% |
| Homecare Settings | 28% |
The clinics segment contributed the largest market share of 40% in 2025, as enhanced contraceptive use, lowered unintended pregnancies, better birth spacing, increased prenatal care, lowered incidence of preterm and low-birthweight offspring, increased breastfeeding, increased HPV vaccinations, and cervical tumor screenings.
The hospitals segment held the second-largest share of 32% of the U.S. contraceptive market in 2025. It supports patients in avoiding unintended pregnancies and planned births, and it lowers pregnancy-related health challenges, specifically for adolescent girls. Efficient contraception advantages both mothers and children by lowering morbidity and mortality, enhancing the social and economic status of women.
Whereas the homecare settings segment held a notable share of 28% in the market in 2025 and is estimated to grow at the highest CAGR of 7.80% during the upcoming period, as this type of care increases the effectiveness of contraceptive use, which could lower the incidence of unintended pregnancies, and if provided after delivery, could decrease short-interval unintended pregnancy. Home visits remove the requirement for transport to clinics, decreasing both the expenses and time challenges.
R&D:
Manufacturing Processes:
Patient Services:
| Company | Latest Update |
| Agile Therapeutics, Inc. | Agile Therapeutics announced that the USPTO has issued three novel patents with claims associated with its contraceptive patch technology. |
| Piramal Healthcare & Sun Pharma | In May 2025, Piramal Consumer Healthcare unveiled a novel campaign for I-pill Daily, reaffirming its commitment to women's health, dignity, and well decision-making. |
| HRA Pharma | The company is mainly focusing on increasing the availability of its flagship OTC daily birth control pill, Opill. |
| Bayer AG | In December 2025, Bayer announced the start of its Phase III study SUNFLOWER, an investigational study assessing the effectiveness and safety of 52mg levonorgestrel-releasing intrauterine system Mirena for the management of nonatypical endometrial hyperplasia (NAEH) in women. |
| Pfizer Inc. | In December 2025, the pharma giant announced that the Food and Drug Administration approved its new safety warning label for Depo-Provera, Pfizer’s birth control injection, which has been a subject of controversy as women in the country have been using it to develop meningiomas. |
Strengths
Weaknesses
Opportunities
Threats
By Product Type
By Gender
By Age Group
By Distribution Channel
April 2026
April 2026
April 2026
April 2026