
The U.S. small molecule innovator API CDMO market size was estimated at USD 9.37 billion in 2025 and is predicted to increase from USD 9.94 billion in 2026 to approximately USD 16.85 billion by 2035, expanding at a CAGR of 6.04% from 2026 to 2035.
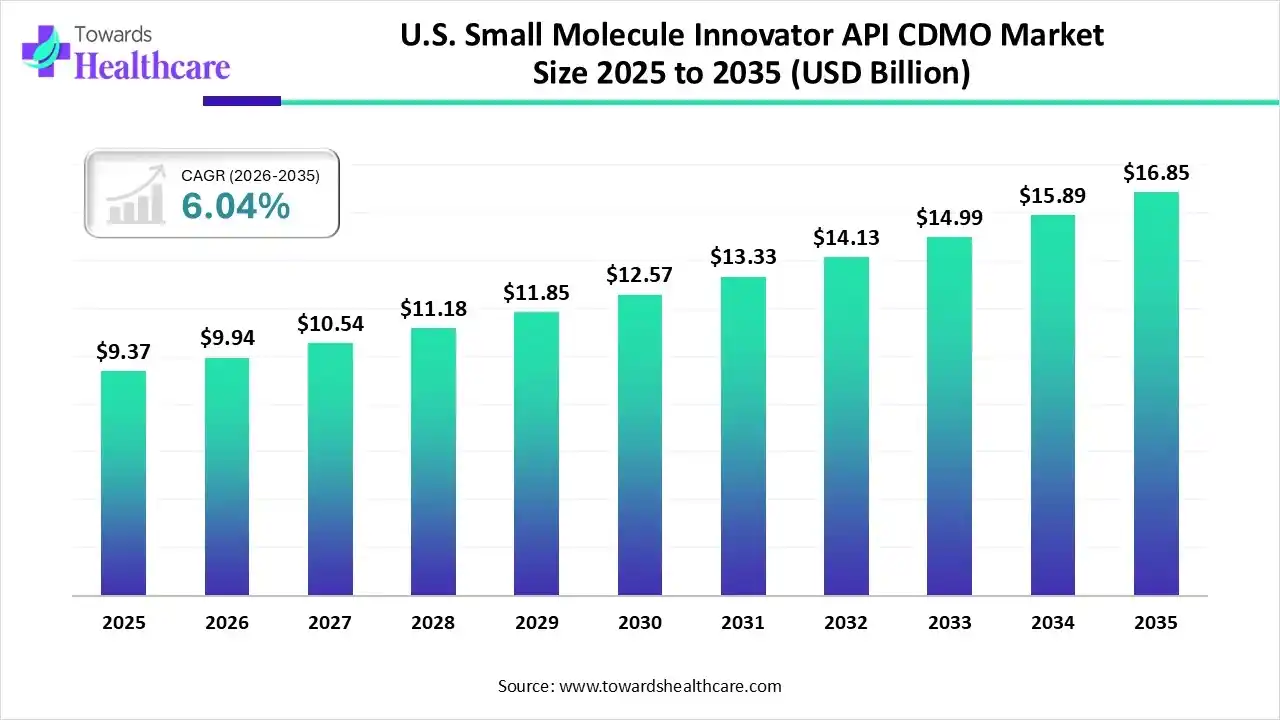
The U.S. small molecule innovator API CDMO market is growing as CDMO supports multifaceted activities, including active pharmaceutical ingredient (API) advancement, formulation development, government compliance, clinical trial management, process development, upscaling, and commercial manufacturing.
The U.S. small molecule innovator API CDMO market is growing because CDMOs assist in rationalization processes, which enable biotech and pharma organizations to not only cut expenses but also enhance their healthcare sector entry timelines, while complying with stringent government requirements. Contract Development and Manufacturing Organization (CDMO) plays a significant role in the medical and biotechnology organizations, yet they serve definitely various functions. CDMO provides a more comprehensive range of solutions that extend beyond production to include product development. CDMO provides a more inclusive range of solutions that extend beyond manufacturing to include product advancement.
Integration of AI-driven technology in the U.S. small molecule innovator API CDMO drives the growth of the market, driven by AI-driven enters the equation, renovating small molecule production by enhancing operational effectiveness, improving quality control, and eventually delivering. The role of AI-based systems becomes more pronounced at the commercial scale, where it allows continuous process confirmation and optimizes control setpoints mid-batch, enhancing operational efficacy. AI-powered systems incessantly monitor manufacturing processes, allowing early complexities identification and reducing disruptions. AI-based system empowers CDMOs to tackle unique complexities while maintaining operational excellence by improving transparency, speeding up timelines, and streamlining technology.
| Table | Scope |
| Market Size in 2026 | USD 9.94 Billion |
| Projected Market Size in 2035 | USD 16.85 Billion |
| CAGR (2026 - 2035) | 6.04% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Stage Type, By Customer Type, By Therapeutic Area |
| Top Key Players | Lonza Group Ltd., Thermo Fisher Scientific Inc., Catalent, Inc., Cambrex Corporation, Recipharm AB,CordenPharma International |
Which Stage Type Led the U.S. Small Molecule innovator API CDMO market in 2025?
In 2025, the clinical segment held the dominant market share of approximately 55% in 2025, due to clinical trials being significant for improved understanding and enhancing health. They support the testing of investigational medical interventions such as drugs, tools, vaccines, and lifestyle changes to see which ones are most likely to benefit patients. Clinical trials also supports to develop treatments for patients with diseases or conditions who have had few or no options.
Preclinical
Whereas the preclinical segment is the fastest-growing in the market, preclinical stages play a significant role in the journey toward novel drug discovery and advancement, evaluating the safety, efficacy, and potential adverse effects. Preclinical research is the foundation of drug advancement, marking the start of the journey to identify, characterize, and refine new therapeutic applications. Preclinical research using animals to study the efficacy of a therapeutic drug or strategy are vital step before translation to clinical trials.
Why did the Pharmaceutical Companies Segment Dominate the Market in 2025?
The pharmaceutical companies segment was dominant in the U.S. small molecule innovator API CDMO market, with approximately 92% share in 2025, as small molecule companies offers large scale advanced intermediates and APIs to end drug products for the GMP outsourcing supply chain. It offers drug development, manufacturing, and analytical solutions. These organizations help pharma and biotech organizations via supplying scientific skill, advanced technology, and operational capacity that do not exist in-house.
Biotechnology Companies
Whereas the biotechnology companies segment is the fastest-growing in the market, as these companies are progressively prioritizing collaborations with CDMOs that provide regional manufacturing abilities. CDMOs provide process optimization, analytical testing, government support, formulation, and manufacturing on different scales, specifically catering to novel chemical entities (NCEs).
Why did the Oncology Segment Dominate the Market in 2025?
The oncology segment was dominant in the U.S. small Molecule innovator API CDMO market with approximately 42% share in 2025, as small molecule is relatively simple structure, compared to that of biologics, allowing for oral administration, easier formulation, and affordable production. CDMOs possess specialized containment services necessary for the production of cytotoxic, targeted cancer drugs, safeguarding quality and safety.
Infectious Diseases
Whereas the infectious diseases segment is the fastest-growing in the market, as a CDMO in pharma is an important partner that assists pharmaceutical organizations with the development of formulations, clinical production, enhancing processes, and commercial-scale manufacturing. CDMOs reduce coordination complexity, accelerate time-to-market, and offer better cost control.
How did the U.S. dominate the market in 2025?
In 2025, the U.S. dominated the U.S. small molecule innovator API CDMO market, as a strong R&D start-up ecosystem is significant in driving innovation and increasing economic growth. Major healthcare companies attract and retain skilled talent, lower brain drain, and encourage technological self-reliance. With a vigorous innovation ecosystem and high levels of spending in research, the U.S. maintained its leadership in innovation. In the United States, the biotech sector is maintained by scientific expertise, reliable spending, and a long history of research and innovation. U.S.-driven CDMO specializing in the advancement and production of small molecules, involving high-potency APIs, for organizations advancing complex and time-sensitive services, which drives the growth of the market.
For Instance,
R&D:
Manufacturing Processes:
Patient Services:
| Company | Headquarters | Latest Update |
| Lonza Group Ltd. | Switzerland | Lonza Group AG has quietly become one of the most influential biomanufacturing solutions on the planet. |
| Thermo Fisher Scientific Inc. | United States | In July 2025, Thermo Fisher Scientific Inc. expanded its strategic collaboration with Sanofi to allow additional U.S. drug product production. |
| Catalent, Inc. | Florida | Catalent delivers end-to-end pharma services as a trusted CDMO, advancing drug development and manufacturing worldwide |
| Cambrex Corporation | United States | In October 2025, Cambrex announced a $120 million spending to expand its U.S. operations, addressing increased demand for API development and production, and accelerating the company’s leadership role in the fast-growing peptide therapeutics. |
| Recipharm AB | Sweden | Recipharm expands its industrial operations to meet demanding FDA requirements. |
| CordenPharma International | Switzerland | CordenPharma's Small Molecule platform provides large-scale advanced intermediates and APIs to final drug products for your GMP outsourcing supply chain. |
Strengths
Weaknesses
Opportunities
Threat
By Stage Type
By Customer Type
By Therapeutic Area