Primarily, the blockchain in the pharmaceutical field is referred to as a decentralized, immutable digital ledger technology that is leveraged to preserve, track, & control drug supply chains, clinical trials , & patient data. Moreover, this industry facilitates end-to-end traceability from manufacturer to patient, eliminating counterfeit drugs, simplifying recalls, & verifying data integrity through cryptographic, tamper-proof records.
In terms of data security, these blockchain technologies are increasingly providing high-level security for patient information, clinical trials, and intellectual property by employing cryptographic approaches. Blockchain solutions also help in monitoring conditions for sensitive biologics, tracking temperature & humidity during transit to mitigate spoilage.
What are Counterfeit Drugs & the Impact of the Blockchain on these Drugs?
- Counterfeit drugs are known as forgery or falsified medicines developed & sold with the planned intention to deceive consumers about their origin, authenticity, or effectiveness. Also, these kinds of drugs are illegal, which can lead to serious health risks, like treatment failure, adverse reactions, & death.
- To raise the focus on discarding or removing counterfeit drugs, the pharmaceutical blockchain is actively assisting through
- Real-time authentication, while considering specific, scannable QR codes or serial numbers that are linked to physical drugs and should be registered on the ledger. This further enables consumers for immediate verification of these types of product�s authenticity.
- Also, blockchain is fostering visibility among stakeholders/regulators, which confirms drugs are managed properly & declining the risk of theft.
How do Stringent Regulations & Surging Needs Drive the Progression of Blockchain in the Pharmaceutical Industry?
- Diverse catalysts are driving prospective developments and ground-breaking advances in the pharmaceutical blockchain across the globe.
- One of the key drivers, the gradually rising counterfeit drugs, which account for nearly 10% of the global drug supply, is demanding extensive blockchain. This will provide a rigid, end-to-end audit trail, enabling instant monitoring, tracking & verification of drug authenticity.
- However, the emergence of strict regulations, including the US Drug Supply Chain Security Act (DSCSA) & the EU Falsified Medicines Directive (FMD), requires electronic, interoperable systems for tracing prescription drugs, for which blockchain serves as a viable solution to meet these essentials.
- Often, leaders are facing segmented conventional systems, resulting in a deficit & a lack of transparency, where blockchain provides a shared, tamper-evident ledger, with optimized recall management, finding product diversion, & also enables real-time tracking from producer to patient.
Impressive Trends are Focusing on the Broader Use of Advanced Technologies
- The worldwide smart contracts automate compliance processes, specifically instantaneously validating, documenting, & releasing, or blocking, batches based on quality control data, that comply with the Drug Supply Chain Security Act (DSCSA).
- Alongside, rigorous blockchain technology is integrated with AI and IoT devices, which are assisting in tracking environmental conditions in real-time, minimizing waste & promising safety during transport.
- A pivotal trend includes a notable step toward decentralized networks to record transactions, which secures patient privacy and protects sensitive medical data, and omits single points of failure.
Powerful Smart Contracts are Supporting the Exploration of Revolutionary Cold Chain Management�
- More sophisticated smart contracts have various applications across the cold chain system of pharmaceuticals, which mainly covers�
- The adoption of IoT sensors to monitor temperature, humidity, and location, with direct transmission of data to the blockchain. Somehow, settings move beyond expected limits, these contracts show instance notification to stakeholders and enable corrective action before products get damaged.
- Extensively, smart contracts are terminating human paperwork, as they automate verification by complying with cold chain regulations. In some instances, crossing of temperature thresholds in shipment uses contracts for automatic rejection of the shipment, and also updates the product status to impose penalty clauses in the contract.
- After the successful, ensured delivery, with no issues, the smart contracts activate automatic payment from the retailer to the supplier & carrier, which confirms expedited, protective transactions by eliminating middleman timelines.
Describing the Regulatory Framework of Major Areas for Pharmaceutical Blockchain in 2026
| United States | Drug Supply Chain Security Act (DSCSA) | This shows a need for a fully secure, interoperable system that enables unit-level tracking from manufacturer to dispenser.� |
| Europe | Falsified Medicines Directive (FMD) | This highly emphasises the adoption of a repository-based system; blockchain is paired as a trust layer to ensure data integrity & track-and-trace needs. |
| China | National Medical Products Administration (NMPA) | This has been fostering immersive digital serialization & monitoring to ensure the security of supply chains, with a goal to combat counterfeit drugs. |
| India | Central Drugs Standard Control Organization (CDSCO) | Regulatory guidelines are broadly pushing digital compliance, supporting manufacturers to shift away from manual record-keeping to ensure data provenance & authenticity. |
Arising Challenges in the Pharmaceutical Blockchain that Affect the Global Revolution
- For technology, infrastructure, and training, there is a need for a substantial initial investment, which restricts certain emerging firms.
- Nowadays, systems are facing concerns with raised transaction speed, which is demanded, & this leads to minimal handling needed for huge-scale logistics.
- Uncertainty in the regulatory landscape & limitations in integrating decentralized systems are also impacting the overall development.
- The emergence of secure, public blockchains shows risks related to sensitive patient data (GDPR compliance) & potential security flaws.
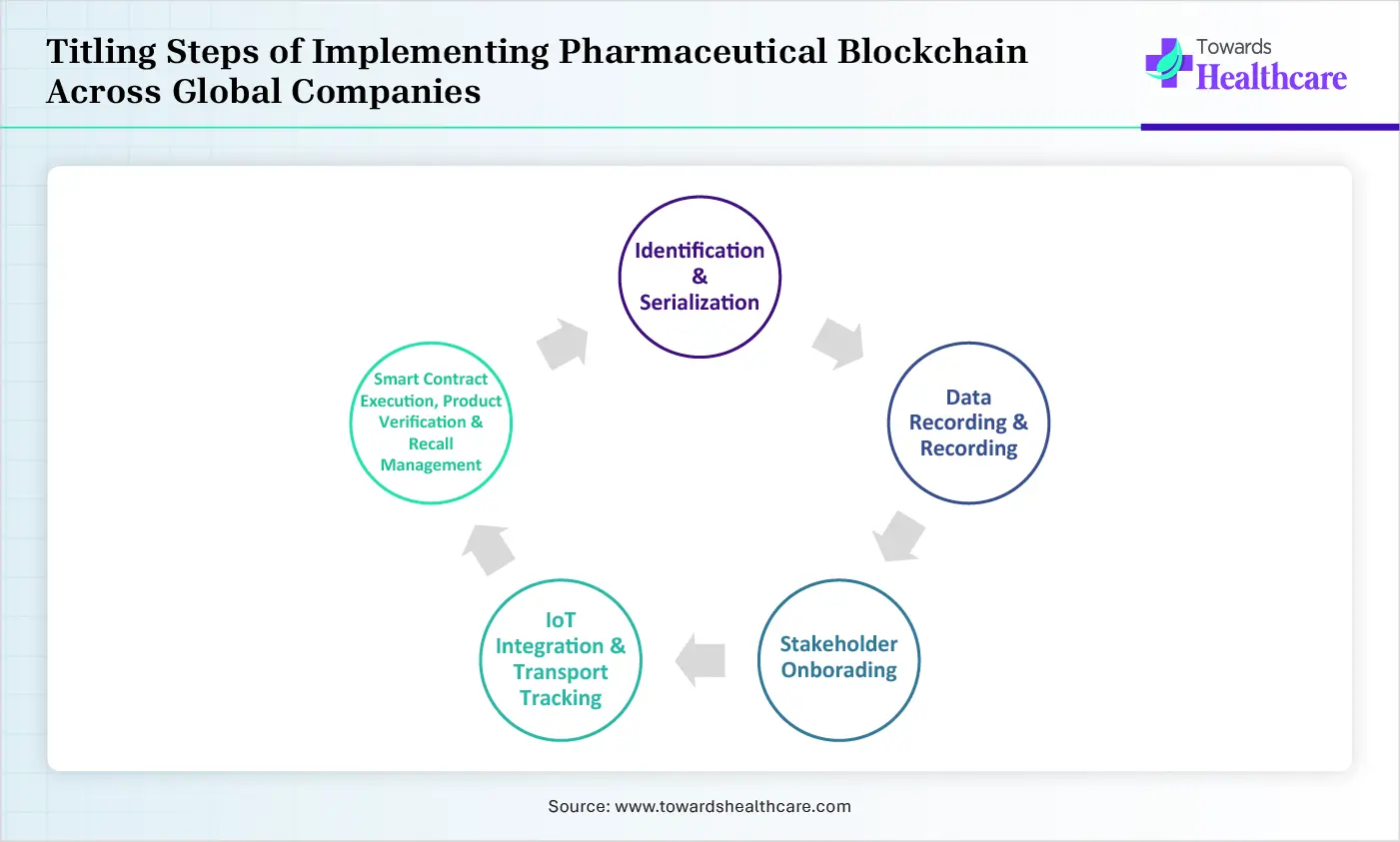
Titling Steps of Implementing Pharmaceutical Blockchain Across Global Companies
Appealing AI Roles for the Prospective Breakthroughs of Pharmaceutical Blockchain
Foremost AI engagement includes the analysis of blockchain-stored supply chain data, i.e. from raw materials to final delivery, to address anomalies or fraudulent patterns in real time, which will prevent falsified drugs from arriving on the market. Alongside, AI solutions can project demand and probable disturbances during smart contracts on the blockchain and automatically implement actions, like reshaping inventory levels or reordering supplies, based on these assumptions.
On the other hand, in the case of clinical trials, AI spurs the study of patient recruitment data and trial results protected on a blockchain to adjust protocols dynamically, & also ensure data privacy while raising trial pace & safety.
Eventually, smart contracts are becoming more autonomous by utilizing predictive intelligence to validate contract performance, handle cold-chain logistics, and process payments automatically.
Involvement of Top Companies in the Blockchain in Pharmaceutical Industry, with Their Significant Efforts
| PharmaLedger Association | This firm has been actively providing e-prescriptions, clinical trials, and supply chain management to eliminate fraud and boost transparency. |
| IBM | It specializes in the deployment of blockchain for drug traceability and supply chain transparency. |
| Chronicled | This has explored the MediLedger Network, a permissioned blockchain solution that employs smart contracts to track & trace pharmaceuticals. |
| Guardtime | A company focuses on executing blockchain in health infrastructures, with secure audit trails for pharma supply chains, & preserving sensitive data. |
| Blockpharma | It emphasizes the identification & mitigation of counterfeit drugs from entering the supply chain. |
| Triall | Its offering comprises �Blockchain Proof,� which assists in securely managing clinical trial data and documentation to prevent tampering & accelerate regulatory approvals. |
| FarmaTrust | This firm facilitates the Ethereum blockchain to foster the reliability of clinical trials, confirming secure and anonymized data collection for regulatory agencies. |
| Embleema | This operates a network that surges drug development by enabling real-world evidence (RWE) to be safely shared among patients & regulators. |
| Blockchain Industry Group (BIG) | A firm assists in developing blockchain applications to support tracking drugs, mitigating counterfeits, and securing supply chains within the pharmaceutical sector. |
Highlight Notable Unveilings Across the Blockchain in Pharmaceutical Industry in 2025
- In January 2026, the Indian Pharmacopoeia Commission (IPC) launched a novel QR code-based verification system to assist consumers in checking the authenticity of medicines and protect them from counterfeit drugs.
- In November 2025, Propanc Biopharma, the Australian-based pharmaceutical company, launched a cryptocurrency treasury worth up to $100 million to secure long-term funding for its cancer drug development programs.
- In November 2025, GeekStake introduced its innovative developer-focused toolset to empower the security, reliability, and lifecycle management of smart contracts through advanced AI-powered automation.
- In September 2025, Bio Protocol raised $6.9m in a funding round led by Maelstrom Fund to unveil Bio Protocol's growth into a full-stack platform for AI-assisted decentralised science, scientific funding and drug discovery.
- In August 2025, Sharps Technology closed more than $400 million & introduced a digital asset treasury strategy, with the significant holding being the cryptocurrency SOL.
- In August 2025, Wellgistics Health, Inc. rolled out its XRP Implementation Program that will enable independent pharmacies to leverage blockchain-based payments for prescription drug transactions.
Conclusion
The worldwide promotion of blockchain in pharmaceutical industry has been propelled by the surging focus on combating counterfeit drugs, preserving data, bolstering logistical efficiency by using smart contracts, and also encouraging trust among regulators through immutability. Ongoing efforts will push real-time visibility of raw materials & finished products, which lowers shortages, enhances inventory management, and enables immediate tracking of targeted recalls. Prospective opportunities will also pose integration of IoT sensors with blockchain, which will allow protective, tamper-proof tracking of biologics and vaccines, with ensured temperature stability throughout transit. However, the pharmaceutical ecosystem has been keeping its goal for widespread AI adoption to ensure data integrity in drug discovery, protect clinical trials, & improve the supply chain by estimating demand & decreasing counterfeiting through IoT integration, smart contracts, & AI analytics.
Expert Insights
According to a recent report on the Blockchain in Pharmaceutical Market, published on Towards Healthcare, experts indicate that blockchain adoption is transforming pharmaceutical supply chains by enhancing transparency, traceability, and data security. The technology enables real-time tracking of drug movement, helping to combat counterfeit medicines and ensure regulatory compliance. Increasing digitization of healthcare systems and the need for secure data sharing among stakeholders are key drivers of growth. Furthermore, integration with emerging technologies such as artificial intelligence and IoT is expanding blockchain�s applications in clinical trials and drug distribution, positioning the market as a critical enabler of efficiency and trust in the pharmaceutical ecosystem.
About the Experts

Aditi Shivarkar
Aditi leads as Vice President at Towards Healthcare and brings over 15 years of experience in healthcare research, innovation, and strategy. She works closely with data from across the healthcare sector and turns it into clear direction that companies can actually use. Her work covers pharmaceuticals, medical devices, and digital health. She helps businesses understand where the market is going and how to respond with confidence. Aditi focuses on practical thinking, strong decision-making, and delivering real results that make a difference.

Aman Singh
Aman Singh brings over 13 years of experience in healthcare research and consulting. He studies global healthcare trends and keeps a close eye on areas like biotech, AI in healthcare, and new treatment approaches. At Towards Healthcare, he leads the research team and makes sure the work stays accurate, useful, and easy to understand. Aman breaks down complex changes in the industry and helps businesses make smart, informed decisions.

Piyush Pawar
Piyush Pawar works as Senior Manager for Sales and Business Growth at Towards Healthcare, with more than 10 years of experience in the healthcare space. He works directly with clients and helps them find the right research for their needs. He makes sure clients understand the insights and know how to use them in their business. Piyush builds strong relationships and focuses on helping companies grow by turning research into clear, practical action.




 Request Consultation
Request Consultation


