April 2026

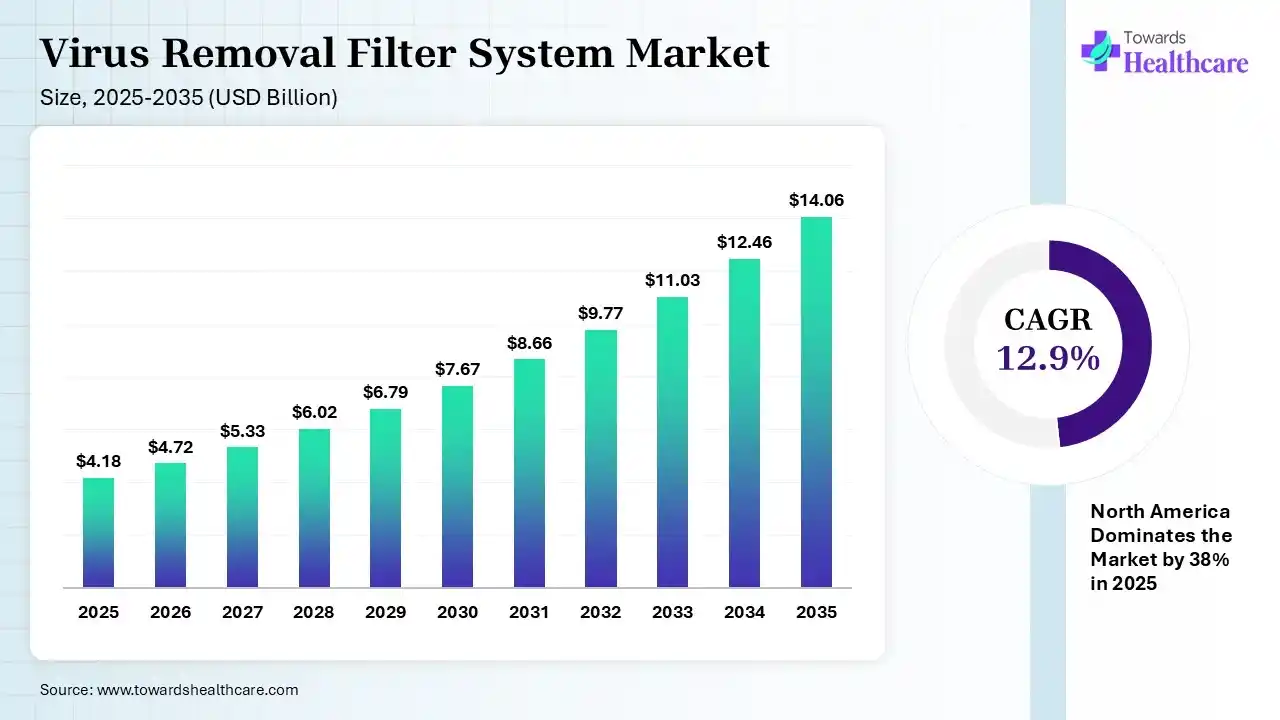
The global virus removal filter system market size was estimated at USD 4.18 billion in 2025 and is predicted to increase from USD 4.72 billion in 2026 to approximately USD 14.06 billion by 2035, expanding at a CAGR of 12.9% from 2026 to 2035. The market is growing steadily due to rising demand for biologics, vaccines, and strict safety regulations in pharmaceutical manufacturing, which require high-level virus clearance during production.
A virus removal filter system is a specialized filtration technology used to eliminate or reduce viruses from biological fluids, air, or water by passing them through a membrane with extremely small pore sizes. It is widely used in biopharmaceutical and healthcare to ensure product safety, especially in the production of vaccines, biologics, and blood-derived therapies. The virus removal filter system market is growing due to the rising global demand for vaccines, especially after pandemics, and the need for high-purity, virus-free biological products. Strict regulatory standards for vaccine safety and quality are pushing manufacturers to adopt advanced filtration technologies.
AI is transforming the market by enabling smarter process optimization, predictive maintenance, and real-time monitoring in biopharmaceutical manufacturing. Advanced analytics improve filtration efficiency, reduce contamination risks, and enhance quality control. AI-driven modeling also accelerates filter design and validation, lowering costs and development time. As automation expands, manufacturers can achieve higher productivity, consistent performance, and faster compliance with stringent regulatory standards.
Shift Toward Single-Use and Disposable Systems
The market is increasingly adopting single-use filtration systems due to their cost-effectiveness, reduced contamination risk, and operational flexibility. These systems eliminate cleaning requirements, support faster production cycles, and are highly suitable for modern biopharmaceutical and vaccine manufacturing environments.
Advancements in High-Performance Membrane Technologies
Continuous innovation in membrane materials and nanofiltration techniques is improving virus clearance efficiency and throughput. Enhanced filter durability and selectivity are enabling better performance, supporting the growing demand for high-purity biologics and advanced therapeutic products.
Integration of Automation and AI in Filtration Processes
Automation and AI-driven monitoring are transforming filtration operations by enabling real-time control, predictive maintenance, and improved process optimization. These technologies enhance consistency, reduce human error, and ensure compliance with stringent regulatory standards, shaping the future of smart biomanufacturing.
| Table | Scope |
| Market Size in 2026 | USD 4.72 Billion |
| Projected Market Size in 2035 | USD 14.06 Billion |
| CAGR (2026 - 2035) | 12.9% |
| Leading Region | North America by 38% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product Type, By Technology, By Application, By End User, By Membrane Material, By Region |
| Top Key Players | Merck KGaA, Danaher Corporation, Sartorius AG, Thermo Fisher Scientific, GE HealthCare, Charles River Laboratories |
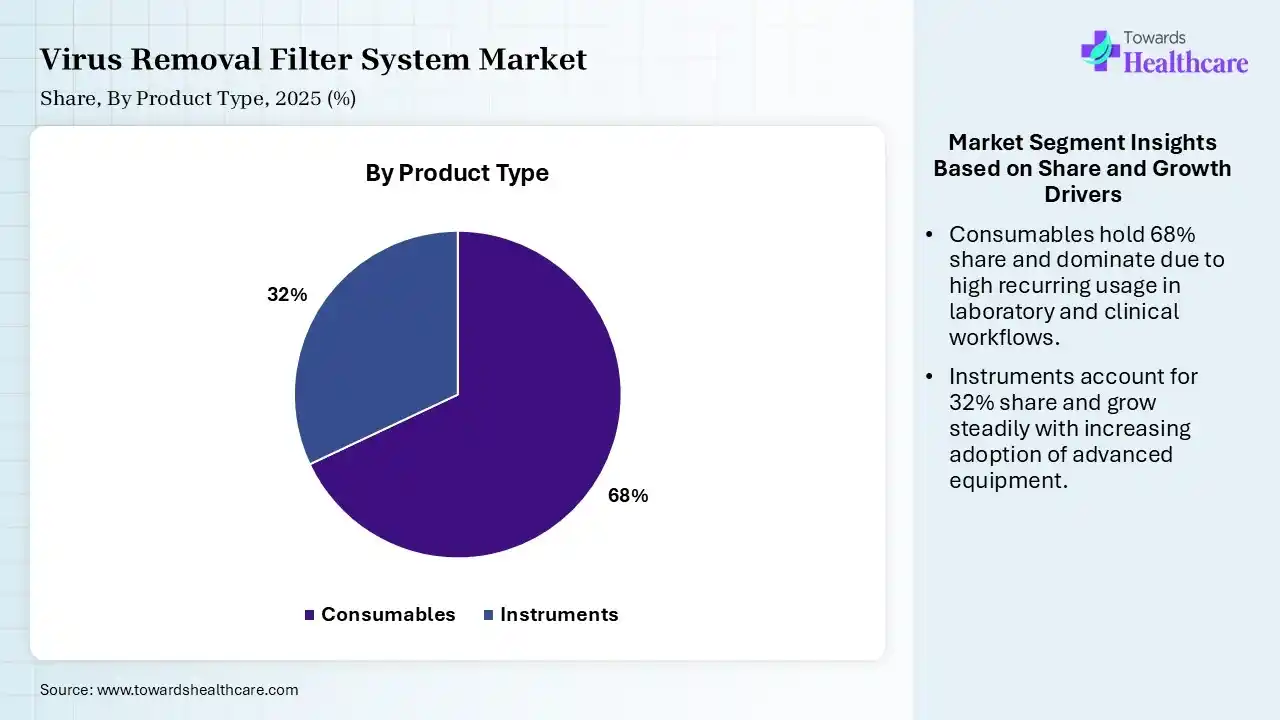
| Segment | Share 2025 (%) |
| Consumables | 68% |
| Instruments | 32% |
The Consumables Segment Dominated the Market in 2025
The consumables segment dominated the virus removal filter system market with a revenue share of 68% in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to its recurring usage in biopharmaceutical production processes, where filters, membranes, and cartridges require frequent replacements to maintain efficiency and sterility. High demand for continuous manufacturing, strict regulatory standards, and the consumption, making consumption a reliable and significant revenue-generating segment compared to a one-time equipment purchase.
The instruments segment held the second-largest share of 32% in 2025 due to its essential role in supporting filtration processes, including systems for monitoring, pressure control, and automation. Although purchases are less frequent than consumables, these systems are high-value and critical for ensuring process efficiency and compliance. Growing adoption of advanced bioprocessing equipment and automation in pharmaceutical manufacturing continues to drive steady demand for filtration instruments.
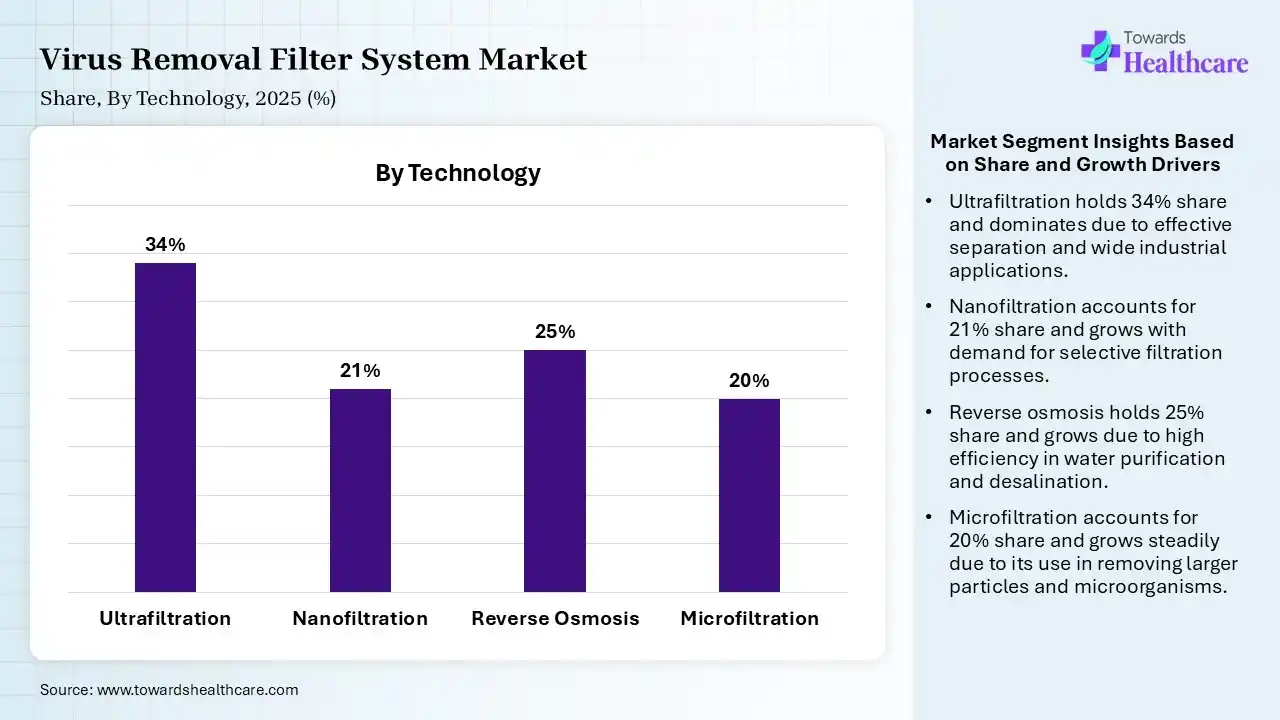
| Segment | Share 2025 (%) |
| Ultrafiltration | 34% |
| Nanofiltration | 21% |
| Reverse Osmosis | 25% |
| Microfiltration | 20% |
The Ultrafiltration Segment Led the Market in 2025 with the Largest Share
The ultrafiltration segment held a dominant share in the virus removal filter system market of 34% in 2025 due to its high efficiency in removing viruses while maintaining protein integrity, making it ideal for biologics and vaccine production. Its cost-effectiveness, scalability, and compatibility with existing bioprocessing systems support widespread adoption. Additionally, strong regulatory acceptance and its ability to handle large volumes with consistent performance further strengthen its leading position in the market.
The reverse osmosis segment held the second-largest share of 25% in 2025 due to its strong capability in removing viruses, dissolved solids, and contaminants with high precision. It is widely used in water purification and plays a support role in pharmaceutical processes. Although highly effective, higher energy consumption and operational costs compared to ultrafiltration limit its dominance, while its reliability and broad application sustain significant market demand.
The nanofiltration segment held 21% share in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to its ability to effectively remove smaller virus particles while maintaining high permeability and lower energy consumption compared to reverse osmosis. Its increasing adoption in biopharmaceutical processing, water purification, and advanced separation applications, along with continuous technological advancements, is driving rapid demand and positioning it as a high-growth segment in the market.
The microfiltration segment held 20% of the virus removal filter system market share in 2025 due to its cost-effectiveness, high flow rates, and efficiency in removing larger particles, bacteria, and some viruses in pre-filtration stages. It is widely used as a primary clarification step in biopharmaceutical and water treatment processes. Increasing demand for scalable and energy-efficient filtration solutions, along with their compatibility with other advanced filtration technologies, is driving their steady adoption and market growth.
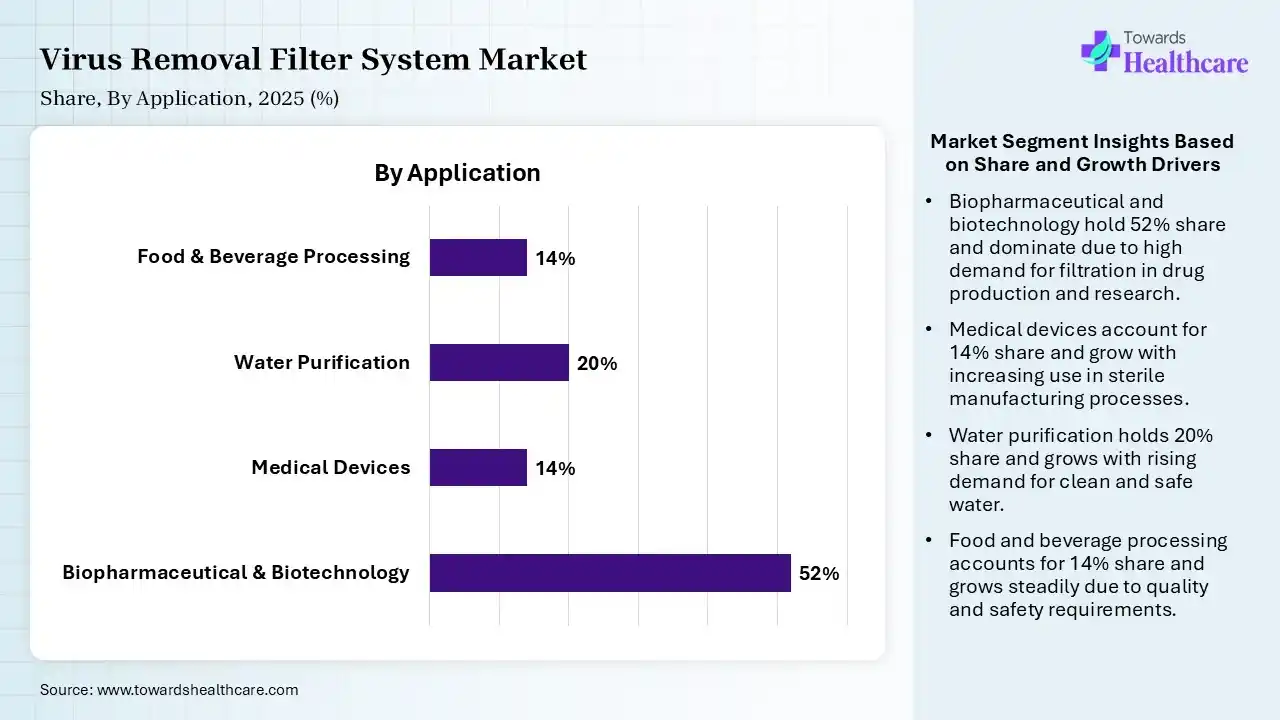
| Segment | Share 2025 (%) |
| Biopharmaceutical & Biotechnology | 52% |
| Medical Devices | 14% |
| Water Purification | 20% |
| Food & Beverage Processing | 14% |
The Biopharmaceutical & Biotechnology Segment Led the Market in 2025 with the Largest Share
The biopharmaceutical & biotechnology segment held a dominant share in the virus removal filter system market of 52% in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to the high demand for safe and contamination-free production of biologics, vaccines, and advanced therapies. Strict regulatory requirements for virus clearance, along with increasing investment in drug development and biologics manufacturing, drive strong adoption. Additionally, the rapid growth of cell and gene therapies further boosts the need for reliable and efficient virus filtration systems in this segment.
The water purification segment held 20% of the market share in 2025 due to rising concerns over waterborne viruses and increasing demand for safe drinking water globally. Growing urbanization, industrialization, and stricter environmental regulations are driving the adoption of advanced filtration systems. Additionally, government initiatives and investments in water treatment infrastructure are accelerating the use of virus removal technologies in municipal and industrial applications.
The medical devices segment held 14% of the virus removal filter system market share in 2025 due to increasing demand for sterile, contamination-free products and stricter infection control standards in healthcare settings. Virus removal filtration is essential in device manufacturing processes, especially for implantable and fluid-contact devices. Rising surgical procedures, growth in advanced medical technologies, and regulatory emphasis on patient safety are driving greater adoption of reliable filtration systems within this segment.
The food & beverages processing segment held 14% of the market share in 2025 due to rising demand for safe, contamination-free products and strict hygiene regulations. Virus filtration helps ensure product quality, extend shelf life, and maintain compliance with food safety standards. Growing consumer awareness, expanding packaged food industry, and the need for advanced purification technologies are driving greater adoption of virus removal systems in this sector.
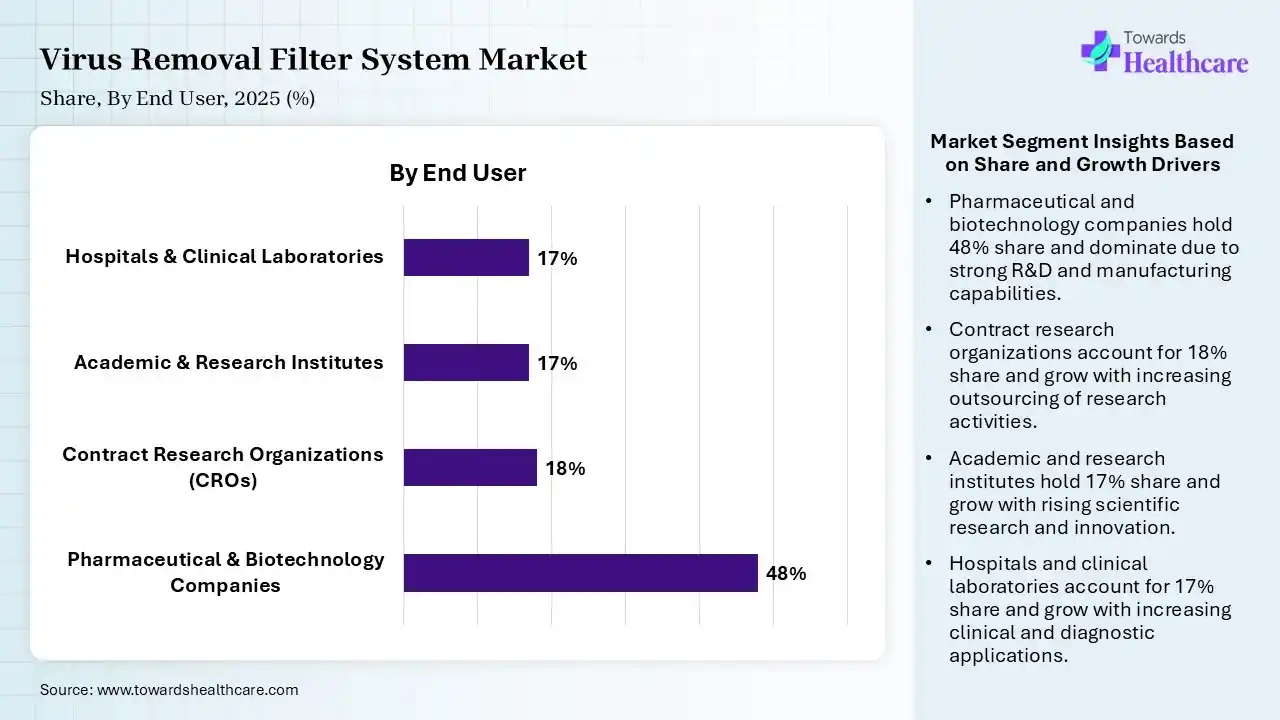
| Segment | Share 2025 (%) |
| Pharmaceutical & Biotechnology Companies | 48% |
| Contract Research Organizations (CROs) | 18% |
| Academic & Research Institutes | 17% |
| Hospitals & Clinical Laboratories | 17% |
The Pharmaceutical & Biotechnology Companies Segment held a Dominant Position in the Market in 2025
The pharmaceutical & biotechnology companies segment held a dominant share in the virus removal filter system market of 48% in 2025 due to high demand for virus-safe production of biologics, vaccines, and advanced therapies. Strict regulatory requirements and large-scale manufacturing needs drive the continuous use of filtration systems. Additionally, strong R&D investments and expanding biopharma pipelines further increase reliance on efficient virus removal technologies, securing this segment’s leading position.
The contract research organizations (CROs) segment held the second-largest share of 18% in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to increasing outsourcing of drug development and biopharmaceutical testing by pharmaceutical companies. CROs require reliable virus removal systems to ensure safety and regulatory compliance during research and production processes. Their expanding role in clinical trials, cost efficiency, and growing partnership with biotech firms are driving strong demand in this segment.
The academic & research institutes segment held 17% of the virus removal filter system market share in 2025 due to increasing focus on virology, biologics development, and advanced life science research. Rising government and private funding supported studies requiring safe handling and purification of biological samples. Additionally, growing collaborations with biopharmaceutical companies and expanding research in vaccines and gene therapies are driving demand for reliable virus removal filtration systems in these settings.
The hospitals & clinical laboratories segment held 17% of the market share in 2025 due to rising demand for accurate diagnostics and strict infection control measures. Virus removal filtration is essential for maintaining sterile environments and safe sample handling. Increasing patient volumes, growth in diagnostic testing, and heightened awareness of healthcare-associated infection are driving the adoption of advanced filtration systems in these settings.
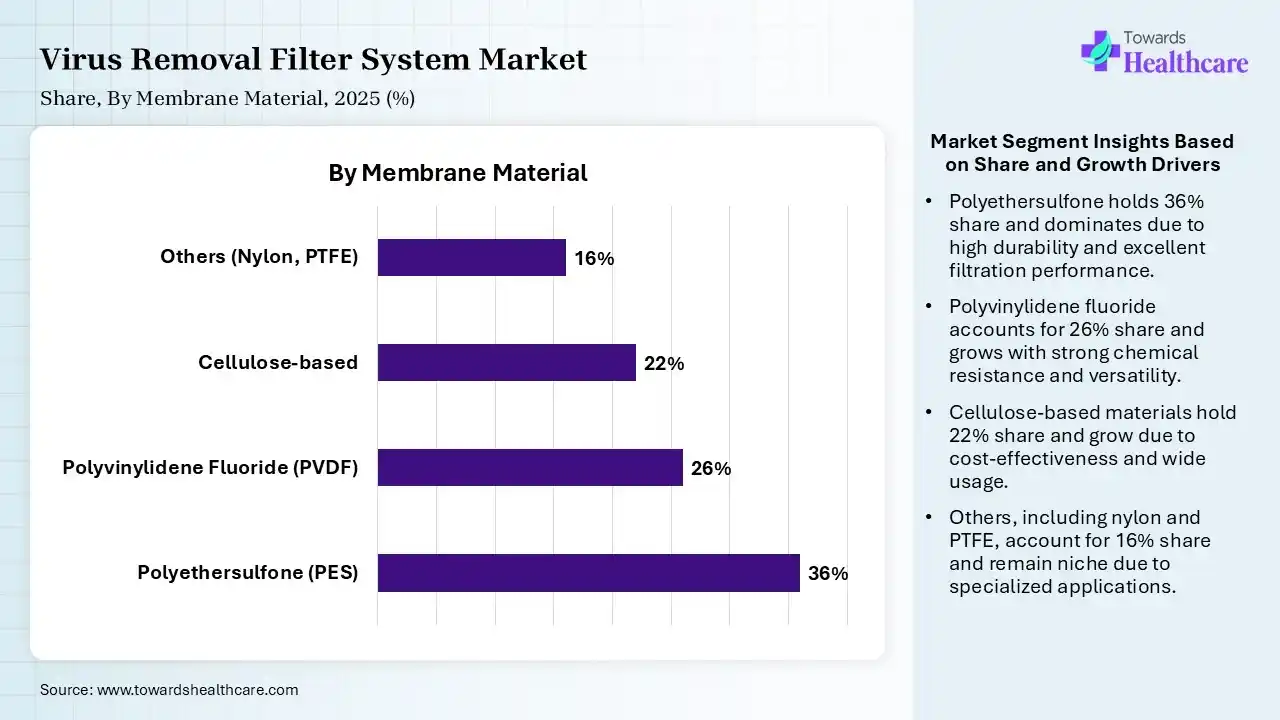
| Segment | Share 2025 (%) |
| Polyethersulfone (PES) | 36% |
| Polyvinylidene Fluoride (PVDF) | 26% |
| Cellulose-based | 22% |
| Others (Nylon, PTFE) | 16% |
The Polyethersulfone (PES) Segment Dominated the Market in 2025
The polyethersulfone (PES) segment led the virus removal filter system market with a share of 36% in 2025 due to its excellent filtration efficiency, high flow rates, and strong chemical and thermal stability. PES membranes offer low protein binding, making them ideal for biologics and vaccine processing. Their durability, consistent performance, and compatibility with a wide range of biopharmaceutical applications drive widespread adoption and market leadership.
The polyvinylidene fluoride (PVDF) segment held the second-largest share of 26% in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to its strong chemical resistance, mechanical durability, and excellent filtration performance. PVDF membranes are widely used in biopharmaceutical and water filtration applications for their ability to handle harsh conditions. Their high versatility, reliability, and compatibility with sterilization processes support steady demand and significant market presence.
The cellulose-based segment held 22% of the virus removal filter system market share in 2025 due to its cost-effectiveness, biodegradability, and wide availability, making it an attractive option for sustainable filtration solutions. These membranes offer good hydrophilicity and low fouling characteristics, supporting efficient filtration in various applications. Increasing focus on eco-friendly materials and expanding use in research and water treatment are driving steady demand for cellulose for cellulose-based filtration systems.
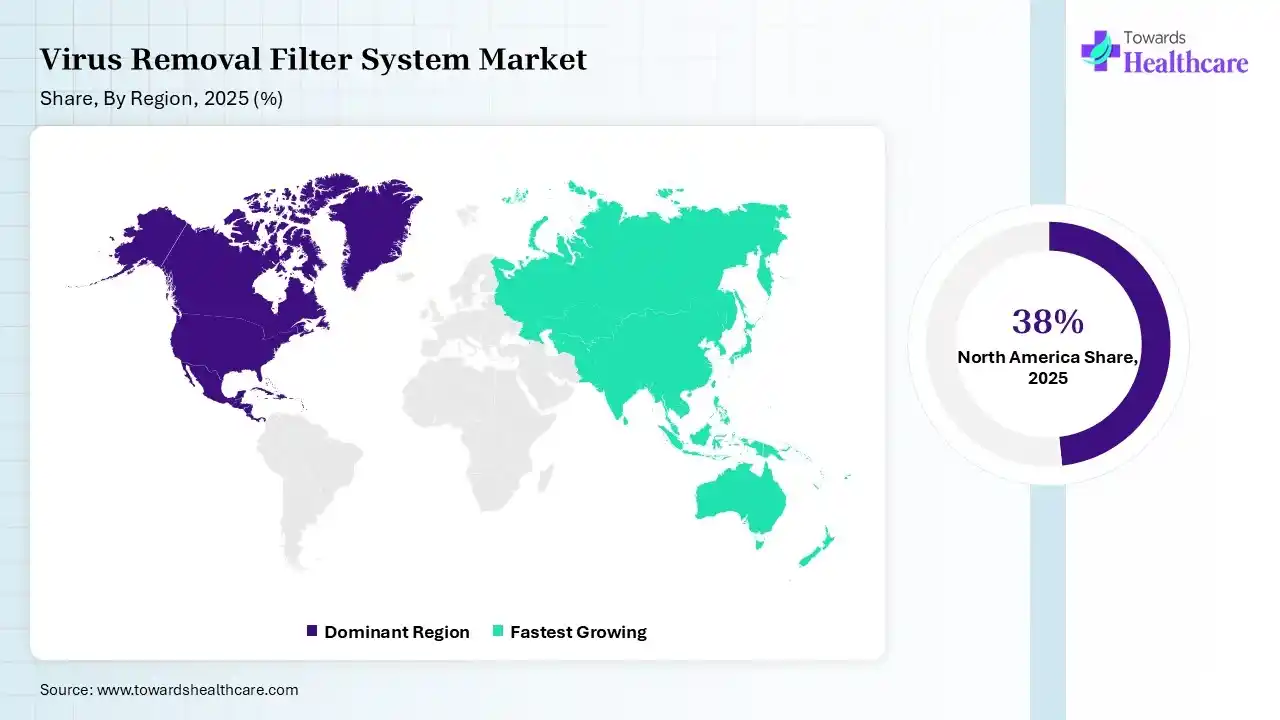
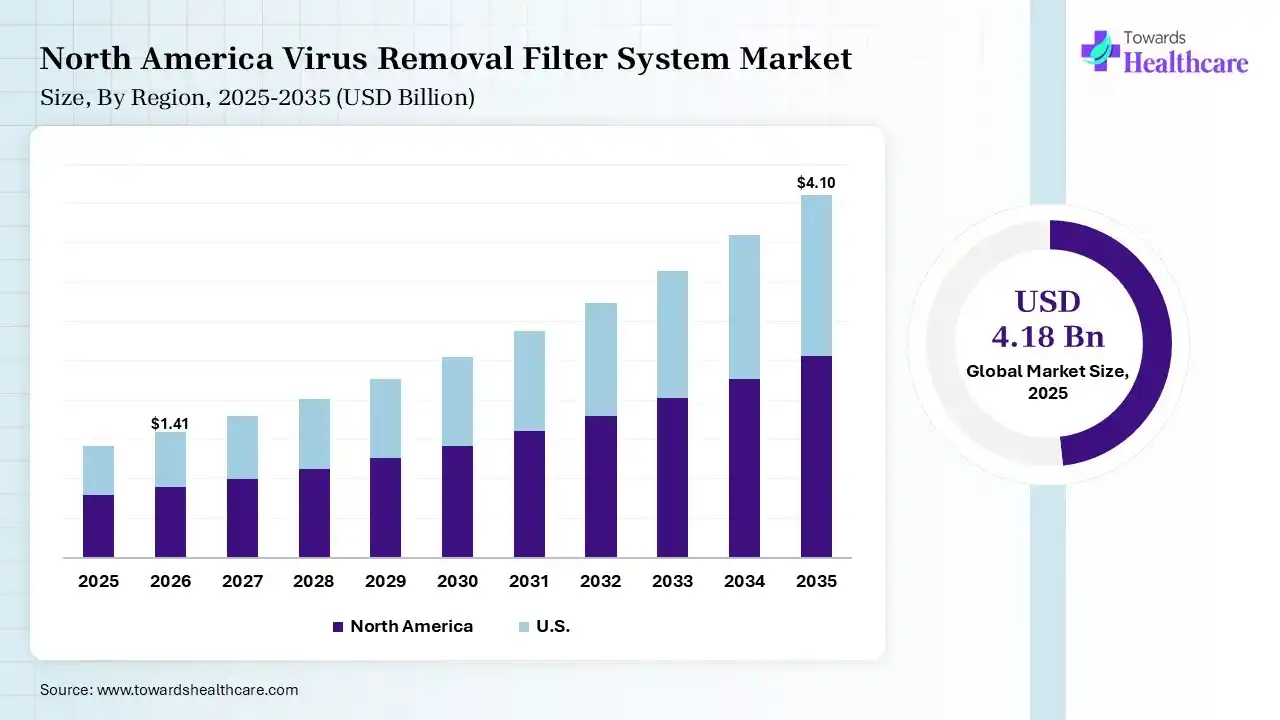
North America dominated the virus removal filter system market with 38% share in 2025 due to its well-established biopharmaceutical industry, strong presence of leading market players, and advanced manufacturing infrastructure. High R&D investment, strict regulatory standards, and rapid adoption of innovative filtration technologies further drive demand. Additionally, increasing biologics and vaccine production significantly contributes to the region’s market leadership.
U.S. Market Trends
The U.S. virus removal filter system market leads due to its advanced biopharmaceutical industry, strong presence of global manufacturers, and high investment in research and development. Strict regulatory frameworks ensure widespread adoption of high-quality filtration systems. Additionally, increasing production of biologics, vaccines, and cell and gene therapies continues to drive strong and consistent market demand.
Asia Pacific held 23% of the market share and is expected to grow at the fastest CAGR in the virus removal filter system market due to expanding biopharmaceutical manufacturing, increasing healthcare investments, and rising demand for vaccines and biologics. Rapid industrialization, supportive government initiatives, and improving regulatory frameworks are accelerating adoption. Additionally, growing research activities and the presence of emerging biotech hubs are further driving strong market growth across the region.
India Market Trends
India is anticipated to grow at the fastest CAGR in the virus removal filter system market due to its rapidly expanding biopharmaceutical industry, increasing vaccine production, and strong government support for healthcare infrastructure. Rising investments in biotechnology, growing contract manufacturing activities, and improving regulatory standards are accelerating adoption. Additionally, a large patient population and increasing focus on affordable biologics are further driving demand for virus removal filtration systems.

| Companies | Headquarters | Offerings |
| Merck KGaA | Darmstadt, Germany | Provides virus filtration membranes, single-use systems, and advanced bioprocessing solutions for safe production of biologics, vaccines, and cell & gene therapies. |
| Danaher Corporation | D.C., USA | Through subsidiaries, it offers virus filtration technologies, membranes, and integrated bioprocessing systems supporting large-scale biologics manufacturing. |
| Sartorius AG | Göttingen, Germany | Supplies sterile and virus filtration products, membrane filters, and single-use solutions for efficient downstream bioprocessing. |
| Thermo Fisher Scientific | Massachusetts, USA | Offers filtration systems, purification technologies, and analytical tools for virus removal in drug development and biologics production. |
| GE HealthCare | Illinois, USA | Provides bioprocessing and virus filtration solutions along with purification systems for vaccine and biologics manufacturing. |
| Charles River Laboratories | Massachusetts, USA | Focuses on virus safety testing, validation services, and support for ensuring effective virus clearance in biopharmaceutical processes. |
Strengths
Weaknesses
Opportunities
Threats
By Product Type
By Technology
By Application
By End User
By Membrane Material
By Region
April 2026
April 2026
April 2026
March 2026