
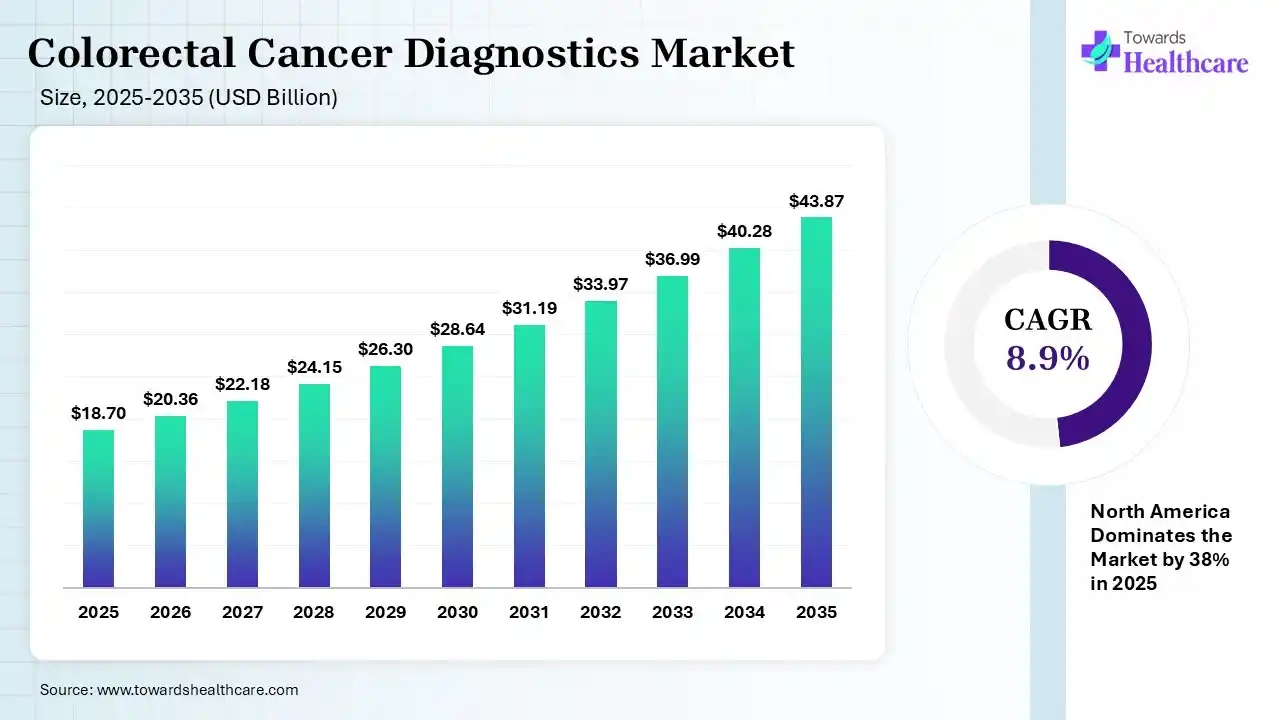
The global colorectal cancer diagnostics market size was estimated at USD 18.7 billion in 2025 and is predicted to increase from USD 20.36 billion in 2026 to approximately USD 43.87 billion by 2035, expanding at a CAGR of 8.9% from 2026 to 2035. A gradual rise in CRC incidence & broadening awareness, with robust screening programs, are driving the demand for these diagnostics. Besides this, the market is transforming AI-assisted solutions & the latest molecular diagnostics.
The colorectal cancer diagnostics market covers the integrated, systematic application of medical tests & imaging procedures to detect malignant cells in the colon or rectum. These approaches support locating tumors, finding pre-cancerous polyps, & ensure cancer presence by using tissue sampling. However, the global demand is driven by the increasing cases, broader adoption of non-invasive screening, and the latest molecular diagnostics.
Primarily, the leading healthcare organizations are employing AI algorithms to assess histopathologic images & whole-slide images (WSIs) to classify benign from malignant tissues. Whereas, eventual advances are promoting machine learning in the analysis of blood-based biomarkers, fluorescence spectroscopy, & complete blood counts. Additionally, the trend is rigorously fostering AI by utilizing CT & MRI imaging data to assist in anticipating tumor staging, grading & patient response to therapy.
Developing Liquid Biopsies & Biomarkers
Emerging blood tests spur the detection of circulating tumor DNA (ctDNA) or methylated DNA to find early-stage CRC and monitor recurrence.
Seeking Advanced Stool-Based Molecular Tests
Gradual efforts are innovating multitarget stool DNA tests & analyzing fecal miRNA signatures to enhance detection accuracy, especially for precancerous lesions.
Prospective Integrations
The market will explore the unification of AI with endoscopy (Computer-Aided Detection - CADe) & imaging to boost adenoma detection rates & lower false negatives during colonoscopy.
| Table | Scope |
| Market Size in 2026 | USD 20.36 Billion |
| Projected Market Size in 2035 | USD 43.87 Billion |
| CAGR (2026 - 2035) | 8.9% |
| Leading Region | North America by 38% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Test Type, By Technology, By Application, By End User, By Region |
| Top Key Players | Exact Sciences Corporation, F. Hoffmann-La Roche Ltd (Roche Diagnostics), Quest Diagnostics Incorporated, Danaher Corporation, Guardant Health, Inc., Abbott, Fujifilm Holdings Corporation, Epigenomics AG, Eiken Chemical Co., Ltd , Olympus Corporation |
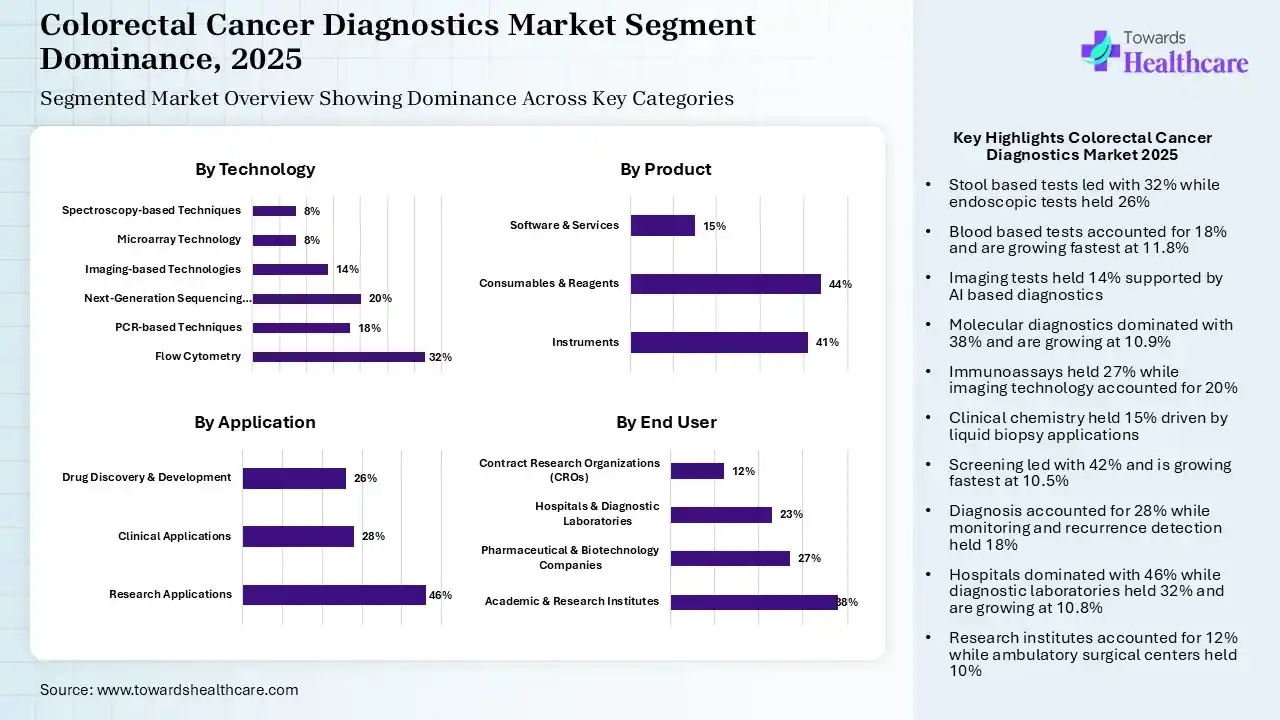
| Segment | Share 2025 (%) |
| Stool-Based Tests | 32% |
| Blood-Based Tests | 18% |
| Imaging Tests | 14% |
| Endoscopic Tests | 26% |
| Biopsy | 10% |
The Stool-Based Tests Segment Dominated the Market in 2025
The stool-based tests segment captured a major share of 32% of the colorectal cancer diagnostics market in 2025, due to the growing demand for non-invasive, home-based, & inexpensive tests to raise screening adherence, particularly among asymptomatic individuals. Along with government programs, firms are advancing next-generation multitarget stool RNA, which has approximately 94.4% sensitivity for colorectal cancer & 45.9% for advanced adenomas.
In 2025, the endoscopic tests segment held the second-largest share of 26% of the market. In the worldwide rising CRC instances, it is considered the gold standard in diagnosis, & also supports colorectal cancer (CRC) management, & facilitates both diagnosis & mitigation in one session. The latest adoption includes Confocal Laser Endomicroscopy (CLE), which offers real-time, microscopic imaging during the procedure.
However, the blood-based tests segment held 18% of the total market share and is predicted to expand rapidly at 11.8% CAGR in the coming era. Globally surging preference for minimally invasive procedures, these tests are offering more convenient, less patient anxiety & resolving hurdles. The FDA has authorized novel tests, including Shield for average-risk individuals.
The imaging tests segment accounted for a notable share of 14% of the colorectal cancer diagnostics market. These kinds of tests are improving detection accuracy & are also highly adopted in healthcare systems by combining with AI. The CT Colonography offers an alternative for incomplete colonoscopies or frail patients.
| Segment | Share 2025 (%) |
| Molecular Diagnostics | 38% |
| Immunoassays | 27% |
| Clinical Chemistry | 15% |
| Imaging Technology | 20% |
The Molecular Diagnostics Segment Led the Market in 2025
The molecular diagnostics segment dominated with a 38% share in 2025 & is estimated to expand at 10.9% CAGR. These tests are ensuring the section of precision targeted interventions for metastatic CRC. Ongoing utilization of next-generation sequencing (NGS) & molecular biomarkers encourages earlier detection, prognosis & personalised therapy. The market has executed the latest methylation markers & AI-powered analysis.
In 2025, the immunoassays segment held a 27% share of the colorectal cancer diagnostics market, due to the broader use in routine diagnostics & its affordability, with merged automation. The market is increasingly leveraging the lateral flow assays & highly sensitive enzyme immunoassays (EIA), enabling robust identification of hemoglobin or other biomarkers in stool.
The imaging technology captured a lucrative share of 20% in 2025. The progression is driven by massive investments in radiology infrastructure, accelerating screening awareness. This is further promoting the use of AI-driven imaging, which raises diagnostic capabilities.
The clinical chemistry segment held 15% share of the market, due to the advanced infrastructure, which assists in consistent application. This approach is widely implemented in liquid biopsies to detect circulating tumor DNA (ctDNA) & RNA-based biomarkers, boosting accuracy in early-stage detection.
| Segment | Share 2025 (%) |
| Screening | 42% |
| Diagnosis | 28% |
| Prognosis | 12% |
| Monitoring & Recurrence Detection | 18% |
The Screening Segment Was Dominant in the Market in 2025
In 2025, the screening segment accounted for a 42% share & is predicted to expand fastest at 10.5% CAGR in the colorectal cancer diagnostics market. Through the diverse, extensive government programs, the market is experiencing an expansion of screening, mainly for individuals aged 45 and older, led by public awareness initiatives. In these surging testing rates, blood-based & mt-sDNA tests are being employed as first-line alternatives to select patients for essential diagnostic colonoscopies.
The diagnosis segment held the second-largest share of 28% of the market, due to the substantial rise in cases of CRC, with the emergence of diagnostic tools & integrated imaging methods. Booming patient preferences for less invasive methods, with faster evolution of highly sensitive, automated molecular diagnostic kits & liquid biopsies, enable earlier & precise staging.
The monitoring & recurrence detection segment captured a 18% share of the colorectal cancer diagnostics market. A huge burden of the population needs consistent monitoring, where liquid biopsy allows real-time disease tracking. Companies are looking for non-invasive, at-home tests for surveillance instead of repeated colonoscopies.
| Segment | Share 2025 (%) |
| Hospitals | 46% |
| Diagnostic Laboratories | 32% |
| Research Institutes | 12% |
| Ambulatory Surgical Centers | 10% |
The Hospitals Segment Dominated the Market in 2025
The hospitals segment led with a 46% share of the market in 2025. To address the growing incidence of CRC, hospitals are facilitating end-to-end care, from screening to treatment, with significant approaches in the diagnostic & care ecosystem. They also execute well-trained personnel, sophisticated imaging, & gold-standard colonoscopies to find & discard precancerous polyps.
However, the diagnostic laboratories segment captured 32% share in 2025 & is estimated to expand rapidly at 10.8% CAGR. These laboratories are extensively offering precise, non-invasive, & molecular-level insights for early detection & tailored treatment. Certain key labs are conducting genetic testing on tumor tissues to foster precision therapeutic strategies.
The research institutes segment accounted for a 12% share of the colorectal cancer diagnostics market, due to escalating oncology research funding, which enhances demand. Alongside, these institutes are emphasizing biomarker discovery & alliances with biotech leaders to explore innovations.
The ambulatory surgical centers segment held a notable share of 10% in 2025. This expansion is propelled by the growing step towards outpatient procedures, with the implementation of affordable care models for appealing to patients. ASCs reduce waiting times & increases the availability of cancer screening colonoscopies.
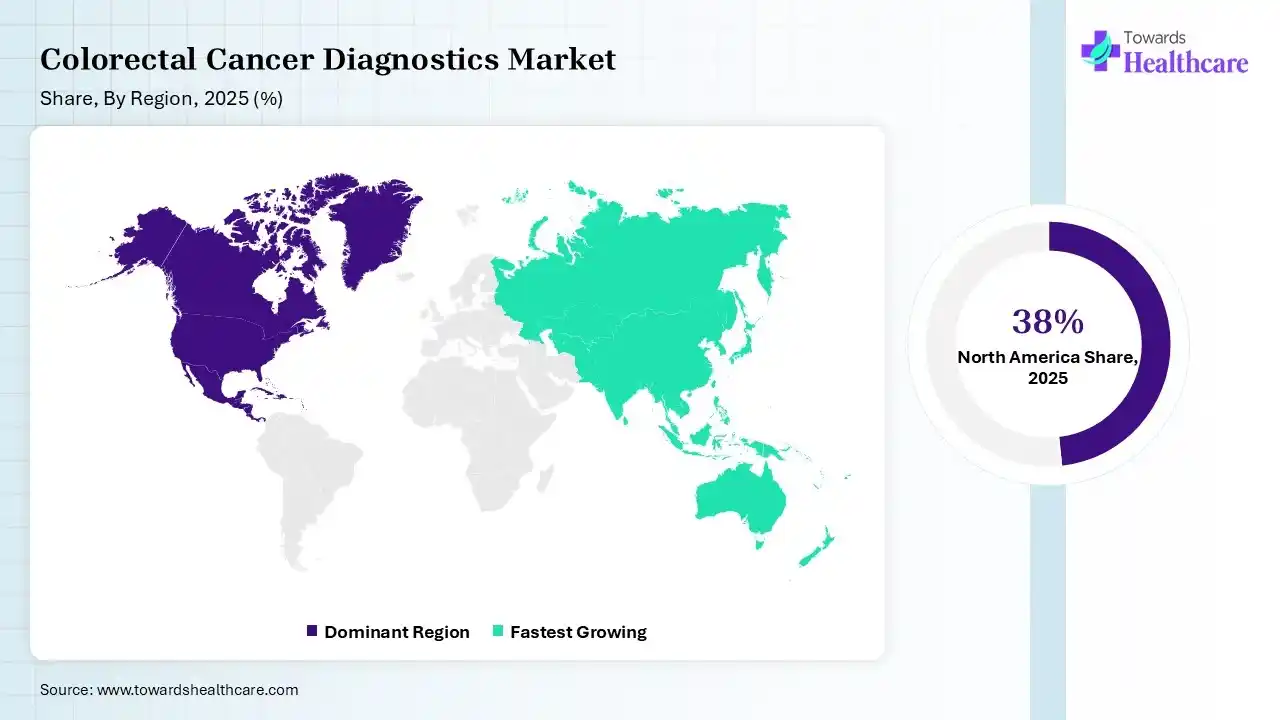
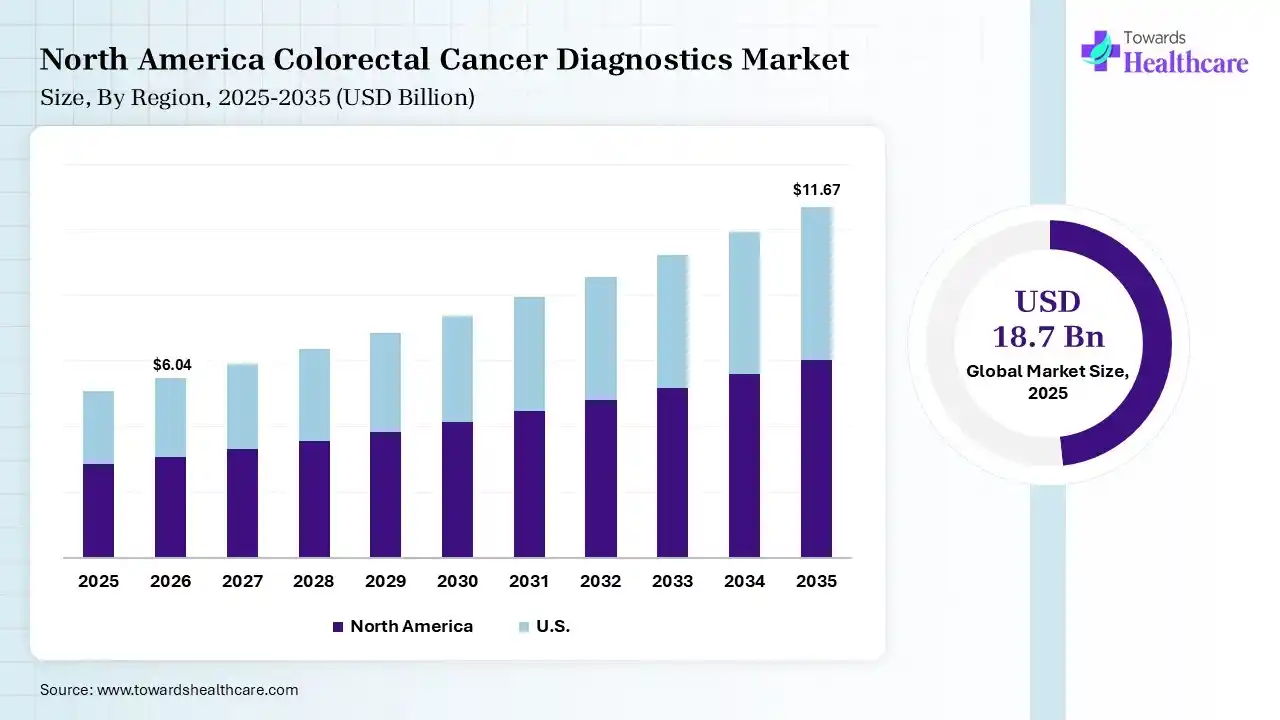
In 2025, North America registered dominance with 38% share of the colorectal cancer diagnostics market. This dominance is fueled by extensive screening programs, well-established healthcare infrastructures & suitable reimbursement policies. Research activities are focusing on educating primary care providers to understand symptoms in younger patients, who are often initially misdiagnosed.
For instance,
U.S. Market Trends
Specifically, the U.S. market held a 30% share, with the promotion of research into ctDNA, DNA methylation markers, & tumor-derived exosomes in both blood & stool to determine early, pre-cancerous stages. Emerging developments cover the FDA approval of blood-based screening tests, & roll out of next-generation stool-based tests.
Asia Pacific held 22% of the total market share in 2025 and is anticipated to witness rapid expansion at 11.2% CAGR in the colorectal cancer diagnostics market in the coming years. This is prominently impelled by the increasing burden of CRC incidences, particularly in China, Japan, & South Korea, along with progressing funding & government-powered, national-level screening incentives.
For instance,
China Market Trends
Whereas China is predicted to expand at 11.5% CAGR, as it has leveraged a two-step approach, i.e., initial risk assessment followed by high-precision diagnostics only for high-risk individuals. Also, China is highly deploying Computer-aided detection (CADe) & characterization (CADx) systems to raise Adenoma Detection Rates (ADR) & lower operator variability.
R&D
Regulatory Approvals
Patient Support & Services
| Company | Description |
| Exact Sciences Corporation | This firm specializes in non-invasive colorectal cancer screening & molecular diagnostics. |
| F. Hoffmann-La Roche Ltd (Roche Diagnostics) | Its portfolio covers integration of in vitro diagnostics (IVD), next-generation sequencing (NGS), & digital health solutions. |
| Quest Diagnostics Incorporated | This company offers various blood-based screening, fecal testing, & sophisticated molecular diagnostics. |
| Danaher Corporation | This mainly facilitates tissue staining for diagnosis, molecular testing for biomarkers, & 3D biology technologies for drug discovery. |
| Guardant Health, Inc. | Its offerings cover the FDA-approved Shield test & Guardant Reveal. |
| Abbott | This leader explored Cologuard & Cologuard Plus to their lineup, with existing rapid fecal tests to optimize early detection. |
| Fujifilm Holdings Corporation | A firm provides AI-enabled endoscopic imaging, therapeutic devices, & specialized screening centers. |
| Epigenomics AG | Its prominent offering is blood-based molecular diagnostics for colorectal cancer. |
| Eiken Chemical Co., Ltd | This offers screening through its OC-SENSOR line of Fecal Immunochemical Tests (FIT), with emphasis on non-invasive detection of human hemoglobin in stool. |
| Olympus Corporation | It has unveiled the EVIS X1 endoscopy system with TXI technology, OLYSENSE AI-driven diagnostic software, & the Endocuff Vision device. |
Strengths
Weaknesses
Opportunities
Threats
By Test Type
By Technology
By Application
By End User
By Region