April 2026

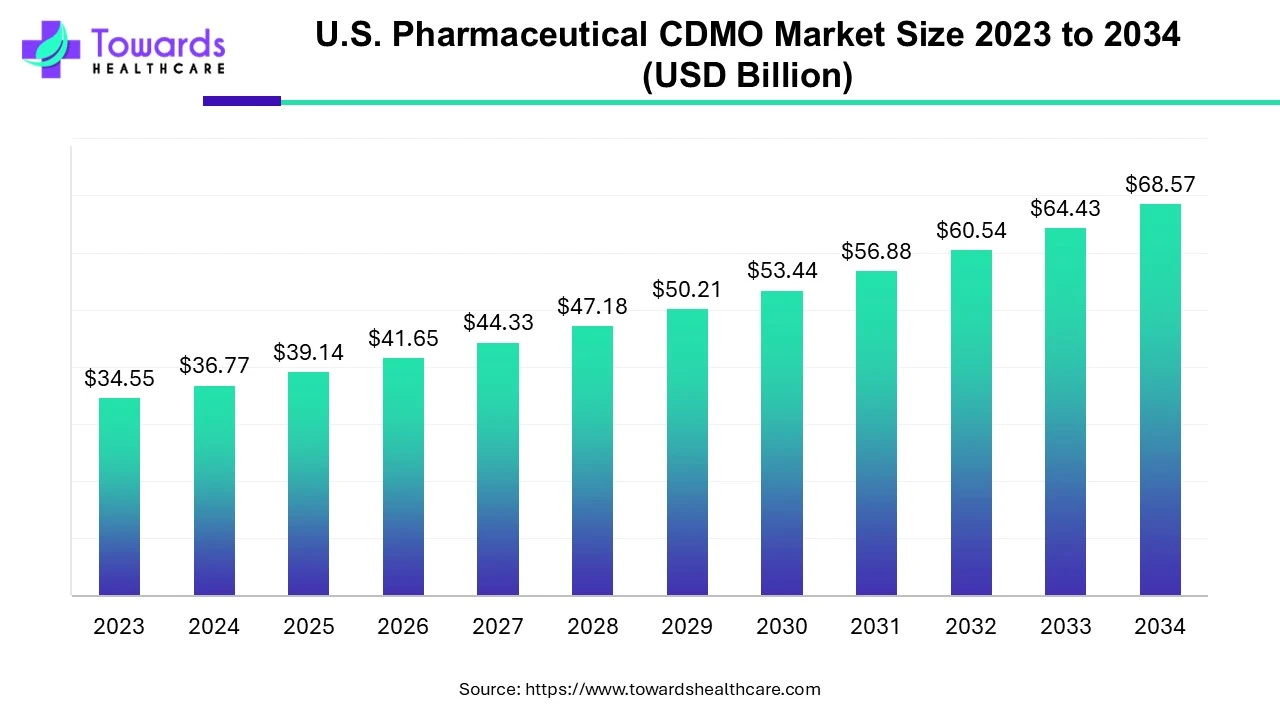
The U.S. pharmaceutical CDMO market size is calculated at USD 39.13 billion in 2025, grew to USD 41.65 billion in 2026, and is projected to reach around USD 72.98 billion by 2035. The market is expanding at a CAGR of 6.43% between 2026 and 2035. Rising mergers and acquisitions, growing research and development, and technological advancements drive the market.
A pharmaceutical contract development and manufacturing organization (CDMO) is an entity that provides specialized services to pharmaceutical companies related to drug development and manufacturing. The wide range of services offered by CDMOs includes formulation development, regulatory support, clinical trial services, product packaging, supply chain management, quality assurance, and technology transfer solutions. Outsourcing enables companies to have cost-effective large-scale production, eliminating the need to set up their facility. CDMOs possess the expertise and technological capabilities to assist companies in major aspects of drug development. This allows companies to focus more on research and marketing.
The growing research and development activities in pharmaceutical companies increase the demand for pharmaceutical CDMOs. The increasing collaboration and mergers & acquisitions majorly contribute to the market. The burgeoning pharmaceutical sector promotes the use of CDMO services. Additionally, technological advancements govern the latest innovations in services offered by CDMOs, potentiating market growth. The increasing investments in pharmaceutical research and manufacturing support the development of more CDMOs.
Increasing Advanced Therapies
Due to the growing diseases, there is a rise in the development of cell and gene therapies along with biologics and biosimilar which is increasing the collaboration with the U.S. pharmaceutical CDMOs to leverage their services.
Growth in Clinical Trials
The growing advancements in the development of immunotherapies, personalized medicine, and orphan drugs are driving their clinical trials, which is increasing the demand for the U.S. pharmaceutical CDMOs for their regulatory expertise.
Growing Outsourcing Trends
The expanding outsourcing trend in the US is increasing the partnership with the pharmaceutical CDMOs to decrease the capital investments, leverage regulatory expertise, and accelerate the product development.
Pharmaceutical CDMOs face challenges related to complexity in manufacturing and demand to reduce the time to market for drug products. Hence, CDMOs leverage artificial intelligence (AI) to overcome such difficulties and provide advanced services. AI helps CDMOs to optimize critical process parameters, streamline development timelines, and enhance product quality. AI introduces automation in the entire development cycle, minimizing human intervention and reducing manual errors. AI can shorten operational cycle times and increase product quality at reduced costs and raw material consumption. AI-enabled predictive analytics can continuously monitor and analyze real-time production data, allowing manufacturers to identify patterns and detect potential errors. This allows for proactive troubleshooting and quick response. Integrating machine learning (ML) algorithms in drug development can enable the analysis of vast amounts of data and suggest suitable excipients for efficient formulations.
Increasing Collaborations
The major growth factor driving the U.S. Pharmaceutical CDMO market is the increasing collaborations of pharmaceutical companies with CDMOs. CDMOs offer advanced facilities to pharmaceutical companies by streamlining the drug development and manufacturing processes. CDMO partnerships make it easier for pharmaceutical companies to access foreign markets through their international presence and regulatory expertise. Such global reach enables companies to navigate international markets seamlessly, ensuring regulatory compliance and market readiness for diverse patient populations worldwide. Collaborating with CDMO also provides firms with the desired flexibility and scalability and accelerates the time-to-market of novel drugs. Moreover, CDMOs also collaborate among themselves and are also involved in mergers & acquisitions activity. These collaborations help CDMOs to expand their client base and access new technologies. These also augment their geographical presence and diversify their portfolio of services.
Supply Chain Disruption
The major challenge of the U.S. pharmaceutical CDMO market is the supply chain disruption. CDMOs can face supply chain disruption, leading to drug shortages. This may further result in lost market opportunities or an inability to satisfy patient demand, thus restricting market growth.
Rising Number of Startups
The future of the U.S. pharmaceutical CDMO market is promising, driven by the rising number of startups. The U.S. pharmaceutical sector is one of the largest in the world owing to suitable regulatory frameworks and increasing investments in pharmaceutical R&D. The rising prevalence of chronic disorders due to the growing geriatric population increases the demand for developing novel diagnostics and therapeutics. All these aspects lead to the rising number of startups in the U.S. Although these startups receive funding from various investors around the nation, they might lack certain specialized and advanced equipment and infrastructure for large-scale manufacturing. Hence, pharmaceutical startups opt for outsourcing services for drug development and manufacturing. CDMOs provide expertise in the relevant field to such startups, helping them develop cutting-edge solutions for complex healthcare problems.
| Key Elements | Scope |
| Market Size in 2026 | USD 41.65 Billion |
| Projected Market Size in 2035 | USD 72.98 Billion |
| CAGR (2026 - 2035) | 6.43% |
| Market Segmentation | By Product, By Workflow, By Application, By End-Use |
| Top Key Players | Adare Pharma Solutions, AGC Biologics, Agilent Technologies, Catalent Pharma Solutions, Exela Pharma Sciences, PCI Pharma Services, Pfizer CentreOne, Scorpius BioManufacturing, Sharp Services, Single Use Support, Thermo Fisher Scientific, UPM Pharmaceuticals, Inc. |
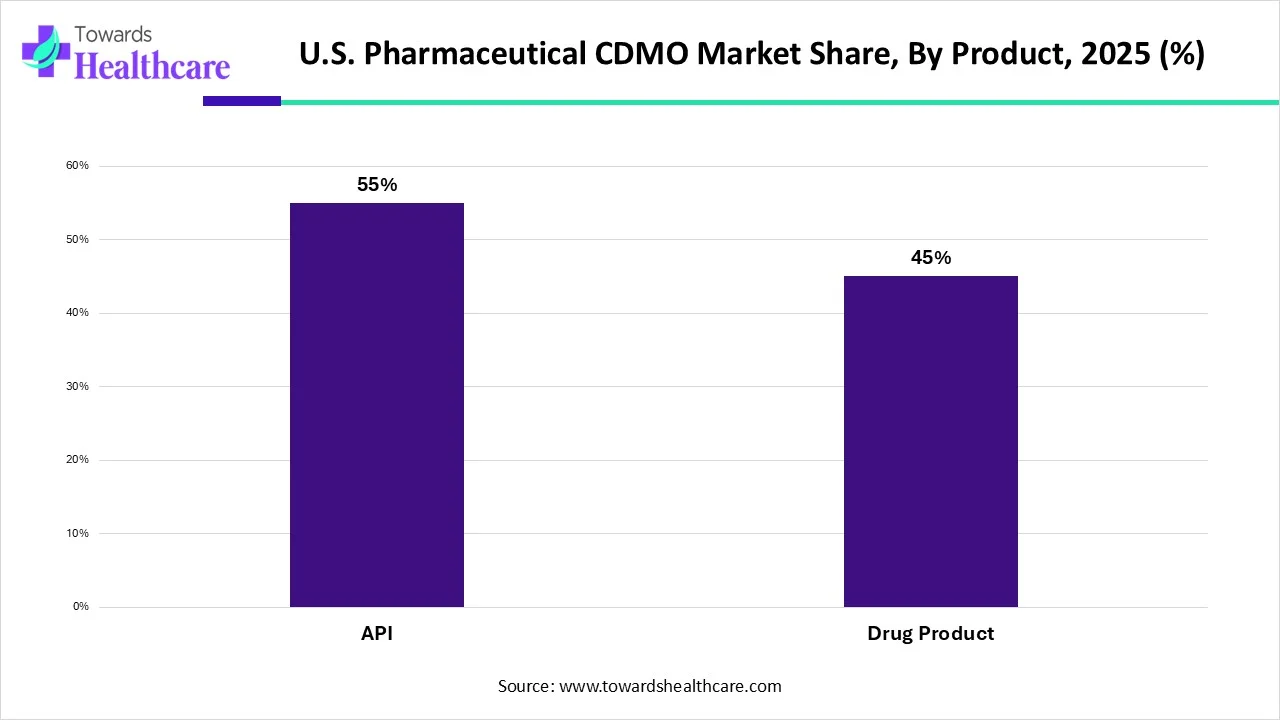
Why Did the API Segment Dominate in the U.S. Pharmaceutical CDMO Market?
By product, the API segment held a dominant presence in the U.S. pharmaceutical CDMO market by 55% in 2025. and is projected to expand rapidly in the market in the coming years. Active pharmaceutical ingredients (API) refer to a wide range of small molecules responsible for therapeutic action. CDMOs provide custom API synthesis and manufacturing, including physicochemical studies, route scouting, route optimization, purification, scale-up, and process development. Hence, CDMOs play an essential role from early process optimization and scale-up to commercial production. Some CDMOs also offer services for the commercial supply of APIs. The growing demand for small molecules as therapeutics and new product launches boost the segment’s growth. CDMOs reduce the time to market approval of APIs.
How the Commercial Segment Dominated the U.S. Pharmaceutical CDMO Market?
By workflow, the commercial segment led the U.S. pharmaceutical CDMO market by 60% in 2025. The growing demand for biologicals and small molecules due to the rising prevalence of chronic disorders augments the segment’s growth. The increasing use of generic pharmaceuticals and personalized medicines also contributes to the segment’s growth. Small to large pharmaceutical companies outsource product development and large-scale manufacturing services to distribute pharmaceuticals to large patient populations. CDMOs are also compliant with regulatory requirements, ensuring product quality and compliance standards for safety, purity, and efficacy. They help companies to maintain the demand-to-supply ratio in different geographical regions.
Which Application Type Segment Held the Dominating Share of the U.S. Pharmaceutical CDMO Market?
By application, the oncology segment held the largest share of the U.S. pharmaceutical CDMO market in 2024. The rising prevalence of cancer in the U.S. and growing research and development activities promote the segment’s growth. The American Cancer Society estimated more than 2 million new cancer cases in the U.S. in 2024. The latest advancements in personalized medicines, cell and gene therapy, monoclonal antibodies, and new drug discovery research for the prevention, diagnosis, and treatment of cancer in the U.S. increase the demand for CDMOs. The increasing funding by government and private organizations for cancer research leads to new product developments, potentiating CDMO services in the U.S. According to the American Society of Clinical Oncology, NIH funding contributes approximately $65 billion to the U.S. economic growth annually.
| Segment | Share 2025 (%) |
| Large Pharmaceutical Companies | 50% |
| Medium Pharmaceutical Companies | 25% |
| Small Pharmaceutical Companies | 25% |
What Made Large Pharmaceutical Companies the Dominant Segment in the U.S. Pharmaceutical CDMO Market?
By end-use, the large pharmaceutical companies segment registered its dominance over the U.S. pharmaceutical CDMO market by 50% in 2025 and is anticipated to grow with the highest CAGR in the market during the studied years. Large pharmaceutical companies conduct multidisciplinary research in various fields that require CDMOs. CDMOs help these companies in process development and manufacturing, eliminating their need to set up a specialized facility based on product requirements. The segmental growth is also attributed to suitable capital investments, new product launches, and state-of-the-art research and development facilities. The increasing number of patents also favors such companies to outsource product development and manufacturing, maintaining trust and confidentiality.
The demand for pharmaceutical CDMOs in the U.S. is rapidly expanding owing to a variety of factors. The increasing number of pharmaceutical CDMOs supports market growth. It is estimated that there are around 300 pharmaceutical CDMOs in the U.S. The rise in the number of new product launches necessitates commercial manufacturing to fulfill the demand, both domestic and international. The U.S. is also one of the top exporters of pharmaceuticals globally. In 2023, the U.S. exported $90.31 billion worth of pharmaceuticals worldwide. Meanwhile, in October 2024, U.S. pharmaceutical exports accounted for $7.69 billion. The increasing investment to conduct advanced research also fuels market growth. President Biden’s budget proposal for FY2025 includes approximately $201.9 billion for R&D. Out of this, around $51.3 billion is estimated for the Department of Health and Human Services. Moreover, rising healthcare expenditure and the growing adoption of advanced technologies are propelling the growth.
R&D
Clinical Trials and Regulatory Approvals
Patient Support and Services

| U.S. Pharmaceutical CDMOs | Headquarters | Services |
| Thermo Fisher Scientific | Waltham, Massachusetts | End-to-end drug substance and drug product manufacturing services |
| Catalent | Somerset, New Jersey | Advanced drug delivery technologies |
| Lonza | Portsmouth, New Hampshire | Mammalian cell culture manufacturing and specialized bioconjugation for antibody-drug conjugates (ADCS) |
| Resilience | San Diego, California | Focus on nucleic acids, vaccines, and biomanufacturing |
| Baxter BioPharma Solutions | Bloomington, Indiana | Sterile injectable manufacturing services |
| Cambrex | East Rutherford, New Jersey | Small molecule API development and manufacturing services |
| Curia | Albany, New York | Integrated R&D and manufacturing services |
| Parimal Pharma Solutions | Sellersville, Pennsylvania | Expertise in high-potency API and integrated oral solid dosage forms |
| PCI Pharma Services | Philadelphia, Pennsylvania | Clinical and commercial packaging, sterilization, and distribution services |
| Alcami Corporation | Wilmington, North Carolina | Analytical testing, formulation development, and sterile fill-finish |
Louis Jerome Weber, Senior Executive at Bora Pharmaceuticals, commented on the opening of its new office in Delaware, U.S., that there is a growing demand for large-scale manufacturing projects having the same diverse and challenging requirements for niche drug products. He added that he is eager to build an ambitious team in North America to continue delivering smart, solution-driven support for clients and their complex modified-release products.
By Product
By Workflow
By Application
By End-Use
April 2026
April 2026
April 2026
April 2026