April 2026

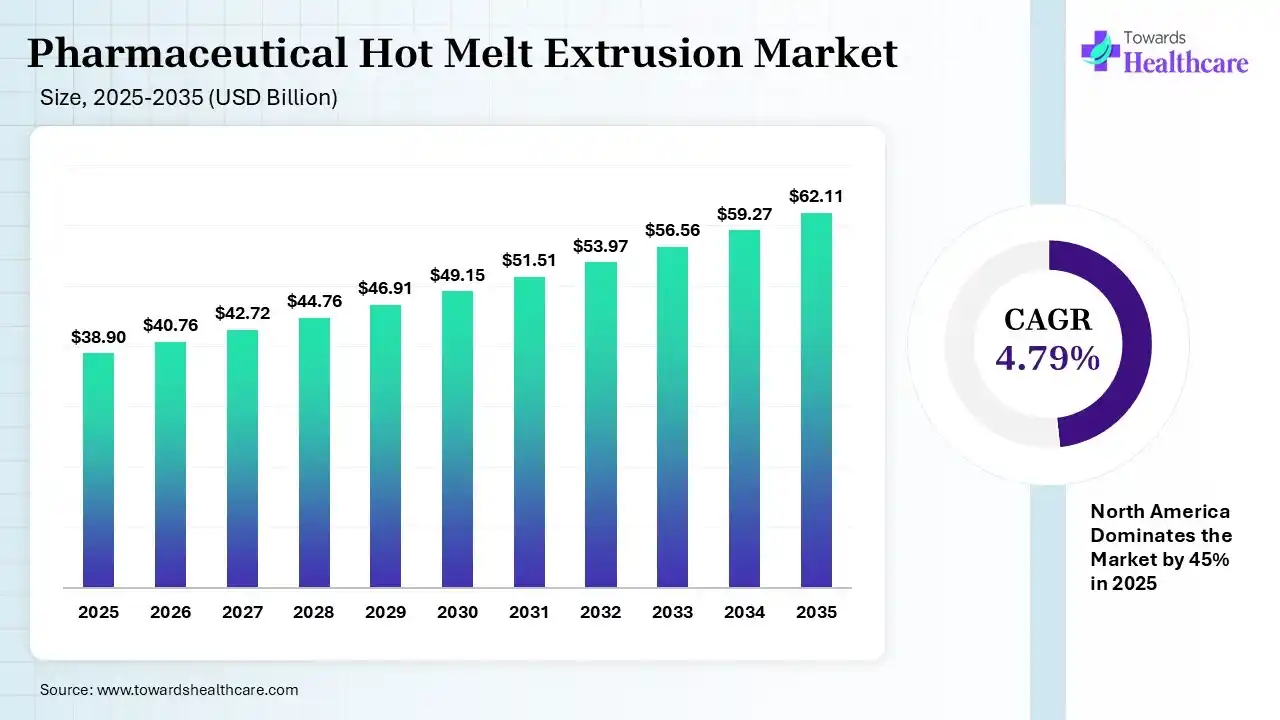
The global pharmaceutical hot melt extrusion market size was estimated at USD 38.9 billion in 2025 and is predicted to increase from USD 40.76 billion in 2026 to approximately USD 62.11 billion by 2035, expanding at a CAGR of 4.79% from 2026 to 2035. The market is expanding as manufacturers use HME to improve drug solubility, bioavailability, and controlled-release performance. Growth is driven by demand for advanced formulations and continuous, solvent-free production, despite high equipment costs and technical challenges.
Pharmaceutical hot melt extrusion is a continuous drug manufacturing process in which active pharmaceutical ingredients (APIs) and polymers are mixed under controlled heat and pressure to form a uniform product with improved solubility, bioavailability, and controlled drug release. The pharmaceutical hot melt extrusion market is witnessing strong growth due to the rising demand for advanced drug formulations that enhance solubility and absorption. Increasing focus on continuous manufacturing, solvent-free processing, and controlled-release dosage forms further supports adoption. Moreover, the growing pipelines of poorly soluble drugs and the need for efficient production technologies are significantly contributing to market expansion.
AI can significantly boost the market by optimizing formulation design, predicting drug–polymer compatibility, and enabling real-time process monitoring. It helps reduce development time, improve product quality, minimize material wastage, and support continuous manufacturing, thereby increasing the adoption of HME technology across pharmaceutical production and research.
| Table | Scope |
| Market Size in 2026 | USD 40.76 Billion |
| Projected Market Size in 2035 | USD 62.11 Billion |
| CAGR (2026 - 2035) | 4.79% |
| Leading Region | North America by 45% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Type, By Application, By Dosage Form, By End-Use, By Region |
| Top Key Players | Baker Perkins Ltd., Coperion GmbH, Leistritz AG, Milacron Holdings Corp., Thermo Fisher Scientific, Inc., Xtrutech Ltd. |
The Twin-Screw Extruder Segment Dominated the Market in 2025
The twin-screw extruder segment dominated the pharmaceutical hot melt extrusion market with a share of 65% in 2025 due to its superior mixing efficiency, precise temperature control, and ability to process complex drug polymer blends uniformly. Its continuous processing capability, scalability for commercial production, and suitability for poorly soluble and controlled-release formulations made it the preferred choice among pharmaceutical manufacturers and contract development organizations.
The single-screw extruder segment held the 35% of market in 2025, and is expected to grow at the fastest CAGR in the market during the forecast period, owing to its lower installation cost, easy operation, and suitability for conventional pharmaceutical formulations. It is projected to register the fastest CAGR during the forecast period due to increasing adoption by small-scale manufacturing, growing pilot production activities, and rising demand for cost-efficient extrusion solutions in drug development.
The Pharma Companies Segment Led the Market in 2025 with the Largest Share
The pharma companies segment led the pharmaceutical hot melt extrusion market with shares of 50% in 2025 due to high investment in advanced drug formulation technologies, large-scale production capabilities, and strong demand for improved drug delivery systems. These companies widely adopted hot melt extrusion for enhancing solubility, bioavailability, and controlled-release profiles, supported by continuous R&D activities and increasing development of complex pharmaceutical products.
The contract manufacturing organizations (CMOs) segment held the second-largest share of 30% of the market in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to rising outsourcing of drug development and manufacturing services. Pharmaceutical companies increasingly rely on CMOs for cost efficiency, technical expertise, scalability, and faster time-to-market, which is significantly boosting demand for hot melt extrusion services.
The research laboratory segment held 15% of the pharmaceutical hot melt extrusion market share in 2025 due to increasing focus on drug formulation studies, early-stage development, and testing of novel delivery systems. Rising investment in pharmaceutical R&D, growing academic industry collaborations, and the need to improve solubility and bioavailability of complex molecules are driving demand for hot melt extrusion systems in research laboratories. Additionally, laboratories use this technology for pilot trials and formulation optimization before commercial-scale production.
The Solid Dosage Forms Segment Led the Market in 2025 with the Largest Share
The solid dosage forms segment dominated the pharmaceutical hot melt extrusion market with a share of 70% in 2025 due to the widespread use of tablets, capsules, and granules in pharmaceutical treatments. Hot melt extrusion is highly effective in enhancing the solubility, stability, and controlled-release properties of solid formulations, making it the preferred choice for manufacturers. Its ease of storage, longer shelf-life, and high patient compliance further supported segment leadership.
The semi-solid/others segment held 30% share in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to rising demand for innovative drug delivery formats such as topical gels, implants, films, and transdermal systems. Hit melt extrusion supports precise formulation and enhanced drug release in these applications, while increasing focus on patient-centric and specialty therapies is further accelerating segment growth.
The Pharmaceutical Manufacturing Segment held a dominant position in the Market in 2025
The pharmaceutical manufacturing segment held a dominant position in the pharmaceutical hot melt extrusion market with a share of 55% in 2025 due to its large-scale production capacity and extensive use of hot melt extrusion in commercial drug manufacturing. Manufacturers increasingly rely on this technology to improve drug solubility, ensure batch consistency, and support continuous processing of tablets, capsules, and controlled-release formulations. Rising demand for advanced therapies and efficient production workflows further strengthened the segment’s leading market position.
The CDMOs segment held the second-largest share of 30% of the market in 2025 and is expected to grow at the fastest CAGR in the market during the forecast period due to increasing outsourcing of drug development, formulation, and commercial manufacturing by pharmaceutical companies. Their specialized technical expertise, cost-efficient production capabilities, and ability to accelerate time-to-market for complex formulations are driving strong demand, particularly for hot melt extrusion-based drug delivery solutions.
The R&D institutes segment held 15% of the pharmaceutical hot melt extrusion market share in 2025 due to increasing investments in pharmaceutical research, formulation innovation, and early-stage drug development. These institutes extensively use hot melt extrusion to study drug polymer compatibility, improve solubility, and develop novel delivery systems. Growing collaborations with pharmaceutical companies and rising focus on personalized medicine and advanced therapeutics are further accelerating segment growth during the forecast period.
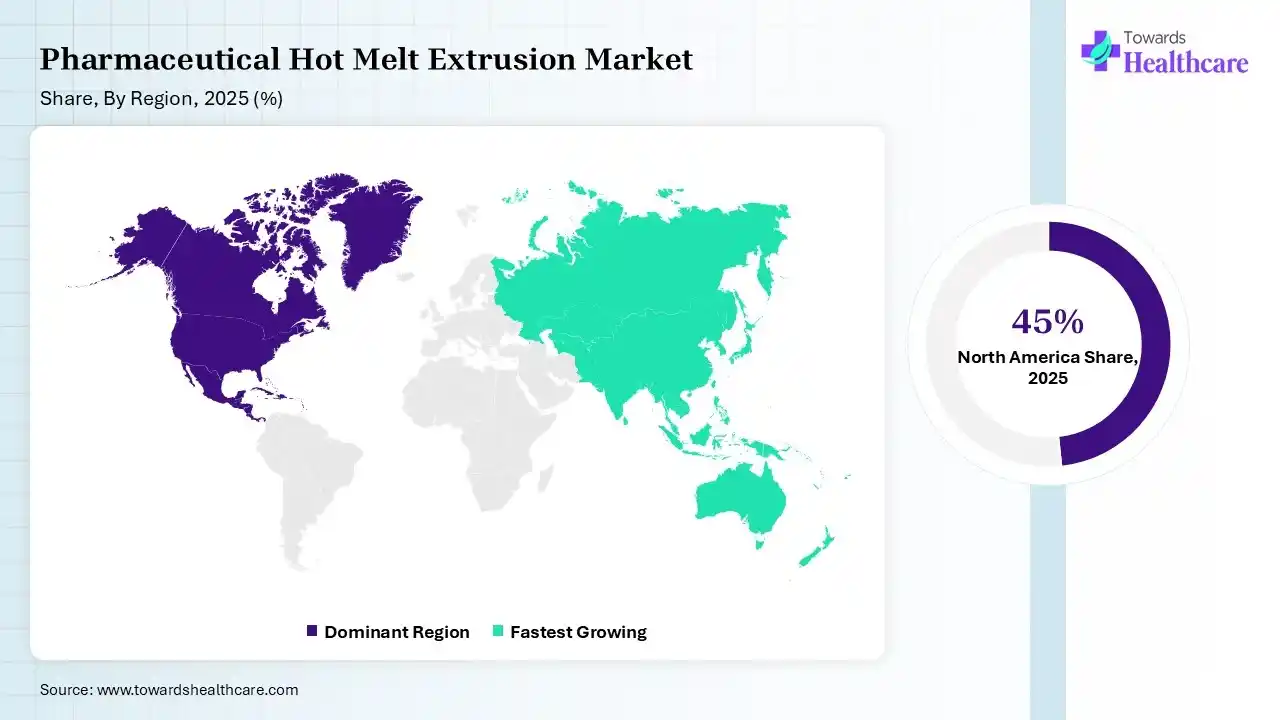
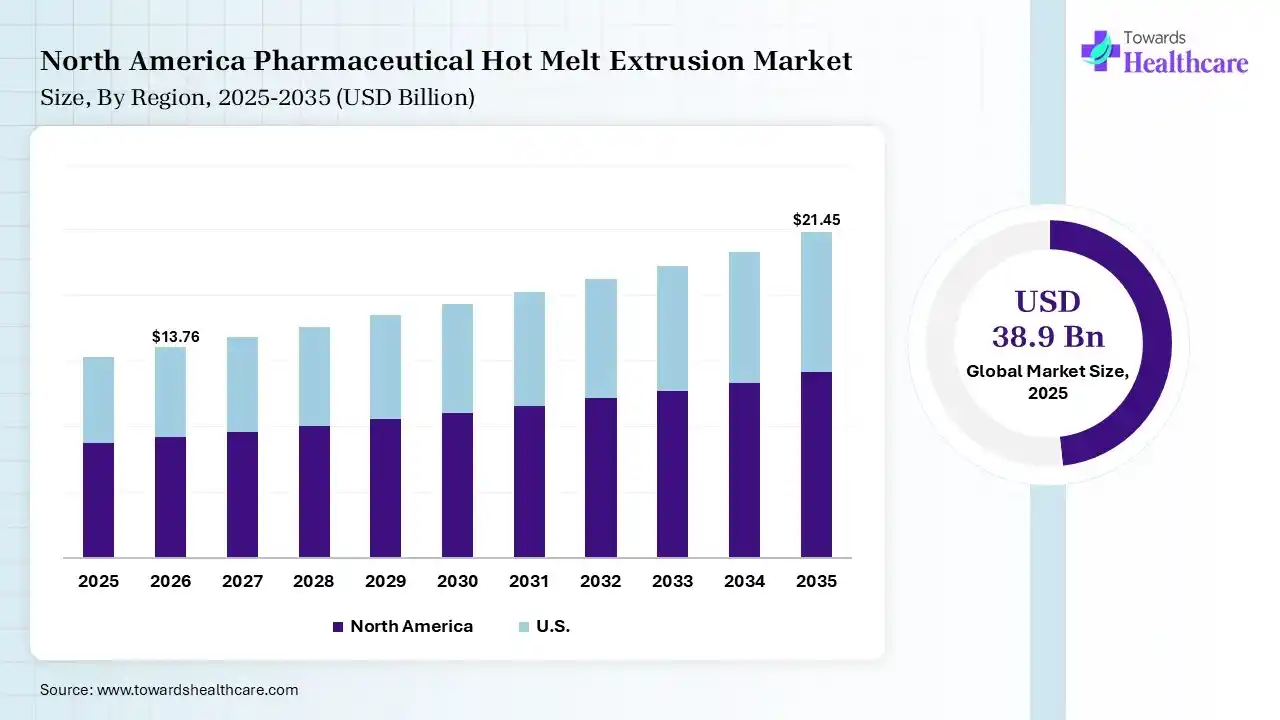
North America dominated the pharmaceutical hot melt extrusion market with a revenue share of 45% in 2025 due to its strong presence of global pharmaceutical manufacturers, advanced research capabilities, and rapid adoption of innovative drug formulation technologies. High spending on drug development, increasing focus on bioavailability enhancements, and widespread use of continuous manufacturing processes further support the region’s leading position in the market.
U.S. Market Trends
The U.S. pharmaceutical hot melt extrusion market is leading due to its advanced pharmaceutical manufacturing ecosystem, strong focus on formulation innovation, and substantial investment in drug research. Increasing demand for enhanced bioavailability solutions, rapid adoption of continuous production technologies, and the presence of specialized CDMOs and research institutions continue to strengthen the country’s dominant position.
Asia Pacific held 20% of the market share and is anticipated to grow at the fastest CAGR during the forecast period due to the rapid expansion of pharmaceutical manufacturing, increasing investments in drug research, and rising adoption of advanced production technologies. The growing presence of CDMOs, cost-effective manufacturing capability, and strong demand for innovative drug formulation across emerging economies are further accelerating regional market growth.
India Market Trends
India is expected to grow at the fastest CAGR during the forecast period due to its rapidly expanding pharmaceutical manufacturing sector, rising investments in drug research, and strong presence of generic drug products and CDMOs. Cost -effective production capabilities, increasing adoption of advanced formulation technologies, and supportive government initiatives for pharmaceutical innovation are further driving market growth across the country.
R&D
Clinical Trials
Formulation and Final Dosage Preparation
| Companies | Headquarters | Offerings |
| Baker Perkins Ltd. | Peterborough, United Kingdom | Peterborough, United Kingdom |
| Coperion GmbH | Stuttgart, Germany | Offers advanced extrusion machinery, precision feeding systems, and process solutions for solubility enhancement and controlled-release drug formulations. |
| Leistritz AG | Nuremberg, Germany | Specializes in high-performance twin-screw extruders designed for pharmaceutical R&D, pilot testing, and commercial production applications. |
| Milacron Holdings Corp. | Ohio, U.S. | Delivers extrusion and polymer processing equipment suitable for pharmaceutical blending, dosage form manufacturing, and specialty formulation applications. |
| Thermo Fisher Scientific, Inc. | Massachusetts, U.S. | Offers laboratory-scale extruders, analytical tools, and formulation development systems for research and process optimization. |
| Xtrutech Ltd. | Blackburn, United Kingdom | Develops customized extrusion systems and continuous manufacturing solutions for formulation development and production scale-up. |
Strengths
Weaknesses
Opportunities
Threats
By Type
By Application
By Dosage Form
By End-Use
By Region
April 2026
March 2026
March 2026
March 2026