
The global cell and gene therapy manufacturing isolators market size is expected to be worth around USD 321.3 million by 2035, from USD 140.16 million in 2025, growing at a CAGR of 8.65% during the forecast period from 2026 to 2035.
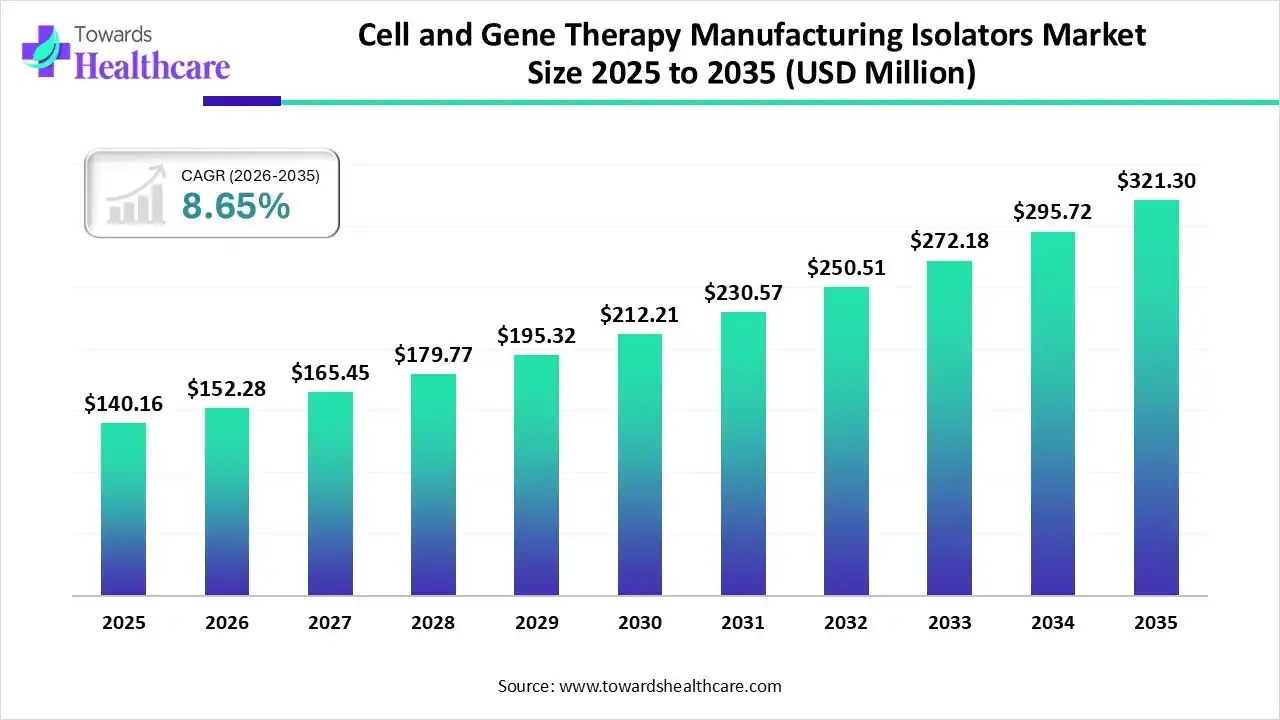
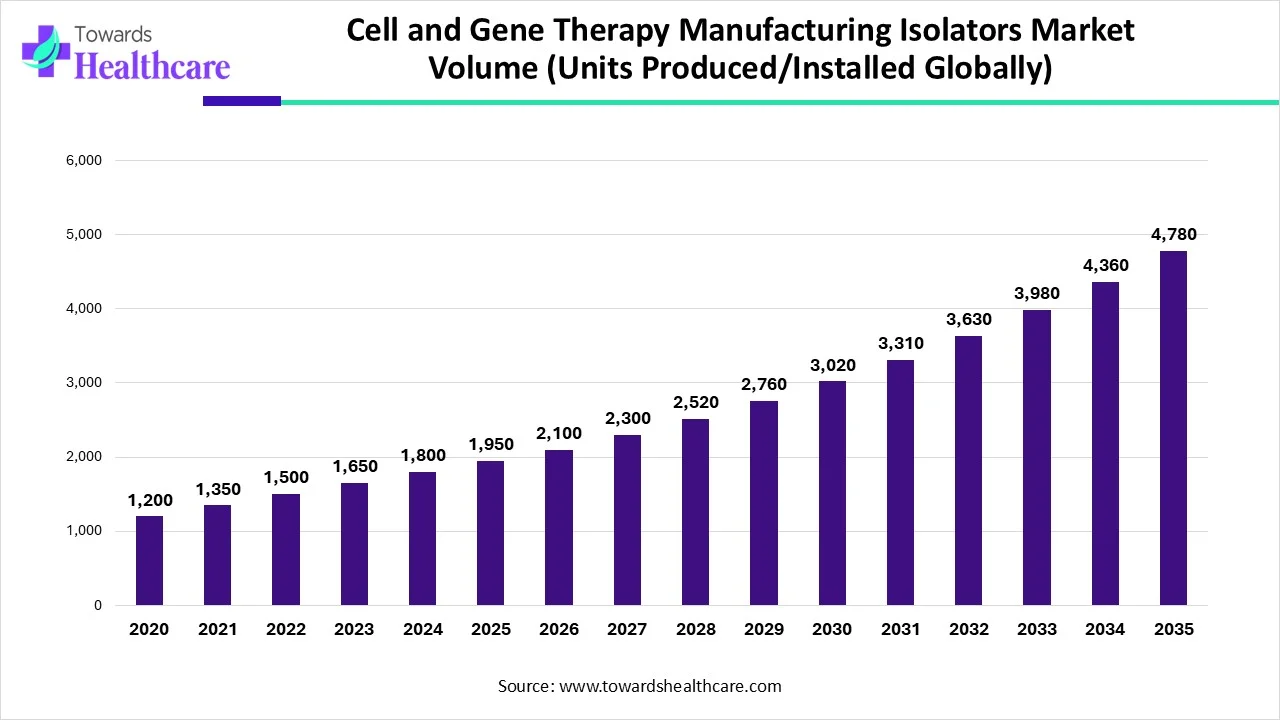
The cell and gene therapy manufacturing isolators market is experiencing robust growth, driven by the increasing development of cell and gene therapy (CGT) products, rising investments, and technological advancements. The increasing collaboration among key players enables them to access advanced technologies and manufacture proprietary products. Regulatory agencies and manufacturers make constant efforts to fulfill global patient needs, accelerating CGT manufacturing.
| Key Elements | Scope |
| Market Size in 2026 | USD 152.28 Million |
| Projected Market Size in 2035 | USD 321.3 Million |
| CAGR (2026 - 2035) | 8.65% |
| Leading Region | North America |
| Market Segmentation | By Isolator Type, By Application Process, By Technology/Design Feature, By End-Use Facility, By Region |
| Top Key Players | Thermo Fisher Scientific, Inc., Bio-Rad Laboratories, Inc., Miltenyi Biotec, Takara Bio, SKAN, Comecer, Ecolab, Akadeum Life Sciences, Sony Biotechnology, Extract Technology |
The cell and gene therapy manufacturing isolators market refers to the development and distribution of tailored isolators for large-scale manufacturing of CGT. CGT isolators provide closed environments and enhance sterility by reducing manual intervention and minimizing contamination risks. They offer modular, sterile, automation-compatible environments that support both autologous and allogeneic product manufacturing at clinical sites. They lower facility requirements and enable real-time quality control (QC) testing.
Integrating artificial intelligence (AI) into isolators introduces automation and leads to smarter, data-driven manufacturing. AI and machine learning (ML) algorithms transform CGT production by reducing errors, streamlining workflows, and making life-changing therapies more accessible. They enable real-time monitoring of CGT manufacturing, enabling manufacturers to get real-time updates. AI-based predictive analytics allow manufacturers to detect errors and make proactive decisions.
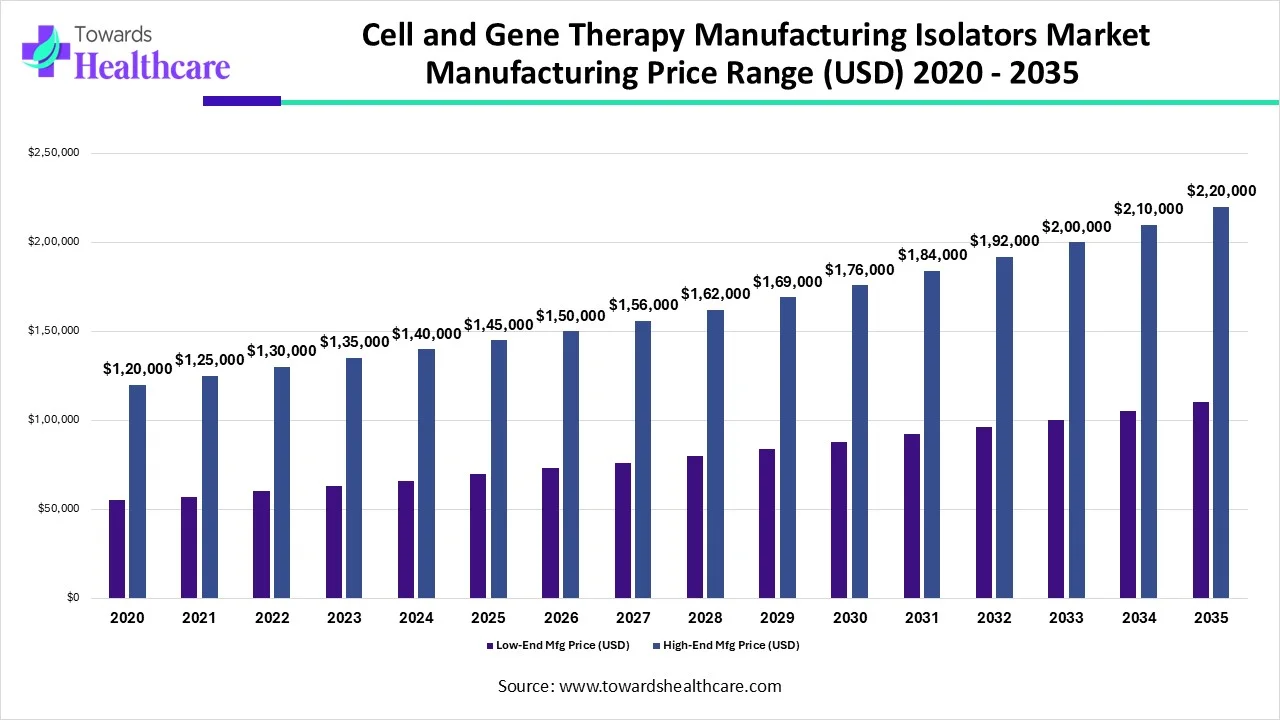
Rationale for ranges:
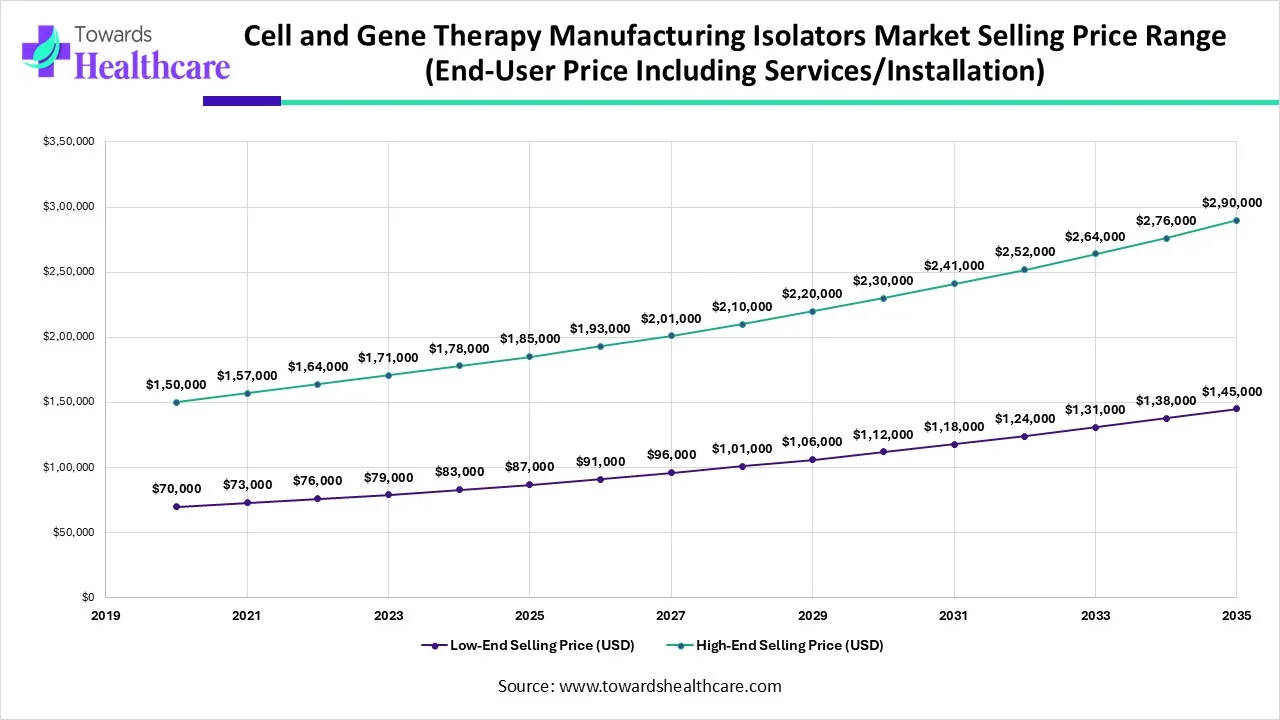
Key Considerations:
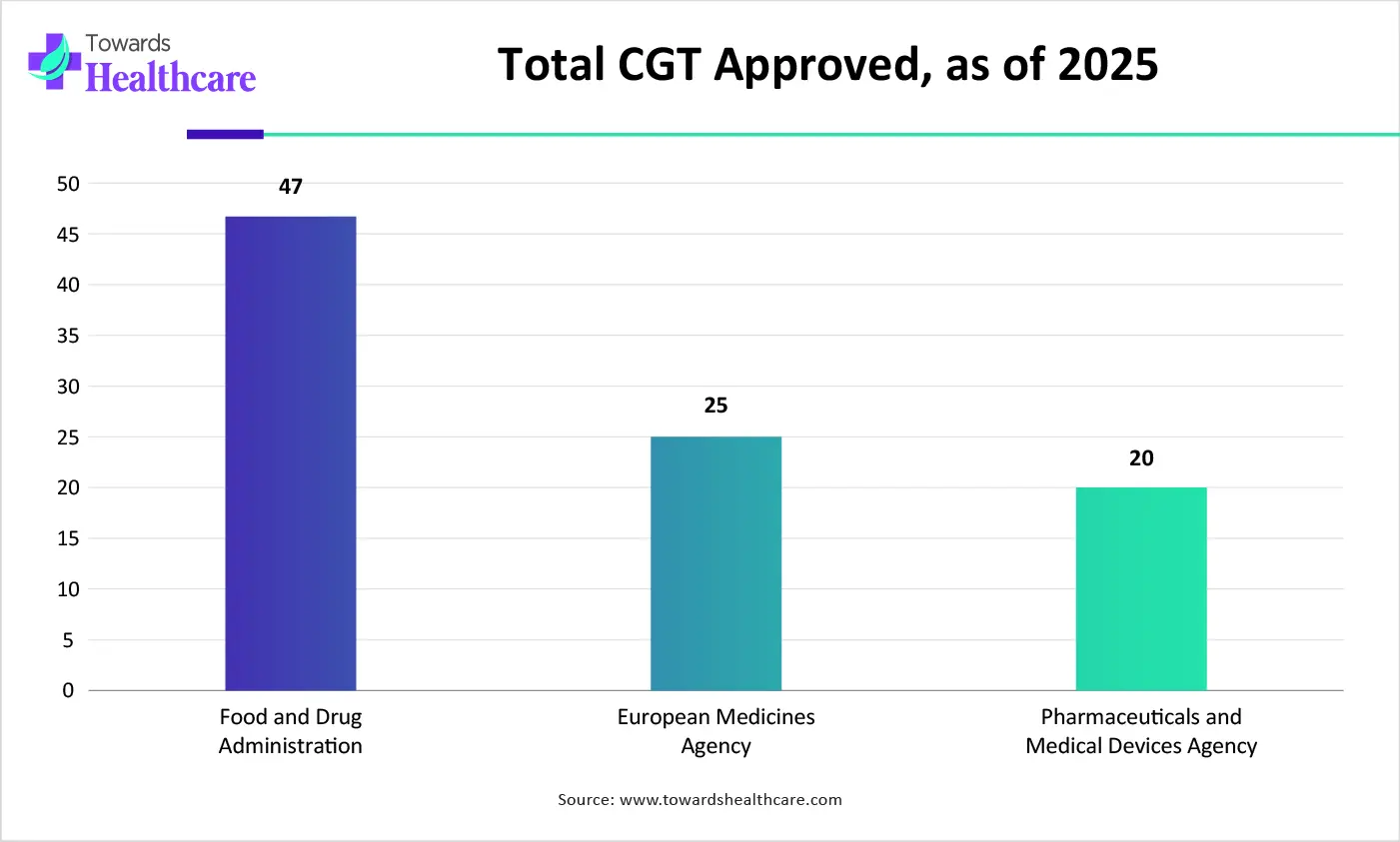
| Regulatory Agencies | Total CGT Approved, as of 2025 |
| Food and Drug Administration (FDA) | 47 |
| European Medicines Agency (EMA) | 25 |
| Pharmaceutical and Medical Devices Agency (PMDA) | 20 |
Which Isolator Type Segment Dominated the Market?
The negative pressure isolators segment held a dominant position in the cell and gene therapy manufacturing isolators market in 2025, due to the high demand for operator protection. Negative pressure isolators provide personnel or environmental protection by containing hazardous substances. They minimize airborne toxins from escaping into the cleanroom equipment. The increasing development of highly potent active pharmaceutical ingredients (HPAPIs) potentiates the demand for negative pressure isolators.
Dual-Pressure/Flexible Isolators
The dual-pressure/flexible isolators segment is expected to grow at the fastest CAGR in the market during the forecast period. Dual-pressure/flexible isolators possess the characteristics of both types of isolators, providing superior benefits. They protect products, personnel, and the environment, eliminating the need for different isolators.
How the Aseptic Processing Segment Dominated the Market?
The aseptic processing segment held the largest revenue share of the cell and gene therapy manufacturing isolators market in 2025, due to the growing need to prevent contamination. Isolators help maintain sterility throughout the complex, multi-step processes, where terminal sterilization isn’t possible. Closed aseptic systems or isolators offer advantages, such as greater process scalability, robustness, flexibility, and consistency with fewer batch failures. They minimize the risk of cross-contamination with microbes.
Viral Vector Production
The viral vector production segment is expected to grow with the highest CAGR in the market during the studied years. Isolators are increasingly used in viral vector production, especially in aseptic fill-finish procedures. This helps manufacturers maintain the sterility of CGT products and maintain a closed environment throughout their manufacturing. Viral vectors are essential components of gene therapy to deliver modified genes directly to the targeted site.
Why Did the Glovebox Isolators Segment Dominate the Market?
The glovebox isolators segment contributed the biggest revenue share of the cell and gene therapy manufacturing isolators market in 2025, due to the ability to manipulate processes without human interference. Glovebox isolators enable manufacturers to conduct the desired operations within a controlled microenvironment. They are specifically designed to thwart contamination from the environment or operator. They allow manual handling of the objects with minimal filtration.
Restricted-Access Barrier Systems (RABS)
The restricted-access barrier systems (RABS) segment is expected to expand rapidly in the market in the coming years. The demand for RABS is increasing due to the presence of safety-locked doors, a rigid machine enclosure, and glove ports. RABS are easy to install, easy to validate, and control temperature and humidity across stringent conditions. They also provide full operator protection, minimizing contamination risk from and to the operator.
Which End-Use Facility Segment Led the Market?
The contract development and manufacturing organizations (CDMOs) segment led the cell and gene therapy manufacturing isolators market in 2025, due to the presence of favorable infrastructure and increasing collaboration with biotech companies. CDMOs invest heavily in adopting specialized equipment for CGT manufacturing. Large companies collaborate with CDMO to expand their product pipelines, focusing on their core competencies. Small companies can have access to equipment and expertise.
Academic & Research Institutes
The academic & research institutes segment is expected to witness the fastest growth in the market over the forecast period. Academic & research institutions receive funding from the government and private organizations to conduct advanced research. Funding also helps them purchase CGT isolators for experiments focusing on CGT development. The growing research and development activities and the increasing public-private partnerships propel the segment’s growth.
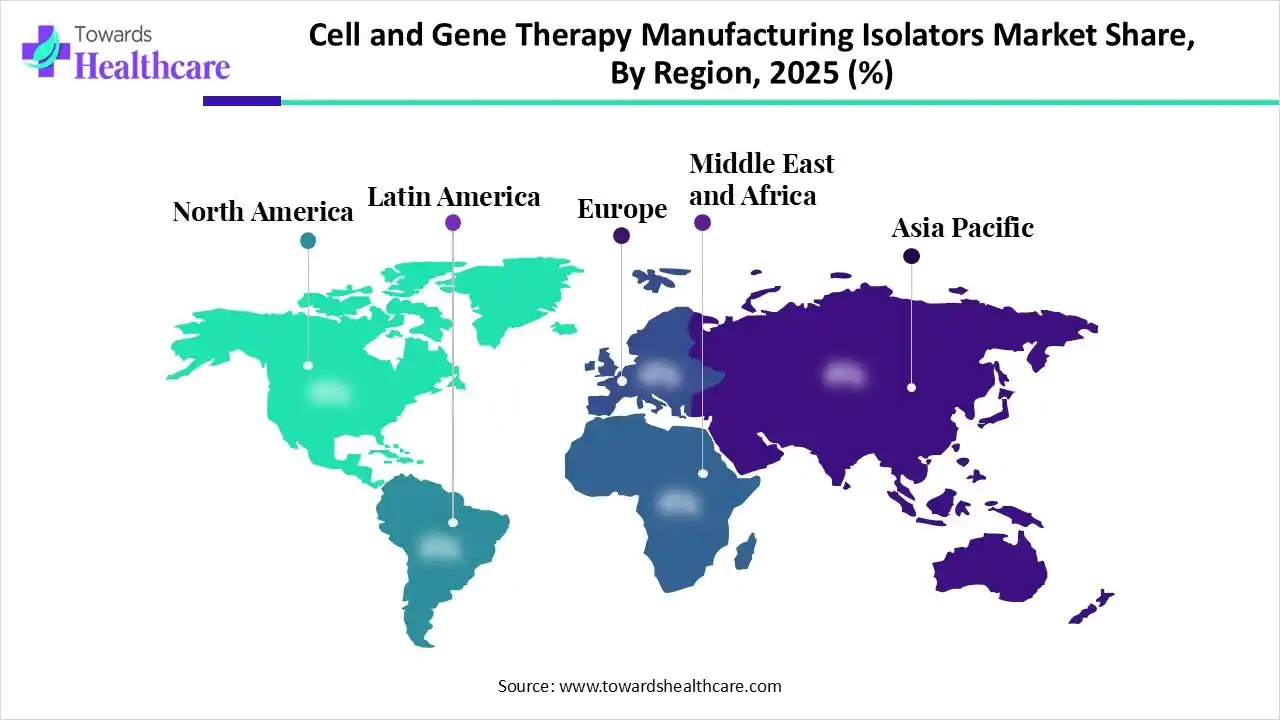
North America dominated the global cell and gene therapy manufacturing isolators market in 2025. The availability of a robust manufacturing infrastructure, the presence of key players, and favorable regulatory support are factors that drive the market in North America. Countries like the U.S. and Canada are home to multiple biotech companies that focus on producing innovative CGT products. They adopt advanced technologies, supporting biotech innovation and strengthening their position in the global CGT market.
Key players, such as Thermo Fisher Scientific, Bio-Rad Laboratories, and Akadeum Life Sciences, are major providers of CGT isolators in the U.S. The U.S. conducts the highest number of clinical trials related to CGT. As of December 2025, a total of 513 clinical trials were registered from the U.S. on the clinicaltrials.gov website.
Asia-Pacific is expected to witness the fastest growth in the cell and gene therapy manufacturing isolators market during the predicted timeframe. The rising prevalence of chronic disorders, the growing geriatric population, and the increasing awareness of early intervention augment the market. Countries like China, India, Japan, and South Korea have a suitable manufacturing infrastructure due to their favorable geographical location and affordable workforce, encouraging foreign companies to set up their facilities in the region. The increasing emphasis on domestic production of pharmaceutical products and biologics necessitates the use of CGT isolators.
The biotech sector in China is booming at a rapid pace, with evolving regulatory landscapes, government initiatives, and a maturing startup ecosystem. More than 130 companies are estimated to produce CGT products in China. As of 2025, the National Medical Products Administration (NMPA) has approved 5 CAR-T cell therapies in China.
Europe is expected to grow at a considerable CAGR in the cell and gene therapy manufacturing isolators market in the upcoming period. European nations are emerging as global hubs for clinical trials and manufacturing of advanced therapy medicinal products (ATMPs). The European Medicines Agency (EMA) regulates the manufacturing and approval of CGTs in Europe. It establishes a favorable regulatory framework to accelerate the development of CGTs. The presence of key biotech companies and the increasing investments also contribute to market growth.
The UK ranks second in Europe in having the largest number of startups for cancer research after France, accounting for 290 companies. Most of the cancer therapeutics’ innovations are in cellular immunotherapy, gene therapy, and image analysis in oncology. The UK also witnessed a surge in equity financing in Q1 2025, reaching £924 million across venture capital and follow-on funding.
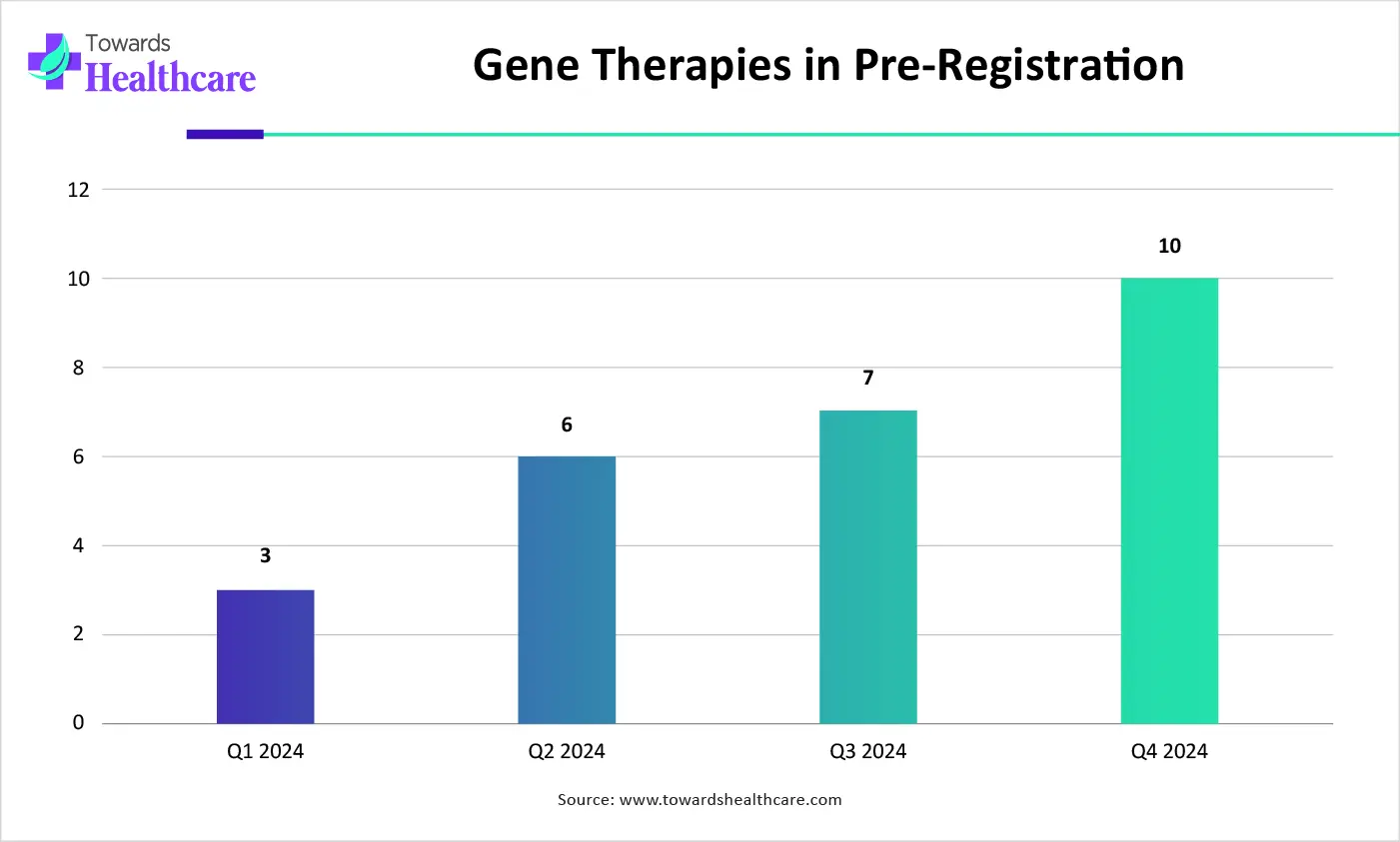
| Period | Number of Gene Therapies in the Pre-Registration Phase |
| Q1 2024 | 3 |
| Q2 2024 | 6 |
| Q3 2024 | 7 |
| Q4 2024 | 10 |

| Companies | Headquarters | Offerings |
| Thermo Fisher Scientific, Inc. | Massachusetts, United States | It offers Dynabeads and MagniSort tube-based cell separation technology for the isolation, expansion, and activation of CGT. |
| Bio-Rad Laboratories, Inc. | California, United States | The ddSEQ Single-Cell Isolator is a droplet-based single-cell isolation system for single-cell gene expression and gene regulation studies. |
| Miltenyi Biotec | Bergisch Gladbach, Germany | It offers a wide range of CGT solutions, including NK cell isolation, expansion, and analysis. |
| Takara Bio | Kusatsu, Japan | It provides kits, reagents, instruments, and services for the comprehensive and seamless manufacturing of CGT. |
| SKAN | Allschwil, Switzerland | Cellana is a highly innovative, cGMP-validated isolator designed for all specific CGT processes. |
| Comecer | Italy | It offers ValueCell COMBI, SOLO, and FLEXY isolators that support cell therapy needs from R&D to much higher volumes in cell factories. |
| Ecolab | Minnesota, United States | The Bioquell Qube Aseptic Isolator is ideal for sterility testing, CGT, small batch production, and drug compounding. |
| Akadeum Life Sciences | Michigan, United States | The company offers buoyant microbubble cell isolation technology to improve the production and engineering efficiency of therapeutic CAR-T cells in a scalable manner. |
| Sony Biotechnology | California, United States | The CGX10 Cell Isolation System is the only fully closed cell isolation system for GMP-compliant cell production and cell sorting applications. |
| Extract Technology | Huddersfield, United Kingdom | Extract Technology’s Cell Therapy Isolators provide an ergonomic and practical alternative to traditional clean rooms for stringent cGMP. |
By Isolator Type
By Application Process
By Technology/Design Feature
By End-Use Facility
By Region