March 2026

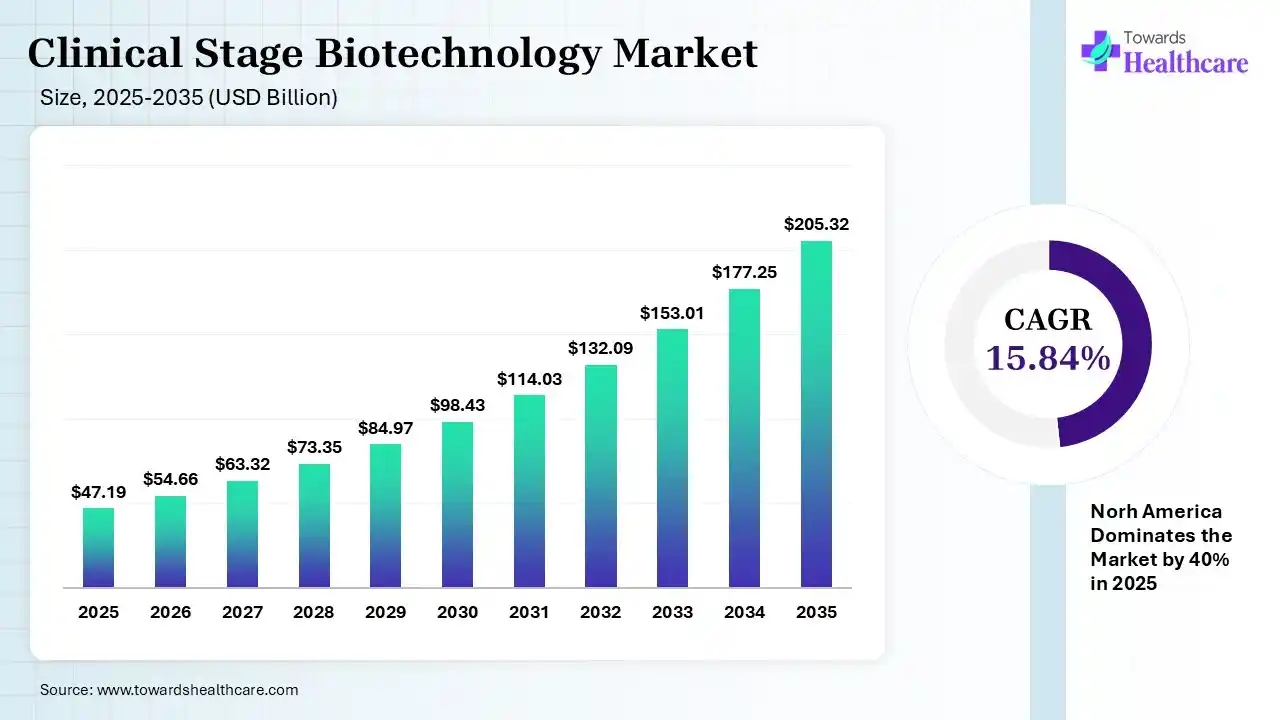
The global clinical stage biotechnology market size was estimated at USD 47.19 billion in 2025 and is predicted to increase from USD 54.66 billion in 2026 to approximately USD 205.32 billion by 2035, expanding at a CAGR of 15.84% from 2026 to 2035.
The clinical stage biotechnology market is growing because this technology provides many advantages, such as improved patient results and targeted therapy, and it offers advanced product safety and efficacy testing.
The clinical stage biotechnology market is growing, as this clinical stage is the most significant milestone for a biotech startup. It includes human testing and is generally divided into three phases, each related to a steep increase in expenses. The increase in biotechnology has transformed pharmaceutical technology by allowing the manufacturing of biopharmaceuticals, involving monoclonal antibodies, recombinant proteins, and gene therapies. Biopharmaceuticals provide intended therapies for challenging diseases like cancer, autoimmune disorders, and genetic conditions. The development in pharmaceutical technology has significantly enhanced healthcare results by offering safer, more efficient, and patient-driven treatments. Technological development has led to the advancement of automated systems, high-output screening techniques, and precision medicine strategies.
Integration of AI-driven technology in clinical-stage biotechnology drives market growth, as AI-driven technology is transforming the significance of biotech, promising to modify precision medicine, clinical trials, and bioprocess optimisation. AI-driven technology has become a driving force for novelty in biotechnology and has sparked significant business interest. AI-driven technology is transforming biotechnology by accelerating development in drug discovery, genomics, healthcare imaging, and personalized medicine, thereby enhancing efficiency and reducing healthcare costs. This solution is transforming biotechnology, accelerating drug discovery, genome sequencing, and targeted medicine. AI-based technology is hastening progress across the whole biomedical pipeline.
Decentralized and Hybrid Trials:
DCTs and hybrid trials provide various benefits, such as enhanced recruitment, patient convenience, and lower costs. This trial has developed as a promising substitute to traditional clinical trials. A decentralized hybrid strategy increases patient participation from the comfort of their homes.
Synthetic Biology in Manufacturing:
Synthetic biology is helpful in pharmaceutical advancement. Synthetic biology allows for the manufacturing of small molecules that are difficult to synthesize with chemical engineering or produce naturally. It has great strength in targeted medicine strategies to fight cancer.
Precision Medicine and Biomarkers:
Precision medicine supports making the prediction of whether a specific treatment works for the patients or not. Precision medicine holds potential for enhancing major aspects of health and healthcare. Design of novel tools for building, analyzing, and sharing big sets of medical information.
| Table | Scope |
| Market Size in 2026 | USD 54.66 Billion |
| Projected Market Size in 2035 | USD 205.32 Billion |
| CAGR (2026 - 2035) | 15.84% |
| Leading Region | North America by 40% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Product Type, By Application, By Phase, By End Use, By Region |
| Top Key Players | Tempus, ElevateBio, Biosplice Therapeutics, Insitro, Recursion Pharmaceuticals, Mirador Therapeutics |
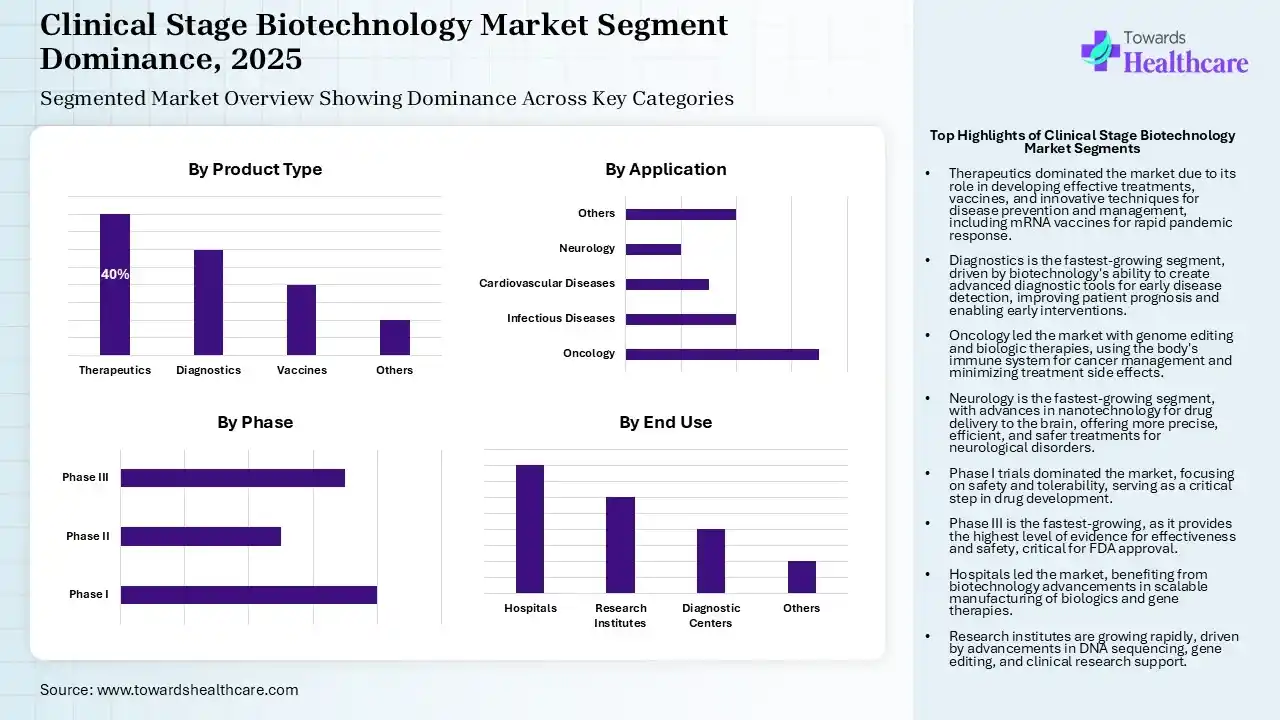
Therapeutics Segment Led the Clinical Stage Biotechnology Market in 2025
| Segment | Share 2025 (%) |
| Therapeutics | 40% |
| Diagnostics | 30% |
| Vaccines | 20% |
| Others | 10% |
Explanation
The therapeutics segment dominated the market in 2025, as it supports offering effective treatments and prevention measures for various diseases via its inventions of new drugs and recombinant vaccines. It is broadly used in the development of many innovative techniques for diagnosing, managing, and preventing diseases. Biotechnology has accelerated the creation of vaccines, including mRNA technology, enabling rapid responses to pandemics and communicable diseases.
Whereas the diagnostics segment is estimated to be the fastest-growing in the market, as biotechnology companies develop accurate diagnostic tools using molecular biology to identify human diseases at early stages. Molecular testing uncovers infections, cancers, or genetic disorders before healthcare symptoms emerge, providing a window for early intervention that enhances prognosis and prevents disease progression.
Oncology Segment Led the Clinical Stage Biotechnology Market in 2025
| Segment | Share 2025 (%) |
| Oncology | 35% |
| Infectious Diseases | 20% |
| Cardiovascular Diseases | 15% |
| Neurology | 10% |
| Others | 20% |
Explanation
The oncology segment dominated the market in 2025, as genome editing is a far better and novel technique to manage cancer. Experts used engineered nucleases that have particular domains that can bind to the target site, followed by their cleavage. Biologic therapies apply the body's own power to manage cancer. These therapies use the body's immune system to get rid of cancer or to reduce treatment side effects. Biological therapy is an efficient substitute treatment for various cancers, as it causes fewer side effects than other treatments.
Whereas the neurology segment is the fastest-growing in the market, as biotechnology is used to develop novel therapies that address the underlying causes of these diseases. It provides other novel services to the problem of neurological disorders. increasing the applications of nanotechnology to deliver drugs straight to the brain, bypassing the blood-brain barrier, and growing the efficiency of treatment. It has the potential to transform the field of medicine. By attaching the power of biotechnology, investigators are allowed to develop therapies that are more precise, efficient, and safe than outdated treatments.
Phase I Segment Led the Clinical Stage Biotechnology Market in 2025
| Segment | Share 2025 (%) |
| Phase I | 40% |
| Phase II | 25% |
| Phase III | 35% |
Phase I segment dominated the market in 2025, as phase I clinical trials allow advanced treatments to progress further in drug advancement or halt that process altogether. Phase I is focused on safety and tolerability, making it the linchpin for any future advancement. This phase significantly involves a small cohort of 20 to 100 healthy volunteers or patients, based on the therapeutic region. The main aim is to find how the investigational compound interacts with human physiology, focusing on pharmacokinetics and pharmacodynamics.
Whereas the phase III segment is the fastest-growing in the market, as phase III trials are the standard method to find a novel standard for treatment. Phase III trials offer the highest level of evidence for presenting the effectiveness of novel treatments or interventions. Phase III trials offer the bulk of information required for the package insert and labeling of a medicine, after it is FDA-approved. Phase III clinical trials are huge, late-stage research comparing novel treatments to standard therapies to establish effectiveness and monitor safety.
Hospitals Segment Led the Clinical Stage Biotechnology Market in 2025
| Segment | Share 2025 (%) |
| Hospitals | 40% |
| Research Institutes | 30% |
| Diagnostic Centers | 20% |
| Others | 10% |
Explanation
The hospitals segment dominated the market in 2025, as biotechnology has been involved in health care by allowing the scalable manufacturing of monoclonal insulin, antibodies, vaccines, and gene therapy products. Novelties in biomedical science lead to smarter therapeutic tools and drug delivery technology. Advances in biotechnology enable the bio-processing of waste, turning biological technology into tools for conservation.
Whereas the research institutes segment is the fastest-growing in the market, recent developments in biotech technology have allowed for more accessible DNA sequencing, quicker DNA synthesis, and more precise gene-editing tools. Clinical research services provider provides intended support to biotechnology and pharmaceutical organizations throughout all stages of their clinical trials.
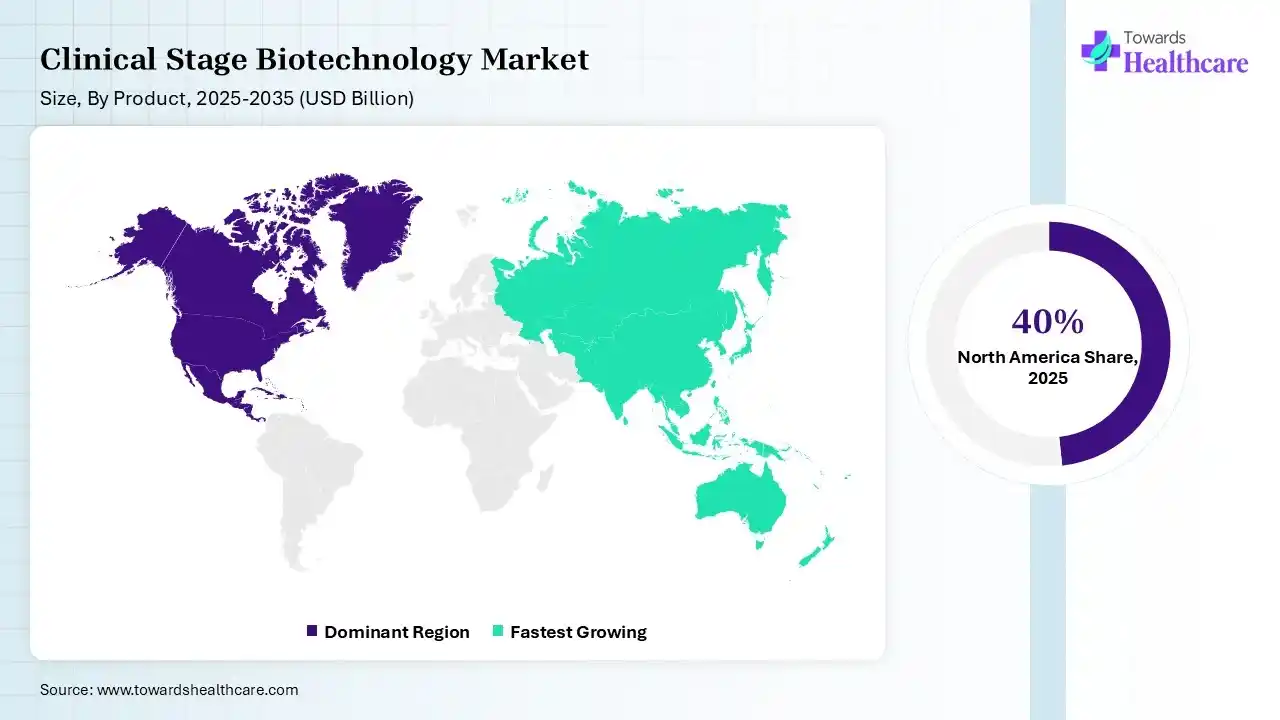
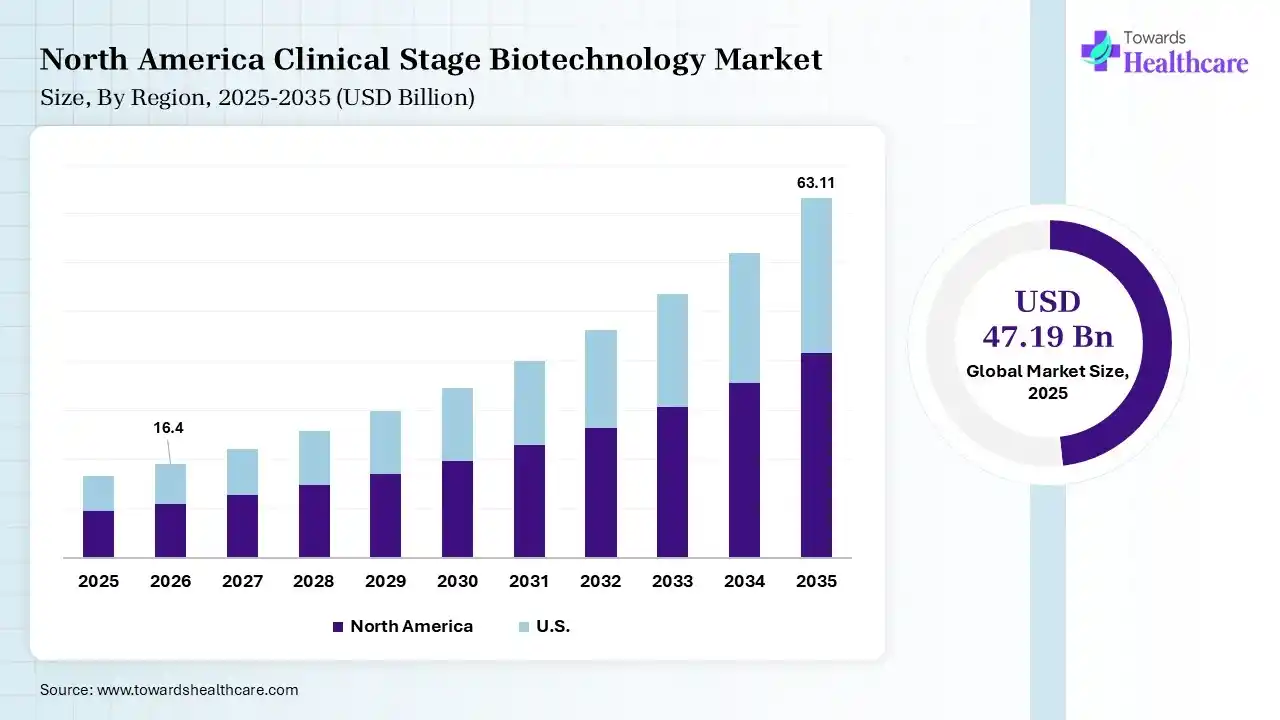
In 2025, North America dominated the clinical stage biotechnology market by 40% because the federal government offers funding for the national Medicare program for adults age 65 and older and people with disabilities, as well as different programs for veterans and low-income people, including Medicaid and the Children’s Health Insurance Program. Major organizations such as San Francisco and Boston provide concentrated talent and infrastructure, reinforcing US leadership. Top-tier biotech clusters are driven by intense VC funding, advanced academic research, and high lab space awareness, which drives the growth of the market.
For Instance,
U.S. Market Trends
In the U.S., increasing spending in R&D and the vast availability of venture capital drive the long, capital-intensive clinical trials. Clinical trials generate vital insights that deepen understanding of health. Academic researchers are generating a plethora of insights and knowledge that have the massive strength to significantly improve patient care.
Asia Pacific is expected to see rapid growth in the clinical-stage biotechnology market, as large patient pools associated with increasing prevalence rates for particular diseases, allowing rapid recruitment for trials. Asia-Pacific countries, such as Japan, India, Australia, and South Korea, present higher patient enrolment rates in clinical trials as compared to many Western nations. Public-private partnerships (PPPs) in the Asia-Pacific region encourage cooperative research initiatives, data exchange, and capacity building in clinical trial management.
India Market Trends
In India, clinical trials are more than 50 % inexpensive as compared to industrialized countries. India is a hub for numerous reputed SMOs providing world-class solutions. Presence of major organizations that work closely with pharmaceutical companies and CROs to deliver seamless integration. India has the maximum number of US FDA-approved production plants.
R&D:
Manufacturing Processes:
Patient Services:

| Company | Headquarters | Latest Update |
| Tempus | United States | In January 2025, Tempus AI, Inc., a technology organization leading the adoption of AI to advance precision medicine, announced the launch of Paige Predict, a suite of cutting-edge digital pathology applications that analyze hematoxylin and eosin (H&E) whole slide images to support and inform testing decisions. |
| ElevateBio | United States | ElevateBio has continued to advance its research related to the breakthrough CRISPR gene editing technology, and treatments ranging from cancer to multiple sclerosis, diabetes, and Huntington’s Disease. |
| Biosplice Therapeutics | United States | In January 2026, Biosplice Therapeutics, Inc., a clinical-stage biotechnology company pioneering first-in-class therapeutics based on small-molecule inhibition of CLK/DYRK kinases, announced that it had submitted its NDA to the FDA for approval of its new therapy for the treatment of knee osteoarthritis. |
| Insitro | United States | In January 2026, AI-based therapeutics company insitro acquired biotechnology organization CombinAbleAI to complete its AI-based platform for drug discovery and design. |
| Recursion Pharmaceuticals | United States | In February 2026, Recursion, a leading clinical-stage TechBio organization decoding biology to radically improve lives, reported business updates and economic results. |
| Mirador Therapeutics | United States | In January 2026, Mirador accelerated its multi-asset clinical pipeline in immuno-fibrotic disease; it closed a $250 million series B with premier investors. |
Strengths
Weaknesses
Opportunities
Threat
By Product Type
By Application
By Phase
By End Use
By Region
March 2026
March 2026
March 2026
March 2026
