March 2026

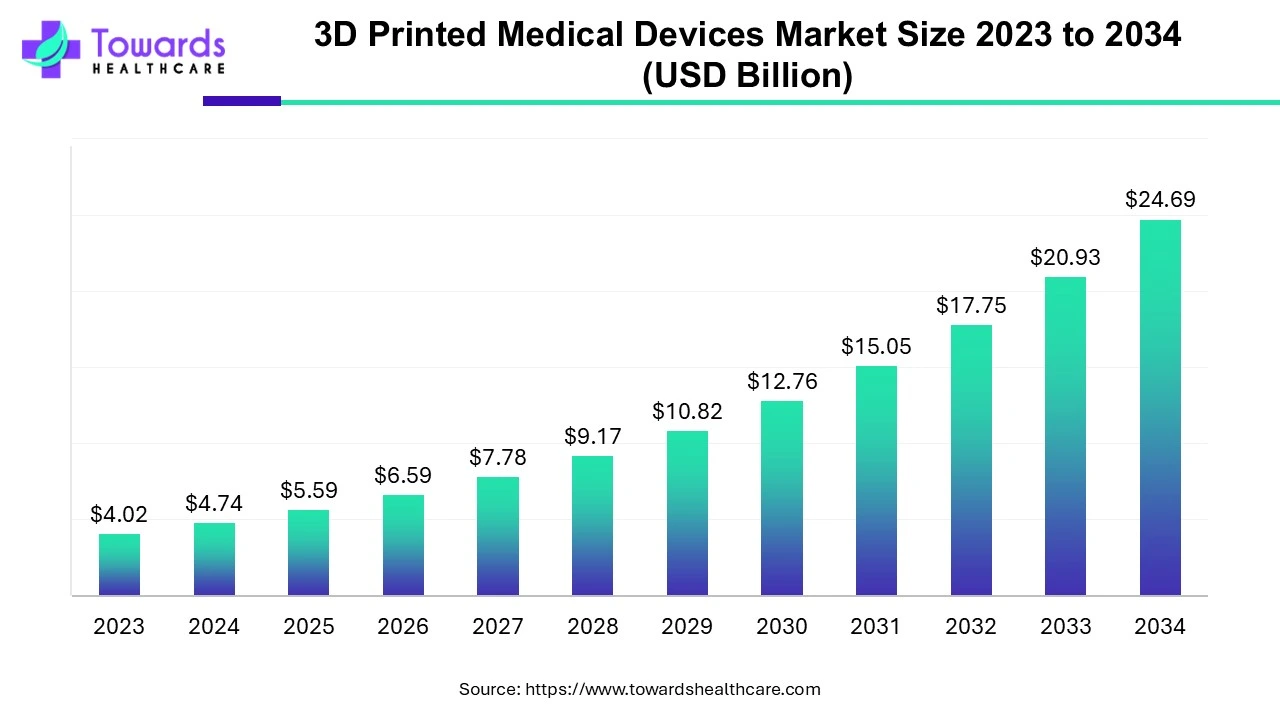
The 3D Printed medical devices market is anticipated to grow from USD 5.59 billion in 2025 to USD 24.69 billion by 2034, with a compound annual growth rate (CAGR) of 17.94% during the forecast period from 2025 to 2034, as a result of rising demand for customized 3D printed solutions.
Customization and Personalization in 3D Printed Medical Solutions are Causing Massive Growth in the Medch Field.
The solutions provided by the 3D printing industry cover a wide range of revolutionary medical and industrial applications. 3D printing has progressed from its beginnings as a prototyping technology to a feasible choice for end-use regions and larger-scale manufacturing applications. 3D printing has expanded beyond consumer markets in the past few years, developing itself as a commonly utilized commercial method of production. In the past two years industry developments have positioned the technology to move beyond its applications in effective prototyping and become an appropriate approach for end-use regions and serial manufacturing.
In January 2025, Axial3D secured $18.2 million in funding, with 57 Stars investing $10 million to become the top shareholder. The company focuses on AI-powered 3D medical imaging and printing for personalized surgeries. Recent milestones include FDA approval, a new Belfast facility, and backing from Stratasys. The funds will drive global growth and wider adoption of patient-specific care.
Artificial intelligence (AI) has been found to play a vital role in 3D printing as a powerful problem-solving tool. AI helps in designing novel and innovative medical devices, enhancing personalization of devices. AI introduces automation in medical device manufacturing through 3D printing, reducing human intervention. This results in decreased manual errors and increased reproducibility. AI-enabled predictive analytics aid in detecting potential errors, allowing manufacturers to take real-time decisions. AI also enhances quality assurance of 3D printing by validating designs for 3D printing suitability and minimizing waste. Moreover, AI can suggest process improvements, enhancing precision and decision-making capability. It also expedites the manufacturing process with faster product development cycles, thereby reducing time-to-market of a medical device.
The healthcare industry has experienced a revolutionary transformation with the advent of 3D printing technology. The healthcare industry has been ultimately impacted by the high cost of medical devices over the years, as traditional medical device manufacturing follows expensive and lengthy processes which accelerates the overall cost of production. On the other hand, 3D printing provides an efficient and cost-effective alternative in which medical devices are directly manufactured with the help of digital designs. This eliminates a number of labor-generated steps and complex processes involved in traditional manufacturing.
In order to create precise prototypes of medical devices as well as equipment and tools to streamline testing, more than 90% of the biggest medical device manufacturers use 3D printing. Cost savings of up to 70% were reported by one medical device manufacturer. In addition, according to a different medical research organization, using 3D-printed models in 10-15% of cases could result in annual savings of up to $1,750,000.
Custom-fit prosthetics are frequently expensive and only available to patients in developed nations who have insurance. To overcome these financial obstacles, prosthetics are increasingly utilizing the adaptable design of 3D printing. Making surgical instruments with 3D printing is an efficient and affordable way to manufacture in-demand medical supplies. Even for complicated instruments, it is effortless to alter designs for surgical requirements.
Furthermore, 3D printing offers customization and personalization which involves the creation of patient-specific medical devices. This reduces the need for costly replacements and improves patient outcomes. Surgery guides and implants that precisely fit a patient's anatomy are made using 3D printing. This lessens the possibility of issues, expensive modifications, and protracted hospital stays. By enabling on-demand and localized production, 3D printing eliminates the need for vast shipping and warehousing networks, which can be expensive.
3D Printed medical devices are usually produced in limited quantities, and patterns can be modified continually to reduce costs, weight, and component counts to produce small, lightweight medical devices with internal performance. Medical devices that are specifically designed for a patient enhance comfort, fit, and outcomes while lowering the risk of complications. With numerous applications in dental, interbody, surgical, orthopedic, and personalized devices, 3D printing earnings in the medical devices sector have been increasing continuously since 2018.
Custom implants and prosthetics can be made using 3D printing to better fit a patient's needs and increase comfort and functionality. Using surgical guides that were 3D printed, surgeons can perform complex procedures like spinal replacements and joint replacements with greater precision and accuracy, increasing their chances of success and speeding up their recovery. This leads to devices that are better matched to the patient and their particular anatomy.
For instance, 3D-printed personalized medical devices include:
In addition, the marginal cost of customization in 3D printing is another benefit included, as 3D printing allows end-to-end, automated, and scalable workflows for personalized products from right from designing to production. Furthermore, 3D printing allows the ultimate creation of biologically relevant structures. Such organic structures are unique and are impossible to manufacture with the help of traditional methods. Medical professionals can create unique-to-patient personalized implants using metal additive manufacturing techniques like SLM by using medical imaging data—from CT scans, for example—as the foundation for a CAD 3D model. The implant can be created using a non-toxic, biocompatible titanium 3D printing alloy (like Ti64).
Moreover, rising investments in 3D printing medical devices for the improved availability of customized products augment the market growth. For instance, in March 2023, the National Science Foundation (NSF)'s Leading Engineering for America's Prosperity, Health, and Infrastructure (LEAP-HI) program awarded the researchers a $2 million grant that will enable them to take on the challenge of designing and 3D printing smart devices using various materials. Together, they intended to create pediatric ventilation masks and other medical devices that can be customized and shaped differently while being used using 3D printing technology. On top of it, there is a lot more to come in the upcoming years as Many medical devices must be able to be customized, and being able to change their shape as the patient grows or heals is a game-changer. Finding a quick, affordable way to do that could greatly simplify life for both patients and medical professionals.
The FDA's Centre for Devices and Radiological Health regulates devices, the most prevalent type of 3D-printed product at the moment. Devices are divided into one of three regulatory categories, or classes. The FD&C Act and its implementing regulations found in Title 21 of the Code of Federal Regulations (CFR) establish regulatory controls for devices in the United States. In overall, businesses that produce, prepare, multiply, compound, assemble, or process medical devices for the benefit of humans, including 3D printing businesses, must register with the FDA and list the medical devices they produce, prepare, multiply, compound, assemble, or process on their registration form.
The FDA continues to uphold a custom device exemption. If a custom device satisfies the criteria outlined in Section 520(b) of the Federal Food, Drug, and Cosmetic Act, it may be exempt from 510(k) or prior to market submissions. For example, these specifications might state that the manufacturer can only produce five units of the device annually or that it must be made to treat a specific pathology or physiological condition that no other device on the domestic market is capable of treating. In 2017, the FDA issued recommendations on what details need to be included in applications for 3D-printed medical devices, including those that are patient-matched and include things like joint replacements and cranial implants.
3D printing provides customizable and affordable solutions for the making of anthropomorphic radiotherapy phantoms through in vitro methods. Such phantoms are significantly used for the treatment planning of cancer patients and make sure that innovative radiation procedures are subject to quality assurance (QA). These phantoms make use of 3D printing technology to allow preliminary confirmation of customized radiation doses and to lessen the adverse effects of ionizing radiation on nearby healthy tissues.
In addition, the increasing rate of commercial accessibility and availability especially in case of medical technology with the intervention of 3D printing, ave created extensive growth opportunities in the medical technology sector. 3D printing also provide opportunities for inexpensive producers of personalized medical devices for radiotherapy phantoms as well as radiotherapy devices consisting of bolus & compensators to attain customized dose distribution intended for electron beam shielding devices, irregular surfaces, brac, brachytherapy and several others. This further bolsters the extent of growth opportunities in the 3D-printed medical devices market.
Furthermore, market players can further expand their capacities and avail themselves of more profits in the field by establishing collaborations with other healthcare and technology market players.
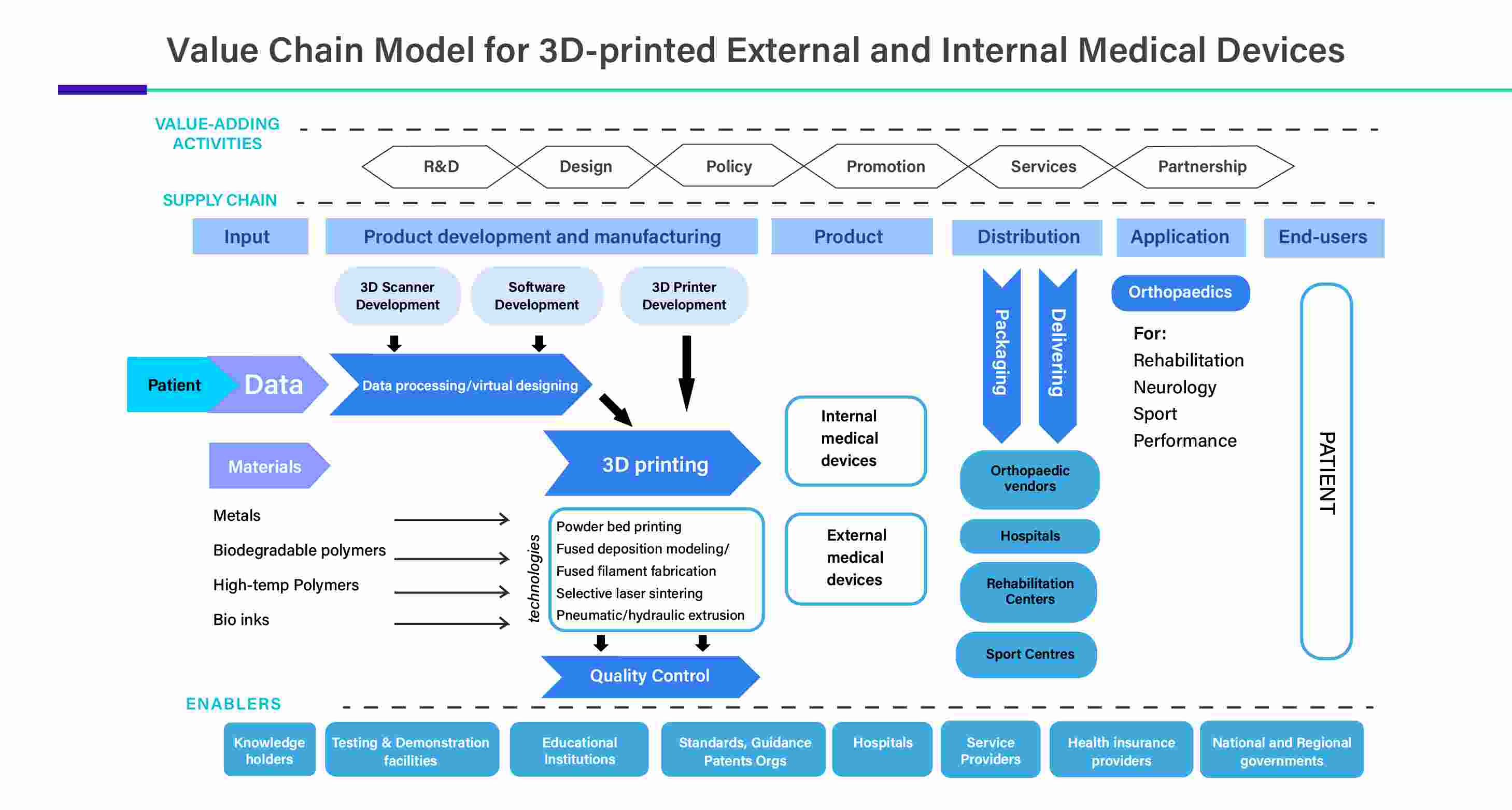
By type, the prosthetics segment held a dominant presence in the 3D printed medical devices market in 2024 and is predicted to witness significant growth in the market over the forecast period. Prosthetics are in high demand due to the rising prevalence of chronic disorders such as diabetes, vascular diseases, and traumatic incidents leading to amputations. It is estimated that approximately 185,000 people have an amputation annually, accounting for 300 to 500 amputations per day. The increasing demand for personalized prosthetics potentiates the need for 3D printing, augmenting the segment’s growth.
By component, the materials segment held the largest share of the market in 2024 and is projected to expand rapidly in the market in the coming years. The widespread adoption of 3D printing devices necessitates manufacturers to purchase materials for manufacturing medical devices. The type of materials to be used depends on the type of medical devices manufactured. Generally, polymers or thermoplastics like ABS or PLS are used for manufacturing medical devices. Other materials that are commonly used include engineered thermoplastics, resins, metals, and carbon fiber to maintain the strength and durability of medical devices.
By application, the implants segment led the global 3D printed medical devices market in 2024. Implants are used as prosthetics, to deliver drugs, monitor body functions, or provide support to organs and tissues. 3D printing is predominantly used for developing implants as their size and shape can be customized. The growing demand for personalized implants and treatment care boosts the segment’s growth. 3D-printed artificial skin can meet the needs of skin transplantation and reduce the pain of patients. Hence, 3D-printed implants offer superior benefits over conventional implants, propelling the segment’s growth.
North America dominated the global 3D printed medical devices market in 2024. The presence of key players and favorable regulatory frameworks drive the market. The Food and Drug Administration and Health Canada regulate the approval of 3D-printed medical devices in the U.S. and Canada, respectively. The U.S. FDA has approved more than 100 3D-printed medical devices. The increasing investments and collaborations also favor the development of medical devices using 3D printing. The Government of Canada recently announced an investment of over $20.8 million for job creation, expansion, and access to 3D printing.
Asia-Pacific is projected to host the fastest-growing market in the coming years. The increasing investments, collaborations, and mergers & acquisitions promote the development of 3D-printed medical devices. Favorable government policies related to environmental sustainability and indigenous manufacturing support the market. Since 3D-printed medical devices generate less waste, it has significantly less impact on the environment. Initiatives like “Made in China” and “Make in India” encourage manufacturers to develop 3D-printed medical devices in the respective nations. The Government of India aims to capture 5% of the global market share in 3D printing, contributing to $3 billion in GDP in the next 2-3 years.
The 3D printing medical device market in Europe is growing significantly due to a combination of factors. The region's advanced healthcare system and strong focus on innovation have created a favorable environment for adopting new technologies. Government support through funding initiatives and streamlined regulations has further accelerated this growth. Additionally, Europe’s aging population has increased the need for personalized medical devices, such as custom implants and prosthetics. The presence of top-tier research institutions and a rising number of health-tech startups also contributes to the region’s leadership in medical 3D printing, making it a key player in the global 3D-printed medical devices market.
North America held the largest share in the medical device gaskets & seals market in 2023. The advanced healthcare infrastructure, increased investments & collaborations, and the presence of key players drive the market. The market is also driven by the increasing number of surgeries in the region. It has been reported that more than one million surgeries are performed in Canada annually. The state-of-the-art research & development facilities promote the latest innovations in medical devices. Stringent regulatory bodies like the US FDA regulate the safety and effectiveness of medical devices in the region. In 2022, around 41 new medical devices were approved by the US FDA. The US is the largest medical device market globally, comprising over 40% of the global medtech market.
Asia-Pacific is anticipated to grow at the fastest rate in the market during the forecast period. The rising incidences of chronic disorders, increasing number of surgeries, favorable manufacturing infrastructure, and growing research & development activities drive the market. It has been reported that around 70% of the healthcare expenditure is on in-patient care, and more than 70% of the inpatient care spending is led by surgeries. It is estimated that around 30 million surgeries are performed in India annually. According to the World Bank Data, around 3,235 per 100,000 surgeries were performed in Korea in 2022. The market is also driven by favorable government policies and regulatory standards for medical devices. In 2023, China’s National Medical Products Administration (NMPA) received 13,260 applications for initial registrations, renewals, and changes in licensing items of Class III (Domestic and Overseas) and Class II (Overseas) medical devices, representing a 25.4% increase than 2022. Out of these, 12,213 applications were approved.
Europe is expected to grow at a considerable CAGR in the medical device gaskets and seals market in the upcoming period. The rising prevalence of chronic disorders potentiates the demand for medical devices. The increasing adoption of advanced technologies and the growing research activities propel the market. The rising collaboration among industry players promotes the development of gaskets and seals. Government organizations support the use of medical and wearable devices through funding and launching initiatives.
More than one quarter (26%) of people aged 16-74 years in the EU use smart wearables, including smartwatches, fitness bands, connected goggles or headsets, and safety trackers. Germany, France, the UK, and Italy are the top four largest countries for medical equipment in the EU. Public hospitals in Italy account for over 75% of medical device purchases, and the private sector accounts for 25%. Additionally, Europe exported 11 billion EUR worth of medical devices in 2023. The U.S., Russian Federation, Australia, India, Saudi Arabia, and Turkey are the top importers of medical devices from Europe. (Source: Medtech Europe)
Brain Garvey, Senior Vice President of Product Development at restor3d, commented that their biomechanics research and commitment to offer surgeons a market leading portfolio for patient-specific care leads to continuous advancements with the Kinos Total Ankle System. He also said that the option to select patient-specific articulation serves as the foundation for their forthcoming revision and fully personalized ankle implant systems.

By Type
By Component
By Application
By Region
March 2026
March 2026
March 2026
March 2026