January 2026

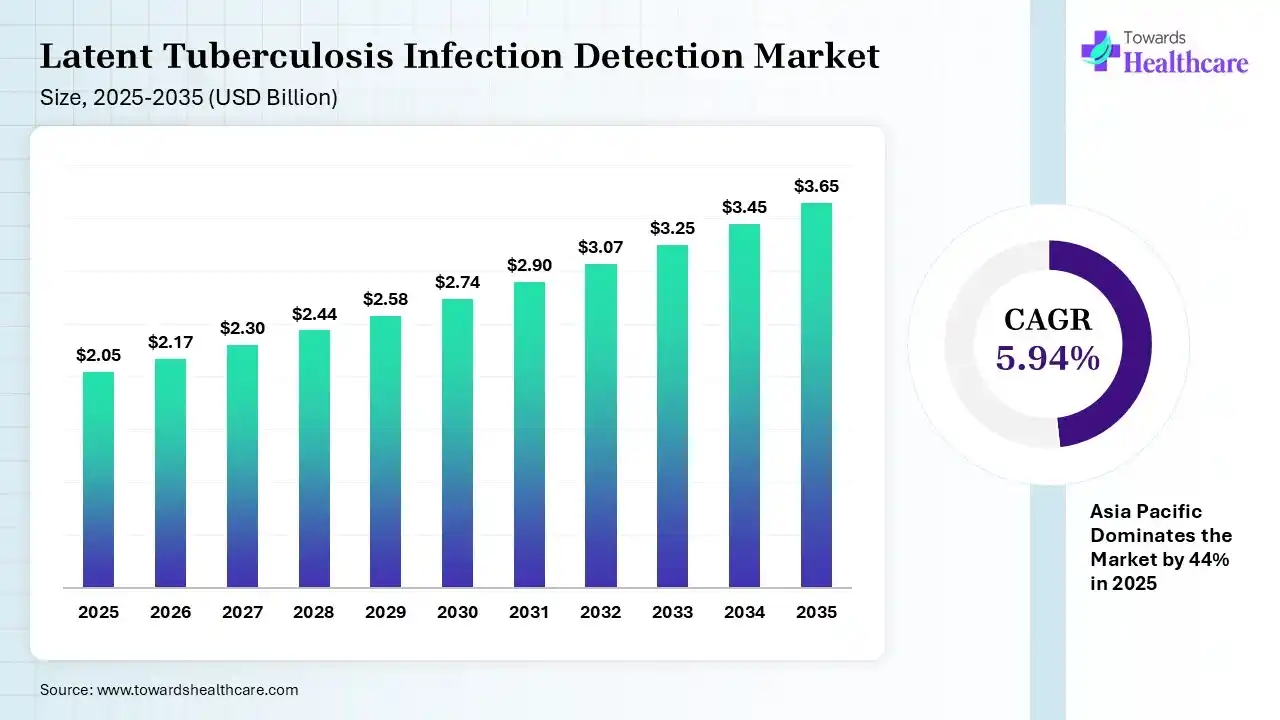
The global latent tuberculosis infection detection market size was estimated at USD 2.05 billion in 2025 and is predicted to increase from USD 2.17 billion in 2026 to approximately USD 3.65 billion by 2035, expanding at a CAGR of 5.94% from 2026 to 2035.
The latent tuberculosis infection detection market is growing due to this treatment allowing preventive treatment that stops the infection from emerging into active, and transmissible TB disease.
The latent tuberculosis infection detection market is growing, as treatment of LTBI is efficient in lowering the challenges of developing subsequent active TB disease. Advancements in present TB diagnostics are immediately required not only to enhance both sensitivity and specificity of Mtb infection detection but also to more precisely determine the challenges of progression or reactivation in the active disease. Treating patients with latent TB infection substantially lowers the risk that latent TB infection progresses to TB disease. The diagnosis of LTBI has always hinged on detecting immune responses to particular mycobacterial antigens, making the selection of these antigens significant for both identifying LTBI and emerging assays that differentiate among infection stages.
Integration of AI-driven technology in latent tuberculosis infection detection drives latent tuberculosis infection detection market growth, as AI-based technology transforms TB control via enhanced ACF processes and triage, improving detection rates in high-burden areas. With increasing precision, AI-based technology can diagnose pleural diseases and differentiate active TB from TB infection. ML diagnoses TB, monitors treatment, and analyzes host biomarkers. This technology provides promising services to improve early detection, increase accuracy, and optimize treatment approaches, thereby potentially transforming TB management globally. AI-based algorithms analyze chest X-rays to identify abnormalities indicative of TB, like nodules, consolidations, or cavities. AI-based technology offers quantitative assessments of TB severity and progression from chest X-ray images, aiding clinicians in making well-informed decisions related to treatment approaches.
Proteomic Advancements:
The application of proteomics technology provides a new methodology for the diagnosis of LTBI. This strategy involves the detection and identification of proteins and their interactions that are closely related to the advancement of disease.
Predictive Diagnostics:
Predictive diagnostics for latent tuberculosis infection (LTBI) enhance traditional technology by identifying individuals at the highest risk of progressing to active disease, rather than detecting prior exposure.
Advancement of Biomarkers:
LTBI-specific biomarkers support the identification of patients who are at increasing risk of developing TB and prepare them to adhere to preventive therapy, thus lowering the worldwide burden of TB.
| Table | Scope |
| Market Size in 2026 | USD 2.17 Billion |
| Projected Market Size in 2035 | USD 3.65 Billion |
| CAGR (2026 - 2035) | 5.94% |
| Leading Region | North America by 27% |
| Historical Data | 2020 - 2023 |
| Base Year | 2025 |
| Forecast Period | 2026 - 2035 |
| Measurable Values | USD Millions/Units/Volume |
| Market Segmentation | By Brand, By Test, By Application, By End Use, By Region |
| Top Key Players | QIAGEN, BIOMÉRIEUX, Oxford Immunotec, SD Biosensor, INC., Wantai BioPharm, Lionex GmbH |

Which Brand Segment Led the Market in 2025?
| Segment | Share 2025 (%) |
| QFT-Plus (QuantiFERON-TB Gold Plus) | 70% |
| Others | 30% |
Explanation
The QFT-plus segment held the major share of the latent tuberculosis infection detection market 70% in 2025 and is expected to grow at the highest rate during 2026-2035, as QFT-plus showed high performance in the identification of TB infection in patients, irrespective of their age or lower CD4 counts. QuantiFERON-TB Plus enhances sensitivity for active TB and preserves high specificity in the unvaccinated controls. QFT-Plus was developed to improve Mtb test sensitivity and diagnostic precision compared with the preceding generation.
Which Test Segment Led the Market in 2025?
| Segment | Share 2025 (%) |
| Tuberculin Skin Test (TST) | 60% |
| Interferon Gamma Release Assay (IGRA) | 40% |
Explanation
The tuberculin skin test segment was dominant in the latent tuberculosis infection detection market by 60% in 2025, as TST is a significant tool applied for active tuberculosis identification, with a moderate worldwide performance and accepted specificity and sensitivity. This test helps to find out if a patient is infected with TB germs. A tuberculin test is a skin test done to check the presence of TB-causing mycobacteria in a patient. It assesses the immune system's response to the antigen. This test supports early diagnosis and management of latent TB infection in high-risk patients who can develop active TB.
Interferon Gamma Released Assay:
Whereas the interferon gamma released assay segment is the fastest-growing in the market, a major advantage of IGRAs is the excellent performance of control by using the patient’s internal positive and negative control, lowering the rate of false-positive and false-negative outcomes of IGRA. IGRAs support lowering the number of patients who need preventive treatment, which is more significant in children.
Why PLHIV Segment Led the Market in 2025?
| Segment | Share 2025 (%) |
| PLHIV | 45% |
| Household Contacts with Pulmonary TB | 35% |
| Others | 20% |
Explanation
The PLHIV segment dominated the latent tuberculosis infection detection market by 45% in 2025, as management of latent tuberculosis infection lowers the challenges of active TB in HIV positive patients, particularly in those with a positive tuberculin skin test. The choice of regimen is based on factors like availability, expenses, adverse effects, adherence, and drug resistance. Patients living with HIV who have a positive test for LTBI benefit more from preventive treatment than those who have a negative LTBI test.
Household Contacts with Pulmonary TB
Whereas the household contacts with pulmonary TB segment is the fastest-growing in the market, as household contacts exposed to patients with pulmonary TB, in a diversity of settings, are at substantial risk of active TB and LTBI. Household contact screening of newly diagnosed TB index cases offers a modest yield of cases. Contact examination links to efficient interventions like treatment for TB disease and TPT, supports to prevent transmission, and enhances TB treatment results for contacts.
Why the Diagnostic Laboratories Segment Led the Market in 2025?
| Segmnet | Share 2025 (%) |
| Diagnostic Laboratories | 50% |
| Hospitals/Clinics | 35% |
| Academic & Research Institutions | 15% |
Explanation
The diagnostic laboratories segment held the major revenue of the latent tuberculosis infection detection market by 50% in 2025, as in diagnostic laboratories, detection and treatment of latent tuberculosis (TB) infection (LTBI) is a key component of TB control efforts in low-incidence settings. The major benefits are agreeing on targeted preventive treatment for those infected, which significantly lowers their risk of developing active tuberculosis.
Hospital & Clinics:
Whereas the hospital & clinics segment is estimated to be the fastest-growing in the market, as hospitals and clinics adopt well-developed technology that treats patients with latent TB infection, significantly lowering the challenges that latent TB infection progresses to TB disease. Advanced tools have evolved, including the interferon-gamma release assays, which more precisely diagnose LTBI. Targeted testing and management of LTBI play a significant role in TB removal from patients.
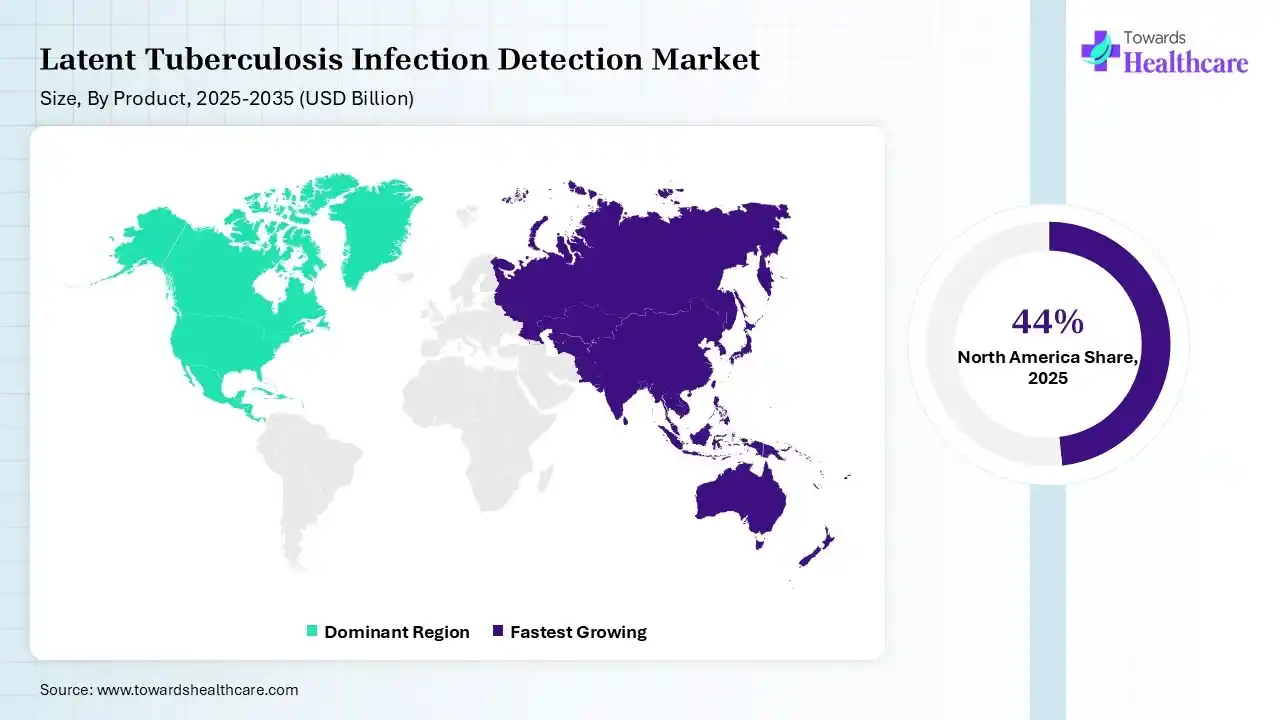
Asia Pacific dominated the latent tuberculosis infection detection market by 44% in 2025, as tuberculosis (TB) is the leading killer of the productive age group in developing countries. More than half of the cases in the world occur in the Asia-Pacific region. The increasing adoption of digital Chest X-rays (CXR) integrated with computer-aided detection (CAD) allows for fast, scalable, and precise screening. major population groups like migrants, miners, ethnic minorities, and indigenous populations, and other poor and remote communities, who have limited access to advanced healthcare and increasing burdens of TB, which contributes to the growth of the market.
India Market Trends
India remains to have the highest TB burden amid all countries, accounting for nearly a third of the incident TB cases and deaths worldwide. India has the strength to lead the world in advancing advanced TB diagnostics. Growing regulatory support, such as the Indian government's launch of the National Tuberculosis Elimination Program (NTEP), aims to eradicate TB. The novel validated TB diagnostic tests provide major key advantages, including rapid outcomes and significantly lower expenses.
North America is estimated to be the fastest-growing region in the latent tuberculosis infection detection market because targeted testing for tuberculosis has a strategy to diagnose and manage latent TB infection in patients who are at risk for emerging TB disease. Increasing focus on latent TB identification, regulatory funding initiatives, and hospital-based testing drives adoption. Strong public-private collaboration and a comprehensive reimbursement strategy drive the growth of the market.
U.S. Market Trends
In the U.S., the applications of digital diagnostic systems are becoming increasingly widespread, and teleradiology or telepathology are used for diagnosis. The quality of scanning, precise transmission of images, turnaround time, patient privacy, and availability of relevant previous images are paramount. Growing international travel and migration, associated with increasing TB cases in the immunocompromised populations, have intensified investigation efforts in the U.S.
Europe is significantly growing in the latent tuberculosis infection detection market, because many countries in Europe have attained a low incidence of TB, with most novel TB diagnoses occurring in migrants from high-incidence countries. Around 60 000 cases of TB are reported yearly in the European Union/European Economic Area (EU/EEA), with maximum cases in countries in the area characterised by low incidence of the disease.
R&D:
Manufacturing Processes:
Patient Services:

| Company | Headquarters | Latest Update |
| QIAGEN | Germany | QIAGEN announced a positive court conclusion, strengthening a significant patent for the QuantiFERON-TB testing technology. |
| BIOMÉRIEUX | France | bioMérieux is massively involved in both latent tuberculosis infection (LTBI) identification and the rapid characterization of drug-resistant tuberculosis. |
| Oxford Immunotec | United Kingdom | Oxford Immunotec continues to hold a significant position in the detection of latent tuberculosis infection (LTBI). |
| SD Biosensor, INC. | South Korea | In August 2025, SD Biosensor Inc. announced that its latent tuberculosis (TB) diagnostic product, STANDARD E TB-Feron ELISA, had been officially included in the World Health Organization (WHO) recommendation list for TB diagnostics. |
| Wantai BioPharm | China | Wantai's TB-IGRA CLIA is designed for application as an aid in the diagnosis of TB infection, including both latent TB infection and Tuberculosis disease. |
| Lionex GmbH | Germany | In November 2025, LIONEX recently launched a novel IGRA diagnostic test called LIOFeron®TB/LTBI for LTBI. |
Strengths
Weaknesses
Opportunities
Threats
By Brand
By Test
By Application
By End Use
By Region
January 2026
April 2026
January 2026
April 2026